All published articles of this journal are available on ScienceDirect.
A Simplified Approach to the Management of Hypercalcemia
Abstract
Introduction:
While patients with hypercalcemia can be treated with various treatment options including volume expansion, loop diuretics, calcitonin, bisphosphonate, sensipar; hypercalcemia can be resistant to these measures.
Case Description:
In this report, we present two elderly patients with hypercalcemia resistant to traditional therapy. Both had parathyroid tumor. Early diagnosis could not be established. Both patients required hemodialysis. Hypercalcemia was controlled in the 74-year old who successfully underwent surgery with normalization of calcium level. The 79-year old was not considered a surgical candidate and was transitioned to hospice.
Discussion & Conclusion:
Primary hyperparathyroidism is an important cause of hypercalcemia. In order to reduce morbidity and mortality, its diagnosis must be established earlier on. These case reports prepared with residents/junior faculty will benefit other trainees with the diagnosis and management of refractory hypercalcemia and highlight a teaching point that prompt diagnosis of primary hyperparathyroidism can have a major positive impact on the overall management of hypercalcemia.
1. INTRODUCTION
Primary Hyperparathyroidism (PHP) results from an inappropriate overproduction of parathyroid hormone from parathyroid gland(s) and presents with hypercalcemia. It is the third most common endocrine disorder affecting 0.3% of the general population (1-3). Approximately 1–3% of post-menopausal women and a total population incidence of 21.6 cases per 100,000 person-years [1-3] have been reported. Majority of patients with hyperparathyroidism are asymptomatic and symptoms associated with hypercalcemia are absent. However, PHP patient can develop life-threatening symptoms, which require early diagnosis and prompt treatment to reduce mortality. In this report, we present the outcomes of two patients with hypercalcemia and discuss the treatment option adopted to manage their hypercalcemia.
2. CASE DESCRIPTION
2.1. Case No. 1
A 79-year-old female with past medical history of lung cancer status post lobectomy, insulin dependent diabetes mellitus, Stage IV chronic kidney disease, hypertension and cholecystectomy was admitted to the hospital for altered mental status and worsening lethargy from a rehabilitation facility. Laboratory tests ordered by the physicians at the rehabilitation center had revealed elevated serum calcium levels of 18 mg/dL and Parathyroid Hormone (PTH) levels of 1848 pg/mL. Sestamibi scan was performed, which revealed a positive uptake. The patient was diagnosed with primary hyperparathyroidism and was treated with aggressive normal saline hydration, sensipar, calcitonin and alendronate. However, the serum calcium level remained persistently elevated at 18 mg/dL. Denosumab (Xgeva) was added, which successfully reduced her calcium levels to 12 mg/dL. Two days later, she was admitted to the hospital again with increased lethargy and altered mental status. On physical examination, her vital signs were stable with blood pressure 124/78 mmHg, pulse 80 beats/minute, respiratory rate 16/minute, and afebrile. The patient was lethargic, barely arousable, with Glasgow Coma scale of 4/15. A detailed neurological examination could not be performed due to altered mental status. There was no cervical or axillary lymphadenopathy; thyroid gland was smooth, non-tender, and mobile with no masses. Cardiac examination revealed normal heart sounds. Laboratory investigation revealed (Table 1) corrected calcium 19.5 mg/dL, intact PTH 3231.2pg/mL, 25-hydroxy vitamin D 23.6 ng/dL, serum creatinine 3.47 mg/dL, blood urea nitrogen 44 mg/dL, urine calcium 19.1 mg/dL, urine creatinine <10 mg/dL. CT scan of the chest/abdomen/pelvis revealed a suspicious nodule of the parathyroid gland adjacent to the right lobe of the thyroid gland extending into the sternum. Further radiological imaging couldn't be performed due to overall medical instability. Patient was intubated for airway protection. Aggressive normal saline hydration along with two more dosages of sensipar and intravenous furosemide did not help Likewise, intermittent low calcium bath (2.0 mEq/L calcium dialysate bath) and continuous venovenous dialysis also failed to control serum calcium level. The patient was considered a poor candidate for surgery and was ultimately transitioned to inpatient hospice care.
| PATIENT #1 | PATIENT #2 | NORMAL RANGE | |
|---|---|---|---|
| Complete Blood Count | |||
| WBC | 20.4 | 5.0 | 4.5-11.0 K/uL |
| HEMOGLOBIN | 10.7 | 11.5 | 12.0-16.0 gm/dL |
| HEMATOCRIT | 31.5 | 35.4 | 35.0-48.0% |
| MCV | 92.9 | 84.3 | 80-100 fL |
| MCH | 31.6 | 27.4 | 25.0-35.0 PG |
| PLATELETS | 346 | 181 | 140-450 K/uL |
| Complete Metabolic Panel | |||
| SODIUM | 140 | 141 | 136-145mmol/L |
| POTASSIUM | 3.9 | 3.1 | 3.5-5.2 mmol/L |
| CHLORIDE | 106 | 105 | 96-110 mmol/L |
| BICARBONATE | 25 | 31 | 24-31 mmol/L |
| CORRECTED CALCIUM | 19.5 | 16.3 | |
| MAGNESIUM | 2.3 | 2.0 | 1.3-2.5 mg/dL |
| PHOSPHORUS | 5.5 | 2.3 | 2.5-4.6 mg/dL |
| BUN | 44 | 32 | 5-25 mg/dL |
| CREATININE | 3.47 | 2.23 | 0.44-1.00 mg/dL |
| eGFR | 13 | 29 | 60 ml/min |
| PTH | 3231.2 | 799.5 | 12-88 pg/ml |
| PTH related Peptide | Normal | 3 | 0.0-2.3 pmol/L |
| URINE CALCIUM | 19.9 | 8.5 | mg/dL |
2.2. Case No. 2
A 74-year-old male patient presented with slurred speech and dizziness for 5 days. He denied any abdominal pain, headaches, loss of consciousness, vision changes, facial or lower extremity weakness or seizures. His past medical history was significant for atrial fibrillation, chronic kidney disease stage IV (GFR 30 ml/min), gout and hypertension. There was no history of malignancy or hereditary disorders in the family. On physical examination, the patient was alert, awake and oriented with fluent speech and no focal neurological deficits except for impaired short term memory. Laboratory tests revealed (Table 1) corrected calcium of 16.3 mg/dL, phosphorus of 2.3 mg/dL, intact PTH 799.5 pg/mL, PTH related peptide of 3 pmol/L, Vitamin D-25OH of 29 ng/mL. Further workup revealed urine creatinine of 15.2 mg/dl, urine calcium of 8.5 mg/dL. The work up for Multiple Endocrine Neoplasia (MEN IIA) was negative. Parathyroid Sestamibi scan showed bilateral uptake in the parathyroid glands. The patient was started on intravenous normal saline, furosemide, and sensipar. He also received one dose of pamidronate. CT scan of the neck revealed a large suspicious mass on the right side of the neck, which anatomically correlated to a para tracheal lymph node. Due to persistent hypercalcemia, the patient received two sessions of hemodialysis with low calcium bath (2.0 mEq/L calcium dialysate bath) that normalized serum calcium at 9.4 mg/dL. Subsequently, the patient underwent parathyroidectomy and the suspicious mass was confirmed to be right inferior parathyroid gland extending into the thoracic cage. Following the procedure, the intact PTH levels improved to 106.4 pmol/L. The parathyroid mass biopsy revealed adenoma of parathyroid gland. During the hospital course, the patient maintained a stable calcium level of 8.6-8.8 mg/dL and was discharged in a stable condition with close endocrine follow-up.
3. DISCUSSION
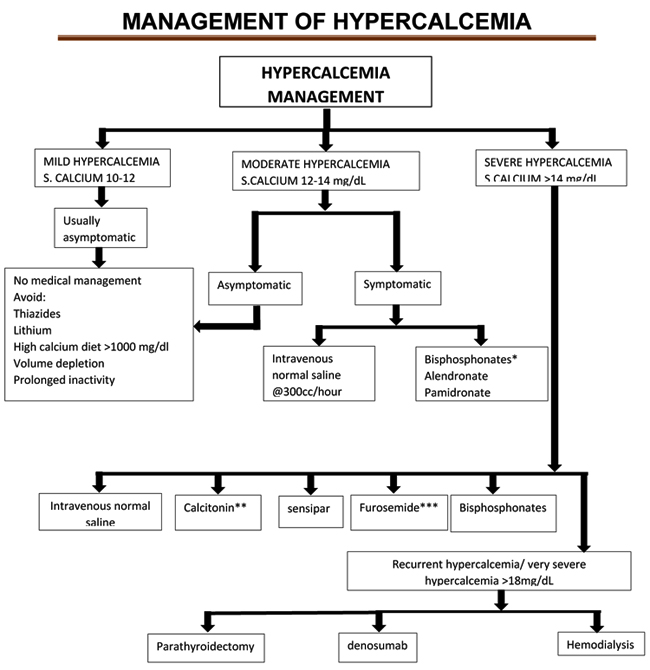
Patients with hypercalcemia can be treated with various treatment options including aggressive volume expansion (usually 1 to 2 L normal saline bolus followed by maintenance fluids at 100 to 150 mL/hour with a goal of urine output of 100 mL/hour and a decrease in serum calcium of 2 mg/dl/24 hours), loop diuretics (to excrete calcium) and calcitonin and bisphosphonate (to inhibit osteoclastic bone resorption) [7, 8]. Calcitonin is very effective in the first 72 hours of use due to tachyphylaxis effect [7, 11]. Due to its ability to control hypercalcemia and safety profile, bisphosphonates are the standard of care in the treatment of cancer-associated hypercalcemia [7]. It is worth mentioning that intravenous zoledronic acid and pamidronate are important bisphosphonates. They attain their maximum effect within 2-4 days. These agents are usually administered along with saline. The anti-PTH related peptide antibodies (Denosumab) are approved for hypercalcemia and are helpful in the management of bisphosphonate refractory hypercalcemia [12]. Finally, mithramycin (a specific inhibitor of bone resorption) can effectively lower serum calcium, however, it is reserved as the last resort treatment due to its toxic profile [7]. Management of hypercalcemia is outlined in Fig. (1).
The two patients presented in this paper demonstrated severe hypercalcemia that was refractory to the above-mentioned approaches. Severe hypercalcemia (serum calcium of >18mg/dL) that is refractory to the traditional measures may require dialysis [4-12]. Hemodialysis with low calcium bath can be very helpful to lower serum calcium quickly. While our case number 2 responded to hemodialysis therapy effectively, case number 1 failed to reduce serum calcium with intermittent hemodialysis or continuous venovenous hemodialysis.
Both cases demonstrated a parathyroid mass; high PTH, serum calcium and urinary calcium as well as a positive Sestamibi scan attesting to diagnosis of primary hyperparathyroidism. Patient number 2 was successfully treated with surgical removal of the adenoma. However, serum calcium could not be normalized in patient number 1. This patient was not considered a surgical candidate. All attempts, including dialysis therapy, failed to control serum calcium. This patient was referred to hospice [13].
Selective Percutaneous Ethanol Injection Therapy (PEIT) guided by color Doppler flow mapping is an effective and safe adjunct to medical therapy with a low risk for hypoparathyroidism, which is a viable option and has been well described in literature [14-20]. The patients who are resistant to medical therapy and are considered as high risk surgical candidates could be managed successfully by PEIT. In this technique, selected parathyroid gland with nodular hyperplasia are sclerosed following ethanol injection and subsequent enlargement or diffuse hyperplasia is controlled with vitamin D pulse therapy [16-22].
PEIT is now established in Japan as a treatment option for SHPT and its cost is covered by the National Health Insurance (NHI) System [21, 23]. According to new the version of clinical guidelines developed by Japanese Society for Parathyroid Intervention [21, 24] for selective direct injection therapy of the parathyroid glands in chronic dialysis patients are those who meet all three of the following criteria: 1) Difficult cases for continuous treatment, despite medical treatment, showing intact PTH ≥ 400pg/ml or hyperphosphatemia and/or hypercalcemia induced by treatment, 2) Enlarged parathyroid glands with suspected nodular hyperplasia on ultrasonography, 3) Patients who have given informed consent to undergo PEIT [21-24]. In a multicenter trial, the most commonly reported complications of PEIT are 1) possible recurrent nerve paralysis and 2) the presence of adhesions can make future surgical intervention complicated, making parathyroidectomy as the preferred initial surgical treatment [20]
To minimize the risk of adhesions and to prevent the recurrent nerve paralysis the recommended steps are: (1) only inject ethanol after confirming that the needle is definitely within the selected parathyroid gland; (2) limit the volume of ethanol to <80% of the volume of the parathyroid gland, (3) minimize the amount of ethanol used, by using Doppler flow mapping and (4) avoid simultaneous PEIT of the right and left parathyroid gland [21-24].
Though the result of PEIT is promising, it is also necessary to evaluate the result of parathyroidectomy following PEIT, the time for the development of adhesion after PEIT and the number of times PEIT can be repeated before the adhesions become too severe [22-25].
CONCLUSION
Hypercalcemia can be life threatening and requires immediate intervention. While traditional measures of controlling hypercalcemia are effective, primary hyperparathyroidism can be resistant. Surgical removal of the gland is the definitive therapy for these patients. However, not everyone is a surgical candidate. In such patient minimally invasive direct ethanol injection is a viable option.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Decleared none.


