All published articles of this journal are available on ScienceDirect.
Prevalence of Osteopenia and Osteoporosis among Chronic Kidney Disease Patients: A Systematic Review
Abstract
Background:
Complications of Chronic Kidney Disease (CKD) lead to low bone mass and osteoporosis.
Objective:
To review the prevalence of osteopenia and osteoporosis among CKD patients.
Methods:
Potential studies were identified through a systematic search of recognized databases using keywords “prevalence,” “osteopenia,” “osteoporosis,” “Chronic Kidney Disease,” “CKD patients,” “Chronic Renal Failure,” “CRF patients,” “Bone Mineral Density,” and “low bone mass” to identify relevant articles. An article was included in the review if the study met the following criteria: 1) conducted in CKD patients aged 18 years and above, 2) Osteopenia and osteoporosis were determined through Bone Mineral Density (BMD) testing, and 3) T scores were determined for all patients. The studies including transplant patients were not included in this review.
Results:
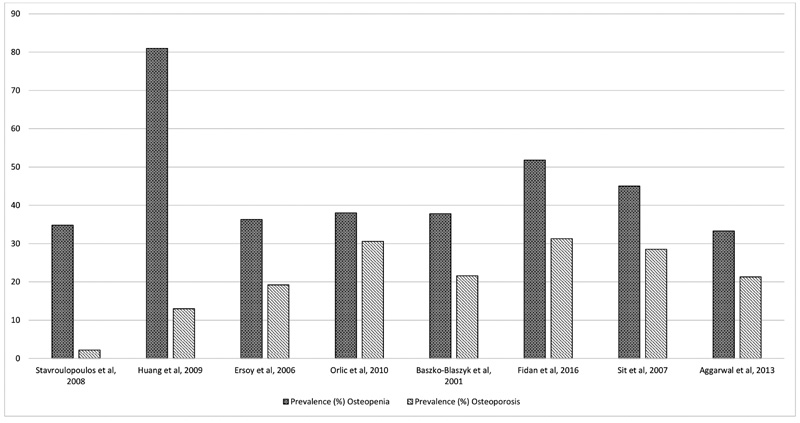
A total of 2142 titles were retrieved and of these 8 studies, based on the prevalence of osteopenia and osteoporosis in CKD patients were included in the final review. The review found that the prevalence of osteopenia reported in the studies was between 33.3% and 81% with an average of 45.91% and the prevalence of osteoporosis was between 2.24% and 31.3% with an average of 23.29%. Higher prevalence rates were observed among female gender and CKD patients with low Body Mass Index. Lumbar spine (LS) was found to be the most vulnerable site for osteoporosis among the majority of the studies.
Conclusion:
The review found a high prevalence rate of osteopenia and osteoporosis among CKD patients. The review further identified that BMD testing at the LS site is more reliable and suitable to determine the risk of fracture and osteoporosis.
1. INTRODUCTION
Chronic Kidney Disease (CKD) is defined as kidney damage or Glomerular Filtration Rate (GFR) less than 60 mL/min/1.73 m2 for 3 months or more, irrespective of the cause. Kidney damage in many kidney diseases can be ascertained by the presence of albuminuria; defined as the albumin-to-creatinine ratio of greater than 30 mg/g in two of three spot urine specimens. Kidney Disease: Improving Global Outcomes (KDIGO) has recommended the estimation of eGFR from calibrated serum creatinine and estimating equations, such as CKD-EPI formula or Modification of Diet in Renal Disease (MDRD) Study equation. Kidney disease severity is classified into five stages based on the level of GFR [1, 2].
The decline in kidney function due to CKD leads to reduced glomerular filtration rate, thus causing phosphorous excretion that develops hyperphosphatemia, which in turn results in low calcium levels and calcitriol formation. This hypocalcemia results in increased levels of parathyroid hormone which further pulls calcium from the bones making them weak with low bone mass developing renal osteodystrophy with a higher risk of fracture [3]. Changes in bone mineral metabolism and alterations in calcium and phosphate homeostasis occur early in the course of CKD and progress as kidney function declines [4]. Low bone mass is frequently seen in patients with CKD stage G5 on dialysis [5]. However, measurement of bone mineral density was not a common practice in the management of CKD G5 patients before the latest Kidney Disease Improving Global Outcomes (KDIGO) guidelines for Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD) which recommended that “ patients with CKD G3a–G5 with evidence of CKD-MBD and/or risk factors for osteoporosis should undergo BMD testing to assess fracture risk if results will impact treatment decisions” [6].
Osteoporosis has been operationally defined on the basis of Bone Mineral Density (BMD) assessment. According to the WHO criteria, the normal BMD value (the average value of young healthy women) is a T-score of -1.0 or above. The T-score values between -1.0 to -2.5 are termed as low bone density or osteopenia. Osteoporosis is defined as a BMD T-score value less than -2.5. The most widely used technique to measure BMD is Dual-energy X-ray Absorptiometry (DXA), and diagnostic criteria based on the T-score for BMD are a recommended entry criterion for the diagnosis and development of pharmaceutical interventions in osteoporosis [7].
It is generally known that impaired kidney function can lead to low bone mass and ultimatelyosteoporosis, thus becoming a risk factor for fracture but most of the research is only focused on CKD G5 patients on dialysis. The patterns and impact of low bone mass in patients with moderate to severe kidney failure are less studied. There is a variation in reported prevalence of osteoporosis and type of fractures among CKD patients of various stages. This review is aimed to study the prevalence of low bone mass including osteopenia and osteoporosis among CKD patients from stage G3a to stage G5.
2. MATERIALS AND METHODS
Studies were identified through a comprehensive literature search of databases including Scopus, PubMed, Medline, Science Direct and Google Scholar, and additional sources including the Cochrane Library from the inception of these sources until August 2019. The keywords used for searching relevant articles were “prevalence,” “osteopenia,” “osteoporosis,” “Chronic Kidney Disease,” “CKD patients,” “Chronic Renal Failure,” “CRF patients,” “Bone Mineral Density,” and “low bone mass.” Boolean operators such as ‘AND’ and ‘OR’ were used to increase the sensitivity and specificity of the search when needed.
The studies in which osteoporosis/osteopenia was not determined using a measure of BMD through Dual-energy X-ray Absorptiometry (DXA) were not included in the final review. Osteoporosis was defined according to WHO criteria, as a BMD T-score below 2.5 and osteopenia was defined as a T-score between –1.0 and –2.5. The articles identified were then screened based on the inclusion and exclusion criteria presented in Table 1.
Initial titles and abstracts were screened by MHT. The exclusion process, using the title/abstract, was also performed by MHT where the reason for exclusion was clear. In case of any uncertainty, the article was reviewed by SAS and any decision on excluding that article was made accordingly. Any disagreements on whether a study should be included/excluded were resolved through mutual discussion and consensus. Data were extracted from a full-text report using a data extraction form, which included study characteristics, participant characteristics, the prevalence of osteopenia and osteoporosis in CKD patients, and osteoporosis prevalence assessment methods. The primary outcomes of the current study were the prevalence rate of osteopenia and osteoporosis among CKD patients. The risk of bias including the quality of the studies for prevalence data was assessed using the Agency for Healthcare Research and Quality (AHRQ) checklist [8].
| Population | Chronic Renal Failure Patients Stage G3 to G5 |
|---|---|
| Phenomenon of interest | The phenomenon of interest included • Measure of bone mineral density in CKD patients • Prevalence of osteopenia and osteoporosis among CKD patients |
| Primary outcome measure | The outcome measures of interest included but were not restricted to the following: • Method used for studying the prevalence of osteoporosis in CKD patients • Impact of CKD stage and other factors on BMD levels • Difference in T score values for different sites among CKD patients |
| Types of studies | Quantitative cross-sectional / longitudinal clinical studies. Studies were included if they reported one or more of the outcomes detailed above |
| Exclusions | Following were excluded • Studies involving renal transplant patients • Studies not measuring bone mineral density • Studies not reporting the T score for BMD test • Studies not measuring both osteopenia and osteoporosis • Studies not following WHO criteria for osteoporosis using BMD T score values. • Studies including CKD patients having GFR values >60ml/min • Studies involving patient receiving medication known to influence bone and mineral metabolism such as corticosteroids, immunosuppressants, hormone replacement therapy, anticoagulants, lithium and anticonvulsants, etc. • Studies involving patients below 18 years of age |
| No. | Authors / Year/ Reference | Study Design | Country | Sample Size (n) | Prevalence (%) | Patient Characteristics | |
|---|---|---|---|---|---|---|---|
| Osteopenia | Osteoporosis | ||||||
| 1 | Stavroulopoulos et al., 2008 [9] | Prospective Cross-sectional study | UK | 89 | 34.83 | 2.24 | • CKD stage G3a and G4 non-dialysis patients • Mean age: 51 years |
| 2 | Huang et al., 2009 [10] | Prospective Cross-sectional correlation study | Taiwan | 63 | 81 | 13 | • ESRD hemodialysis patients • Mean age: 55.7 years |
| 3 | Ersoy et al., 2006 [11] | Prospective cross-sectional, multicenter study | Canada, Greece and Turkey | 292 | 36.3 | 19.2 | • ESRD peritoneal dialysis patients • Mean age: 56 years |
| 4 | Orlic et al., 2010 [12] | Prospective cross-sectional study | Croatia | 134 | 38 | 30.6 | • ESRD hemodialysis patients • Mean age: 56.4 years |
| 5 | Baszko-Blaszyk et al., 2001 [13] | Prospective cross-sectional study | Poland | 37 | 37.8 | 21.6 | • ESRD Peritoneal dialysis uremic patients • Mean age: 46.7 years |
| 6 | Fidan et al., 2016 [14] | Prospective cross-sectional study | Turkey | 83 | 51.8 | 31.3 | • CKD stage G3a to G5 non-dialysis patients • Mean age: 59.9 |
| 7 | Sit et al., 2007 [15] | Prospective cross-sectional study | Turkey | 70 | 45 | 28.55 | • ESRD Hemodialysis uremic patients • Mean age: 45years |
| 8 | Aggarwal et al., 2013 [16] | Prospective cross-sectional study | India | 75 | 33.3 | 21.3 | • CKD stage G3a to G5 pre-dialysis patients • Mean age: 51.7 |
3. RESULTS
A total of n=2142 titles were retrieved and of these 146 were removed due to duplication. The remaining articles were screened for inclusion and exclusion criteria and 1960 studies were excluded. The remaining 36 studies were given a thorough review and finally 8 original studies, meeting all inclusion criteria and fulfilling all primary outcome measures, were included in the final review Fig. (1) shows the flow of study identification. The types of study design and results are summarized in Table 2.
A total of 8 studies were identified from the literature, which investigated the prevalence of osteopenia and osteoporosis among CKD patients through the measure of BMD. All studies were based on prospective cross-sectional study design including one multicenter study. The studies were conducted in different geographic regions, including the developed countries with a well-developed healthcare system like the UK, to the developing countries like India. All studies utilized the DXA scan for the measure of BMD along with other biochemical markers. The studies differ in terms of sample size, ranging from the lowest sample size (n=37) to the highest sample size (n=292). All studies included patients from both genders and dialysis (n=5) as well as non-dialysis patients (n=3). Among the studies on dialysis patients, both hemodialysis (n=3), as well as peritoneal dialysis (n=2) studies, were included. The studies included end-stage renal disease (ESRD) dialysis patients (n=5), CKD patients from stage G3a to G4 (n=1) and CKD nondialysis patients from stage G3a to G5 (n=2). The mean age of the patients, included in these studies, varied from 45 to 59.9 years.
The prevalence rate for osteopenia varied from 33.3% to 81% with an average of 45.91%. The lowest prevalence rate of osteopenia was reported by Aggarwal et al., among pre-dialysis CKD patients of stage G3a to G5. The highest prevalence rate of osteopenia was reported by Huang et al., among ESRD hemodialysis patients. The prevalence rate for osteoporosis varied from 2.24% to 31.3% with an average of 23.29%. The lowest osteoporosis prevalence rate was reported by Stavroulopoulos et al., among CKD patients with moderate to severe kidney failure from stage G3a and G4. The highest osteoporosis prevalence rate was reported by Fidan et al, among CKD stage G3a to G5 nondialysis patients.
Stavroulopoulos et al. [9] reported a higher prevalence of osteopenia in the femur bone as compared to the radial bone. The study did not find any difference in BMD among patients with different levels of vitamin D or even between CKD stage G3a and G4 patients. Huang et al. [10] reported that men had a higher BMD and T-score than women in Lumbar Spine (LS), Femoral Neck (FN) and Total Hip (TH) regions. Overall, the prevalence rate of osteoporosis in women (21%) was higher than that in men (6%). The percentages of subjects with a low bone mass (including osteopenia and osteoporosis) in the LS, FN, TH, and overall were 46%, 73%, 49%, and 81%, respectively. The prevalence rates of osteoporosis at the same sites were 8%, 10%, 5%, and 13%, respectively. FN was found to be the most vulnerable site to low BMD including both osteopenia and osteoporosis. BMD was found to be negatively correlated with age and positively correlated with body weight.

Ersoy et al. [11] reported a higher prevalence of osteoporosis in the FN area (26%) in comparison to LS region (19.2%), but the LS had better agreements T-scores for the diagnosis of osteoporosis as compared to FN. The prevalence of osteoporosis was a bit higher in females (23.3%) than males (16.6%) but this difference was not statistically significant. Low body weight was also reported to be a contributing factor for osteoporosis. BMD among CKD patients was found to be independent of intact Parathyroid hormone (iPTH) levels. Orlic et al. [12] also reported higher osteoporosis prevalence rates in females and LS was the most prevalent site for osteoporosis. A positive correlation between age and BMD was also observed. BMI and BMD were also found to be positively correlated indicating better bone mass among patients with higher body weight. PTH was found to be negatively correlated in females but there was no association found among males. Similarly, other biochemical markers like serum calcium, phosphate, or urea were also non-significant to the osteoporosis prevalence.
Baszko-Blaszyk et al. [13] reported that females had a higher prevalence of osteoporosis but the difference from males was statistically insignificant. As reported by all previous studies, the prevalence of osteoporosis varied depending on the sites examined. Ultradistal part of the forearm yielded the highest prevalence (37.8%), followed by the FN and the total body (21.6% each). Measurements in the LS also yielded similar prevalence results for osteoporosis. Bone mass did not correlate with the duration of dialysis, serum calcium, phosphate, iPTH, creatinine, or urea concentrations. Fidan et. al [14] reported that females had a lower BMD score than males and the difference between genders was statistically significant. The prevalence of osteoporosis was higher in the LS region as compared to the FN region. There was no statistically significant difference in BMD among patients from CKD stage G3a to stage G5. Similarly, there was no correlation between BMD and PTH levels. Sit et al. [15] reported slightly low bone mass in males than females but the difference was statistically insignificant. The higher prevalence of osteoporosis was found in the LS region (47.1%) as compared to the FN region (10%). No statistically significant relationship was observed between BMD and biochemical markers of bone turnover including iPTH levels.
Aggarwal et al. [16] reported that the prevalence of osteoporosis increased from CKD stage G3a to stage G5. The difference in the BMD score among patients of different stages of CKD was statistically significant. Vitamin D levels had a statistically significant and positive correlation with BMD. Similarly, iPTH levels had a negative correlation with the BMD score (Fig. 2).
| ARHQ Methodology Checklist for Cross-sectional study (http://www.ncbi.nlm.nih.gov/books/NBK35156/) |
Stavroulopoulos et al. | Huang et al. | Ersoy et al. | Orlic et al. | Baszko-Blaszyk et al. | Fidan et al. | Sit et al. | Aggarwal et al. |
|---|---|---|---|---|---|---|---|---|
| 1. Define source of information (survey, record, review) | + | + | + | + | + | + | + | + |
| 2. List inclusion and exclusion criteria for exposed and unexposed subjects (cases and controls) or refer to previous publications | + | + | + | + | - | + | + | + |
| 3. Indicate time period used for identifying patients | - | + | - | - | - | + | - | - |
| 4. Indicate whether or not subjects were consecutive if not population-based | - | + | U | U | - | + | U | - |
| 5. Indicate if evaluators of subjective components of study were masked to other aspects of the status of the participants | + | + | - | + | + | + | - | + |
| 6. Describe any assessments undertaken for quality assurance purposes (e.g., test/retest of primary outcome measurements) | + | + | U | + | - | + | + | + |
| 7. Explain any patient exclusions from analysis | + | - | - | + | - | + | - | + |
| 8. Describe how confounding was assessed and/or controlled | + | + | + | + | + | + | + | + |
| 9. If applicable, explain how missing data were handled in the analysis | NA | NA | U | NA | - | + | NA | NA |
| 10. Summarize patient response rates and completeness of data collection | + | + | - | - | U | + | - | + |
| 11. Clarify what follow-up, if any, was expected and the percentage of patients for which incomplete data or follow-up was obtained | NA | + | NA | U | - | NA | NA | NA |
3.1. Quality of Included Studies
The risk of bias including quality of all incorporated studies was determined and found to vary within a narrow range but overall, the quality of all studies was satisfactory. Preferred Reporting Items for Systematic Review and Meta-analysis protocols (PRISMA) has defined the guidelines for systematic reviews and meta-analysis, which prefer the use of good quality studies to avoid the risk of bias and to enhance the strength of evidence of the review [17].
This review was based on observational prospective cross-sectional studies, which had properly defined the sources of information, quality assurance assessment parameters, the procedure of confounding assessment and summary of patient’s response rate and completion of data. The majority of the included studies had properly defined inclusion and exclusion criteria, the time period for identifying the patients and detailed methodologies if evaluators of subjective components of the study were masked to other aspects of the status of the participants. The summary of the quality of observational studies is presented in Table 3.
4. DISCUSSION
Bone Mineral Density (BMD) scan has been demonstrated previously to determine the risk of fracture in CKD patients with a risk of developing mineral bone disorders [18]. In the current study, we have reviewed the prevalence of low bone mass including osteopenia and osteoporosis among CKD patients. The exact diagnosis of osteoporosis can become a challenging task at times due to various factors including hormonal changes especially in females with age and other disease-related complexities [19]. With the development of BMD scan and various studies reporting its effectiveness, it has become relatively easier to determine bone disease, rate of bone loss and changes in bone mineral density over time [20]. All studies included in this search used BMD alone or in combination with X-rays, biochemical parameters, parathyroid hormone, etc. After the use of BMD for the diagnosis of osteoporosis in CKD patients, the risk of bias in diagnosis was minimized. A slight variation in sample size was observed among the included studies. The studies also differed due to the type of CKD patients varying from stage G3a to stage G5 and both dialysis as well as non-dialysis patients. Most of the studies were single centered except one study which involved participants from multiple countries and it reported the prevalence rate as 19.2% [11].
4.1. Prevalence of Osteopenia
All studies have used BMD testing for the measure of bone loss according to the WHO criteria based on the measure of BMD T-score, where osteopenia was defined as the T-score values between -1.0 to -2.5 and osteoporosis was defined as the T score values below -2.5. The prevalence rate for osteopenia varied from 33.3% to 81% with an average of 45.91%. The highest prevalence rate of osteopenia was reported by Huang et al. [10], in its study in Taiwan among 63 ESRD hemodialysis patients. Generally, a higher average prevalence rate (47.6%) was observed in studies including CKD G5 dialysis patients as compared to those studies including CKD G3a to G5 pre-dialysis patients. Almost all studies reported a higher prevalence among females as compared to males, which could be linked to the usual hormonal changes associated with a higher degree of bone loss among females [21]. The National Kidney Foundation in its latest guidelines on diagnosis and management of mineral bone disorders among CKD patients has also recommended BMD testing among CKD G3a – G5 patients to access the risk of fracture [22].
4.2. Prevalence of Osteoporosis
The prevalence of osteoporosis varied from 2.24% to 31.3% with an average of 23.29%. The lowest osteoporosis prevalence rate was reported in CKD G3a and G4 patients while the highest prevalence was reported among CKD G3a to G5 non-dialysis patients. Studies reported that osteoporosis prevalence was slightly lower among CKD G5 patients undergoing dialysis as compared to CKD G5 non-dialysis patients. Most of the studies reported a higher prevalence of osteoporosis among females except one study in which the difference between genders was statistically insignificant. LS and FN were the most common sites for BMD measurement, and the highest prevalence was found at the LS site in most of the studies. One study reported a higher prevalence at ultra-distal forearm site and the femur bone. The majority of studies agreed that the testing at the LS site should be conducted to determine the risk of fracture and osteoporosis. These findings are in line with previously reported literature suggesting testing at the LS site for long-term fracture risk assessment [23, 24].
4.3. Factors Affecting The Prevalence of Low Bone Mass
Chronic Kidney Disease (CKD) affects various biochemical parameters including calcium, phosphorous, vitamin D and parathyroid hormone [25]. Two studies determined the impact of age on BMD levels and both reported different results in terms of the association, one of the studies reported that increasing age leads to low bone mass [10], while the other study reported better bone mass among patients with higher age [12]. Both these studies included ESRD patients with similar age groups, therefore further studies are required to establish the association of age and bone mass among CKD G5 patients. The previously reported literature provides a positive association between age and bone loss [26]. Three studies reported the association of body weight or Body Mass Index (BMI) with the BMD, and among them, two studies provided a positive correlation between body weight and BMD [10, 11] which is in line with the available literature data [27]. The majority of included studies could not find any association of vitamin D levels on the BMD or bone loss among CKD patients which is against the previously reported literature suggesting a negative correlation between vitamin D and bone loss [28]. A recent review of the literature identified the significance of vitamin D beyond the regulation of bone metabolism and calcium-phosphorus homeostasis. Some more reviews recognized the role of vitamin D in gynecological and obstetric diseases and human fertility and reproduction [29-31].
Six studies determined the association of parathyroid hormone levels on BMD and among them, two studies reported a negative correlation between BMD and parathyroid hormone [12, 16]. This association of negative correlation was already reported in the literature [27]. Four studies could not find any such association and reported statistically non-significant differences in BMD scores among CKD patients with different levels of parathyroid hormones [11, 13-15]. This non-significant association contrary to the previous literature data could be due to the lower age group patients (i.e. mean age below 50 years) and few studies involving patients from CKD stage G3a and G4 as well. The association of BMD score and CKD stage was determined in three studies and most of them reported a non-significant association [9, 14] which could conclude that the BMD scores were independent of the CKD stage. These findings were also in line with previously reported studies focusing on the measure of such associations between CKD stage and BMD scores [32].
The review concluded that CKD patients from stage G3a to G5 should be routinely monitored for BMD through DXA scan as well as biochemical parameters affecting bone metabolism to control osteopenia and osteoporosis and to avoid the risk of spontaneous fracture. Further detailed studies on the prevalence and the factors contributing to low bone mass among CKD patients need to be conducted to report the exact associations to fill this gap in the literature.
5. STUDY LIMITATIONS
The study limitations include variation in sample size as well as type of patients since studies including both hemodialysis and peritoneal dialysis as well as non-dialysis patients were included in the review. Furthermore, patients from CKD stage G3a to G5 were included which might present some bias as well.
CONCLUSION
This review found a high prevalence of osteopenia and osteoporosis in CKD patients from G3a to G5. The review further identified that BMD testing at the LS site is more reliable and suitable to determine the risk of fracture and osteoporosis along with other biochemical markers of bone turnover. A higher prevalence of low bone mass among CKD G3a patients calls for early diagnosis and screening of such patients through BMD testing. The prevalence of osteoporosis and fracture can increase if due consideration is not given to diagnose CKD patients for BMD.
AUTHORS’ CONTRIBUTION
All authors contributed to data analysis, drafting and revision of the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
CONSENT FOR PUBLICATION
Not applicable
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGMENTS
The authors would like to thank the Institute of Postgraduate Studies (IPS) Universiti Sains Malaysia (USM) and USM fellowship program.


