RESEARCH ARTICLE
Serum Neutrophil Gelatinase-associated Lipocalin Measured at Admission to Predict Mortality in Sepsis-associated Acute Kidney Injury of Vietnamese Critically Ill Patients
Le V. Thang1, Pham N. H. Tuan5, Nguyen T. Kien1, *, Nguyen T. T. Dung1, Nguyen T. Tue1, Nguyen D. Duong1, Nguyen T. T. Ha1, Diem T. Van1, Nguyen V. Duc1, Vu X. Nghia2, Nguyen H. Dung3, Nguyen T. T. Huong4, Pham T. Dung1
Article Information
Identifiers and Pagination:
Year: 2018Volume: 11
First Page: 100
Last Page: 108
Publisher ID: TOUNJ-11-100
DOI: 10.2174/1874303X01811010100
Article History:
Received Date: 22/8/2018Revision Received Date: 28/11/2018
Acceptance Date: 06/12/2018
Electronic publication date: 31/12/2018
Collection year: 2018

open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: https://creativecommons.org/licenses/by/4.0/legalcode. This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose:
To evaluate incidence of sepsis-associated acute kidney injury (SA-AKI) in the AKI Intensive Care Unit (ICU) patients and predictive value of Neutrophil Gelatinase-Associated Lipocalin (NGAL) measured at the admission in mortality of SA-AKI and non SA-AKI.
Patients and Methods:
A study of 101 consecutive adult patients admitted to the Intensive Care Unit (ICU) diagnosed as AKI in which there were 60 patients with SA-AKI. Acute kidney injury was defined based on Acute Kidney Injury Network (AKIN) criteria. Serum NGAL was measured using the BioVendor Human Lipocalin-2/NGAL ELISA with blood sample taken at admission.
Results:
Incidence of septic acute kidney injury was 59.4%, incidence of death patients reached 20.0%. Mean concentration of serum NGAL in death group was 633.56 ng/ml, higher significantly than that of survival patients (328.84 ng/ml), p<0.005. Serum NGAL in non SA-AKI patients showed a better prognostic value to predict hospital mortality than that in SA-AKI patients (AUC: 0.894 and 0,807 respectively; p < 0.005)
Conclusion:
In SA-AKI patients, serum NGAL and mortality rate increased along with the stage of AKI. Serum NGAL, measuring at admission time, was a good prognostic biomarker of mortality in both SA-AKI and non SA-AKI patients.
1. INTRODUCTION
Acute Kidney Injury (AKI) influences over 13 million people and causes about 1.7 million deaths per year all over
the world [1-5]. There was about 30-60% of ICU patients suffering from AKI [6-12]. Sepsis is a major cause of AKI in ICU patients which is associated with high risk of hospital mortality [13-20]. Sepsis-associated acute kidney injury (SA-AKI) is usually called for acute kidney injury that is caused by sepsis. SA-AKI was diagnosed by clinical features, urinary output and serum creatinine. Neutrophil gelatinase-associated lipocalin (NGAL), which is a member of the lipocalin family of proteins, is expressed and secreted from renal tubular cells at low concentrations. High expression levels of NGAL can be induced by cell apoptosis after epithelial cell damage. A possible mechanism for this phenomenon may be that various factors stimulated renal tubular epithelial cells, leading to increased NGAL expression. NGAL can be absorbed by renal epithelial cells to regulate the expression of apoptosis-related proteins, which in turn promotes cell maturation and induces granulocyte apoptosis [9]. Several studies have shown that NGAL is produced in the kidney after ischemic or nephrotoxic injury [2, 13, 18, 19]. After kidney injury, NGAL can be detected in the plasma of patients with AKI within 2 h, and plasma NGAL levels peak approximately 6 h after injury. Increasing of serum NGAL was related to the number of patients who failed to recover renal function [21, 26]. So that, serum NGAL become an important biomarker for early detection of AKI [6-8], including SA-AKI. In this study, we tried to find out the role of serum NGAL in the prediction of mortality in SA-AKI as compared with non SA-AKI patients.
2. MATERIAL AND METHODS
2.1. Study Design and Setting
We studied prospectively 101 adult patients diagnosed as AKI admitted to the Intensive Care Unit (ICU). Approval for the conduct of the study was taken from the hospital ethics committee, and informed consent was obtained from the patient or surrogate decision maker. We excluded patients below 16 years of age, patients with chronic kidney disease, organ transplant recipients, malignancy, pregnant women and patients admitted for observation to stay in ICU for < 48 h.
Preexisting co-morbidities such as diabetes mellitus, hypertension, liver disease and medications were noted. Demographic data such as age, gender, comorbid conditions, clinical setting such as medical or surgical and presence of sepsis were noted. In our study, we used the Sequential Organ Failure Assessment (SOFA) score to define Single Organ Failure (SOF) and Multiple Organ Failure (MOF) [28]. SOF was defined when SOFA score was more than 3 in a particular organ at any time of hospitalization [27, 28]. MOF was defined when two or more organs failure was confirmed (Table 1) [17].
| Organ | Definition |
|---|---|
| Liver Failure | SOFA ≥3 (serum bilirubin ≥ 6 mg/dL) or Maddrey’s DF >32 |
| Renal Failure | Defined as AKI, according to the Acute Kidney Injury Network criteria |
| Nervous system failure | SOFA ≥3 (Glasgow Coma Scale ≤9) |
| Respiratory failure | SOFA ≥3 (PaO2/FiO2 <200 and mechanically ventilated) |
| Circulatory failure | SOFA ≥3 (use of norepinephrine, epinephrine, or dopamine [dopamine >5 μg/kg/min]) |
| Coagulation failure | SOFA ≥3 (platelet count <50,000/μL) |
2.2. Definitions
Acute kidney injury was defined based on Acute Kidney Injury Network (AKIN) criteria [16]. Patients were classified as having either SA-AKI or non SA-AKI and severity of AKI was graded as: (1) Stage 1 in proportion to 1.5 – 2 fold increasing of SCr level or an acute rise in SCr of more than 26.5 µmol/l (0.3 mg/dl) within 48 h and/or urine output <0.5 mL/kg/h (>6 h); (2) Stage 2 in proportion to 2 - 3 fold increasing of SCr level and/or urine output <0.5 mL/kg/h (>12 h); (3) Stage 3 in proportion to more than 3 fold increasing of SCr or SCr ≥ 354 µmol/l (4 mg/dl) and/or urine output <0.3 mL/kg/h (24 h) or anuria (12 h).
Sepsis in our study was defined as having manifestations of SIRS in combination with evidence of infection in the blood (expressed by positive bacterial cultures). SIRS was defined as having two or more of [1]:
- Temperature >38°C or <36°C;
- Heart rate >90/min;
- Respiratory rate >20/min or PaCO2 <32 mm Hg (4.3 kPa);
- White blood cell count >12 000/mm3 or <4000/mm3 or >10% immature bands.
Septic AKI was defined by AKI in the presence of sepsis without another significant contributing factor that might cause AKI, such as nephrotoxic drugs, poisoning, bleeding, hypovolemic circulatory.
2.3. Laboratory Measurements
Blood urea, SCr, serum electrolytes and other biochemical parameters of organ function were measured at admission. SCr was measured at admission time and daily thereafter. The steady level of creatinine at 4 weeks before admission was estimated to define baseline SCr. Otherwise, the admission value or the lowest SCr during hospitalization was used as a surrogate baseline.
Serum NGAL was measured by the BioVendor Human Lipocalin-2/NGAL ELISA kit based on sandwich enzyme immunoassay method. In the BioVendor Human Lipocalin-2/NGAL ELISA, standards, quality controls and samples are incubated in microplate wells pre-coated with polyclonal anti-human lipocalin-2 antibody. After one hour of incubation and washing, biotin labelled polyclonal anti-human lipocalin-2 antibody is added and incubated with captured lipocalin-2 for one hour. After another washing, streptavidin-HRP conjugate is added. After 30 minutes incubation and the last washing step, the remaining conjugate is allowed to react with the substrate solution (TMB). The reaction is stopped by the addition of acidic solution and absorbance of the resulting yellow product which is measured. The absorbance is proportional to the concentration of lipocalin-2. A standard curve is constructed by plotting absorbance values against concentrations of standards, and concentrations of unknown samples are determined using this standard curve.
2.4. Study Outcomes
The first outcome that we defined was the incidence of SA-AKI occurred during ICU stay, and the second outcome was the recovery, hospital mortality in both SA-AKI and non SA-AKI patients.
2.5. Statistical Analyses
All the continuous data were represented by mean and standard deviation and were analyzed by ANOVA and Student t-test. Categorical data were presented by frequency with percentage and were analyzed using Chi-square test. Receiver Operating Characteristic (ROC) curves with the Area Under the Curve (AUC) was calculated to predict mortality from SA-AKI and non SA-AKI patients. Multivariate regression analysis was performed to identify the predictors of hospital mortality. Statistical analysis was done using Statistical Package for Social Science (SPSS) version 17.1 (Chicago, IL, USA). A p value <0.05 was considered as significant.
3. RESULTS
When compared baseline demographic and laboratory characteristics in patients with and without SA-AKI (Table 2), we found that the mean patient age was 64.41 ± 17.59 years and 59.4% were male. Incidence of MOF patients in SA-AKI was significantly higher than that of non SA-AKI, p value < 0.005. There were no different of diabetes, hypertension and anuria proportion as well as the level of serum urea, creatinine and NGAL between SA-AKI and non SA-AKI patients with p > 0.05. The comparison of hospital outcomes which included duration of ICU stay and hospital mortality rate between SA-AKI and non-SA-AKI patients was established. The results showed no difference between SA-AKI and non-SA-AKI groups (Tables 2 & 3).
| Characteristic | All patients (n=101) | SA-AKI (n=60) | Non-SA-AKI (n=41) | p |
|---|---|---|---|---|
| Ages | 64.41 ± 17.59 | 66.68 ± 16.08 | 61.07 ± 19.31 | 0.116 |
| Number of male | 60 (59.4) | 31 (51.7) | 29 (70.7) | 0.055 |
| Diabetes (n,%) | 26 (25.7) | 17 (28.3) | 9 (22.0) | 0.471 |
| Hypertension (n,%) | 20 (19.8) | 12 (20) | 8 (19.5) | 0.952 |
| Urea(mmol/l) | 11.6 (8.1 – 17.55) |
11.3 (8.2 – 17.17) |
11.6 (8.05 – 22.15) |
0.732 |
| Serum creatinine (µmol/l) | 179 (151.5 – 248.5) |
176.5 (152.25 – 248.75) |
180 (151 – 252) |
0.863 |
| MOF (n,%) | 19 (18.8) | 18 (30) | 1 (2.4) | 0.001* |
| Anuria + Oliguria (n,%) | 36 (35.6) | 23 (38.3) | 13 (31.7) | 0.495 |
| Serum NGAL (ng/ml) | 403.78 (270.37 – 597.01) |
414.87 (273.16 – 601.84) |
379.2 (264.87 – 603.29) |
0.967 |
| – | All patients (n=101) | SA- AKI (n=60) | Non-SA-AKI (n=41) | p |
|---|---|---|---|---|
| Duration of ICU stay (days) | 9 (5 - 13.5) |
9 (5 – 16) |
8 (5 – 13) |
0.383 |
| Hospital mortality (n,%) | 20 (19.8) | 12 (20) | 8 (19.5) | 0.952 |
We also established the baseline laboratory characteristics and outcome in different stages of 60 SA-AKI patients Table 4. Serum urea, creatinine, NGAL concentration in patients with SA-AKI were significantly increased along with higher stage of SA-AKI with p < 0.001. The incidence of death in stage 1, stage 2 and stage 3 SA-AKI were 20%, 30% and 75% respectively.
| – | Stage 1 (n=42) | Stage 2 (n=14) | Stage 3 (n=4) | p |
|---|---|---|---|---|
| Ages | 67.19 ± 17.51 | 64.21 ± 11.24 | 70.00 ± 17.39 | 0.769 |
| Number of male | 20 (47.6) | 9 (64.3) | 2 (50) | 0.556 |
| Urea(mmol/l) | 9.2 (6.9 – 12.72) |
17.15 (9.5 – 21.32) |
32.45 (26.92 – 62.42) |
0.001* |
| Serum creatinine (µmol/l) | 159.5 (147 – 181.25) |
285.5 (248.75 – 323) |
469.5 (442.75 – 528.5) |
0.001* |
| Serum NGAL (ng/ml) | 315.19 (249.42 – 421.47) |
679.02 (601.84 – 770.48) |
806.64 (725.03 – 878.25) |
0.001* |
| Hospital Mortality (n,%) | 5 (11.9) | 4 (28.6) | 3 (75) | 0.007* |
The mean value of serum NGAL concentration (ng/ml) was significantly higher in patients who died compared to those who survived (633.56 compared with 328.84, p < 0.005) (Table 5). The result of logistic regression analysis to identify the independent risk factors for death showed that anuria and MOF were two factors that affected significantly to the proportion of death in SA-AKI patients (OR were 0.013 and 0.004 respectively, p < 0.05) (Table 6).
| – | Death (n=12) | Survival (n=48) | p |
|---|---|---|---|
| Urea(mmol/l) | 20.5 (9.85 – 30.62) |
9.8 (7.57 – 16.05) |
0.011* |
| Serum creatinine (µmol/l) | 255.5 (189.25 – 409.75) |
162 (150.25 – 211.5) |
0.001* |
| Serum NGAL (ng/ml) | 633.56 (426.45 – 794.12) |
328.84 (261.46 – 535.86) |
0.001* |
| Variable | Odds ratio | 95% Cl | p |
|---|---|---|---|
| Serum NGAL > cut-off value (351.59 ng/ml) | 0.005 | 0 – 0.521 | 0.996 |
| Age ≥60 | 0.007 | 0 – 1.409 | 0.067 |
| Sex (male) | 0.007 | 0 – 1.562 | 0.072 |
| Admission serum creatinine (µmol/l) | 1.011 | 0.994 – 1.028 | 0.213 |
| Anuria + Oliguria | 0.013 | 0 – 0.613 | 0.027* |
| MOF | 0.004 | 0 – 0.711 | 0.04* |
We used ROC curve model to determine the predictive value of serum NGAL to predict the hospital mortality of both SA-AKI and non SA-AKI patients. The Area Under the Curve (AUC) was 0.807 (p < 0.005) and cut-off value was 351.59 ng/ml in SA-AKI patients (Fig. 1). In non SA-AKI, the Area Under the Curve (AUC) was 0.894 (p < 0.005) and cut-off value was 631.75 ng/ml (Fig. 2).
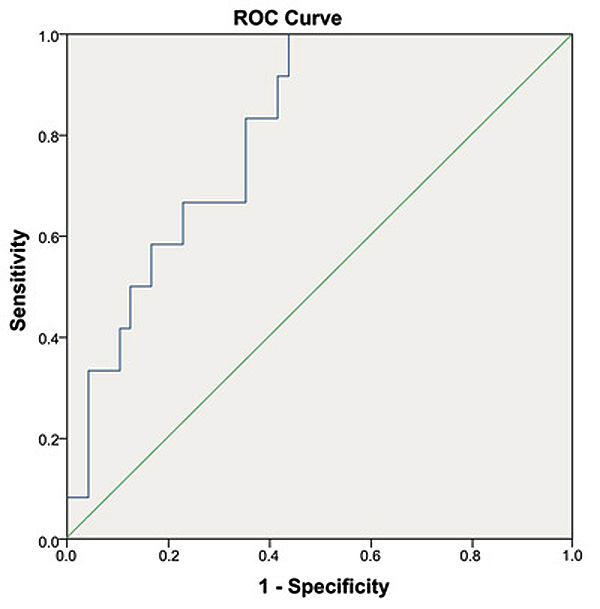 |
Fig. (1). Receiver Operating Characteristics (ROC) curves for prediction of hospital mortality of SA-AKI patients (serum-NGAL). |
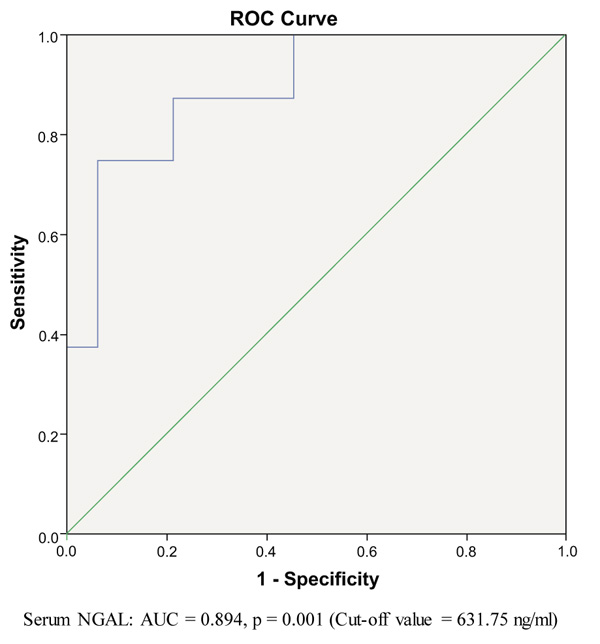 |
Fig. (2). Receiver Operating Characteristics (ROC) curves for prediction of death in non SA-AKI patients (serum-NGAL). |
4. DISCUSSION
In previous study, Wan L. et al. had shown that SA-AKI met in nearly 50% of AKI-ICU patients as well as affected from 15% to 20% of total ICU patients [29]. In our study, patient with SA-AKI was 59.4% of all AKI-ICU patients (60/101 patients). Our result was also similar to that of Bagshaw SM et al. [30], with 51.8% (43/83 patients). In septic patients, both hemodynamic and nonhemodynamic factors contribute separated important roles in the formation of AKI. The hemodynamic factors cause the loss of glomerular filtration rate while nonhemodynamic factors (immunologic, toxic, and inflammatory factors) influence through out the microvasculature and the tubular cells [29].
NGAL is a member of lipocalin superfamily which was first found in human neutrophils. Therefore, serum NGAL level in sepsis is likely to be affected by severity of infection. In our study, serum NGAL concentration in SA-AKI was higher than in non SA-AKI. This might be caused by high expression levels of NGAL can be induced by apoptotic cells after epithelial cell injury. A possible mechanism for this may be various factors stimulated tubular epithelial cells leading to the increasing of NGAL expression. NGAL can be absorbed by renal epithelial cells are regulated by the expression of apoptosis-related proteins, which in turn promotes cell maturation and induces granulocyte apoptosis.
Numerous studies had shown early diagnostic and predictive value of serum NGAL in patients with AKI. In fact, being the ICU doctors, it is very difficult for us to predict patient’s outcome to give patient the best treatment for reducing mortality rate and prolonged survival time purpose, especial with SA-AKI patients. In our study, we used patient’s blood samples immediately after admission to quantitative serum NGAL which was a bit different from some other studies. At the time of hospitalization, there was no intervention, so NGAL concentrations were the least affected. At the same time, we also recognized whether the diagnostic and prognostic value of serum NGAL differed between SA-AKI and non-SA-AKI patients. In our study, we used blood test to measure NGAL concentration instead of urine by two main reasons: Firstly, most of AKI patients, especially SA-AKI patients, suffer severe oliguria, so it was so difficult for us to give urine sample of patients at the time of admission; secondly, the urinary NGAL test results were most accurate when taken on a 24-hour urine sample, but our study was a cross-sectional study at the time of admission, so it was difficult to collect a 24-hour urine sample.
Septic AKI is the dominant problem in managing ICU patients. Although MOF and other complications are responsible for an increasing of mortality rate, AKI itself still contribute an important role in the increasing of morbidity and mortality in ICU patients. We had 19 patients (in total 101 patients) with MOF, in which 18 MOF’s patients in SA-AKI group and only one MOF’s patient in non-SA-AKI Table (2). We did not find any significantly differences about levels of serum creatinine, serum NGAL; or rate of patients with anuria and oliguria or duration of ICU stay, incidence of mortality (Table 3) between SA-AKI group and non-SA-AKI patients. From above results, we thought that SA-AKI patients with MOF related to high mortality rate in Vietnamese critically ill patients.
Serum NGAL had shown excellent performance on early diagnosis of AKI and prediction of clinical outcomes throughout the morbidity and mortality rate, dialysis requirement and severe complications [10]. Our results in table 4 showed that both serum urea, creatinine, NGAL concentration and hospital mortality rate in patients with SA-AKI were significantly increased along with higher stage of SA-AKI. There were about 75% of patients who suffered from stage 3 SA-AKI died while this proportion in stage 1 and stage 2 SA-AKI were about 20% and 30% respectively. Our results were similar to study of Peireira M et al in mortality rate of stage 1 and stage 2 SA-AKI, but our study had a higher proportion in mortality rate of stage 3 SA-AKI (75% compared with 48%) [22]. This might be caused by our limitation in sample size of this group.
When comparing some biochemical kidney function test, we realized that both serum urea, creatinine and NGAL concentration in death patients were significantly higher than in survival patients with p < 0.005. Our results in table 5 showed a link between the mortality of SA-AKI patients and the severity of AKI throughout renal function assessment. This helped us in prognosis for patients, especially those with high serum urea, creatinine and NGAL concentration, which might provide the best treatment for the patients and minimize the risk of death.
We established a multivariate logistic regression to indentify various factors that affected to hospital mortality rate in patients with SA-AKI. We realized that anuria and MOF were two factors that affected significantly to the proportion of death in SA-AKI patients (OR were 0.013 and 0.004 respectively, p < 0.05). Severe AKI is normally associated with a reduction of urine output. This might be caused by the severity of parenchymal structural changing including tubular injuries and tubular back-leakage may be greater in patients with anuric AKI than in patients with nonanuric AKI. Furthermore, anuria frequently occurs together with multi-organ failure that can make AKI becomes overwhelming.
Figs. (1 and 2) showed ROC curve for serum NGAL to predict the hospital mortality of SA-AKI and non SA-AKI patients. Serum NGAL in non SA-AKI patients showed a better prognostic value to predict hospital mortality than that in SA-AKI patients (AUC: 0.894 and 0,807 respectively; p < 0.005). That was an interesting result in our study. With this result, we could realize that serum NGAL level was influenced by various factors other than AKI condition. The inclusion of sepsis in AKI patients led to a variety of other disorders in the body that might affect the biosynthesis of serum NGAL as well as its prognostic value to predict hospital mortality. Therefore, further studies are necessary to assess whether factors affecting the predicted mortality of NGAL in SA-AKI patients.
We also realized some limitations in our study. In our study, SOFA scores were used for determining patients with multiple organ failure. However, it had only 19/101 patients with multiple organ failure in our study (Table 2), so we did not mention the association between SOFA score and NGAL level. Maybe in further study, we might mention this issue.
CONCLUSION
In conclusion, patient with SA-AKI was 59.4% of all AKI-ICU patients. In SA-AKI patients, serum NGAL and mortality rate increased along with the stage of AKI. Serum NGAL, measuring at admission time, was a good prognostic biomarker of mortality in both SA-AKI and non SA-AKI patients.
FUNDING
In this study, we had been strongly supported by clinical application funding of our local hospital and university to complete our research.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Ethical Committee of Vietnam Military Medical University (No. 2890/QĐ-HVQY).
HUMAN AND ANIMAL RIGHTS
Animals did not participate in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2008.
CONSENT FOR PUBLICATION
Informed consent was obtained from all the participants.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
In this study, we had been strongly supported by clinical application funding of our local hospital and university to complete our research.







