All published articles of this journal are available on ScienceDirect.
Survival and Analysis of Predictors of Mortality in Patients Undergoing Hemodialysis in a Rural Hospital in Puducherry, India: A Retrospective Cohort Study
Abstract
Background:
Despite the advent of dialysis technology, the survival of patients on hemodialysis (HD) is not encouraging. There is little information available regarding the prognostic factors of HD, its population characteristics, or outcomes in India.
Objectives:
We attempted to describe the profile of patients on HD, estimate the mean days of their survival and to determine the various factors associated with their survival.
Materials and Methods:
It was a retrospective cohort study conducted on patients who were on maintenance HD, registered during the five-year period between 1st January 2014 and 31st December 2018 in a tertiary care teaching hospital in Rural Puducherry, South India. Demographic, hematological, biochemical, electrocardiographic, echocardiographic parameters and death during follow-up were extracted from the dialysis database after de-identification. The survival rate was calculated using Kaplan- Meir Curves and Cox Regression analysis was done to estimate the adjusted hazard ratio.
Results:
The median survival of the 229 eligible patients on HD was 819 days. Cardiac pathology accounted for 25.7% of those deaths and 33.9% was sudden death. Adjusted Cox proportional model showed the presence of Left Ventricular Hypertrophy (LVH) (HR: 78.1), increase in phosphorus (HR: 1.26), decrease in haemoglobin (HR: 0.78), and decrease in serum albumin (HR: 0.52) were significant bad prognostic factors.
Conclusion:
Screening for LVH, anemia, hypoaluminemia and hyperphosphatemia amongst HD patients and managing them to prevent these bad prognostic factors has the potential to improve their survival.
1. INTRODUCTION
The prevalence of Chronic Kidney Disease (CKD) and its evolution to End-Stage Renal Disease (ESRD) have been increasing steadily at global and national levels [1-3]. It was projected that in 2040 it would be the fifth leading cause of death worldwide [4, 5]. Hemodialysis (HD) is the major treatment modality for renal replacement therapy worldwide. Although maintenance dialysis methods have successfully improved the survival of patients with terminal uremia, mortality remains high [6]. Approximately 9-13% of patients on hemodialysis in India die within one year [7, 8]. The adjusted rates of all-cause mortality are 6.3 to 8.2 times greater for dialysis patients than for the general population [9].
Identifying the prognostic factors that are associated with the mortality of patients with HD certainly helps to impact on the survival of HD cohort [10]. Most of the studies related to the survival of patients with HD have come from the developed world [11]. Hence it is vital for the developing, underdeveloped and the poorer group of patients in the third world to have their own epidemiological studies pertaining to those geographical areas with their own unique disease patterns, etiologies, demographics and differing standards of care. However, there is a dearth of studies in the rural, impoverished patient sector. So we looked at the survival of our HD cohort with the following study objectives.
2. MATERIALS AND METHODS
2.1. Study Setting and Design
The study was carried out in the Nephrology department of a tertiary care teaching hospital located in rural Puducherry in South India. This is one of the largest dialysis centres offering maintenance HD in Puducherry catering mainly to the rural population of lower socio-economic status. Database of the HD unit maintains updated patient information. Nearly 1400 – 1500 patients per month undergo twice weekly intermittent maintenance HD. Everybody is usually dialyzed for the duration of four hours unless there is any compelling indication like fever or hypotension to reduce the dialysis time to three hours. Retrospective analytical cohort design was adopted to address the mentioned objectives.
2.2. Study Participants and Duration
Those patients who were on maintenance HD for chronic renal failure registered during the five year period between 1st January 2014 and 31st December 2018 were studied retrospectively. The patients who were 18 years of age and above were included and those patients who had incomplete information recorded in the Comprehensive Case Record (CCR) were excluded.
2.3. Study Variables and Data Abstraction
The CCR contains the demographic and clinical details of the patient which were recorded by the consultant during every visit of the patient. This information was regularly updated and maintained in the database of the HD unit. Data abstraction sheet was prepared based on the list of study variables required. The study parameters included were demographic details,hematological, biochemical, electrocardiographic and echocardiographic parameters. The data was extracted from the dialysis database and was verified from the individual CCR after deidentification by the trained unit clerk and entered in the data abstraction sheet. The outcome of interest was the time to death during the follow up period.
2.4. Statistical Analysis
The survival rate was calculated using Kaplan- Meir Curves, and were compared by Log- Rank Test. Cox Regression analysis was done to estimate the unadjusted and adjusted hazard ratio and its 95% confidence intervals amongst the variables studied. The hazard is actually the likelihood of experiencing an event of death, assuming that the patient has survived up to a point in time. Data was entered in EpiData Manager (Software version 4.2, EpiData Association, Odense, Denmark) and IBM SPSS Statistics for Windows, version 24.0 (Armonk, NY: IBM Corp.) was used for data analysis.
3. RESULTS
During the study, there were 264 patients registered for HD. Out of them, 35 had incomplete information on the study variables and hence were excluded from the study. Finally, 229 eligible HD patients were included, out of which 112 (48.9%) expired (outcome of interest) during the study period and 117 were alive. The majority of the participants (176(76.91% were males. Most of them had hypertension 148(64.4%) and/or diabetes mellitus as a comorbidity and 20(8.7%) had other forms of kidney pathology in form of pyelonephritis (n=6), chronic glomerulonephritis (n=8) and Autosomal Dominant Polycystic Kidney Disease (n=6). Left Ventricular Hypertrophy (LVH) was found in 139 (60.7%) participants. The distribution of comorbidities and LVH were statistically significant (p<0.001) between patients who died and survived (Table 1).
The mean (SD) age of all the patients included was 51.1 (12.5) years. The mean serum phosphorous, QTC interval, blood urea, URR, and KTv values were significantly higher and mean haemoglobin and serum albumin values were lower among patients who expired (Table 2). Of the 112 expired patients, the major cause of death was due to cardiac pathology 29 (25.7%) followed by sepsis 21 (18.6%). The other causes were cerebrovascular accident 8 (7%), gasto-intestinal bleed 6 (5.4%), brain tumor 3 (2.6%), respiratory failure 4 (3.6%) and hemoptysis 3 (2.6%). Sudden death was recorded in 38(33.9%) patients. The number of patients in this cohort who had a dialysis catheter were only 26.
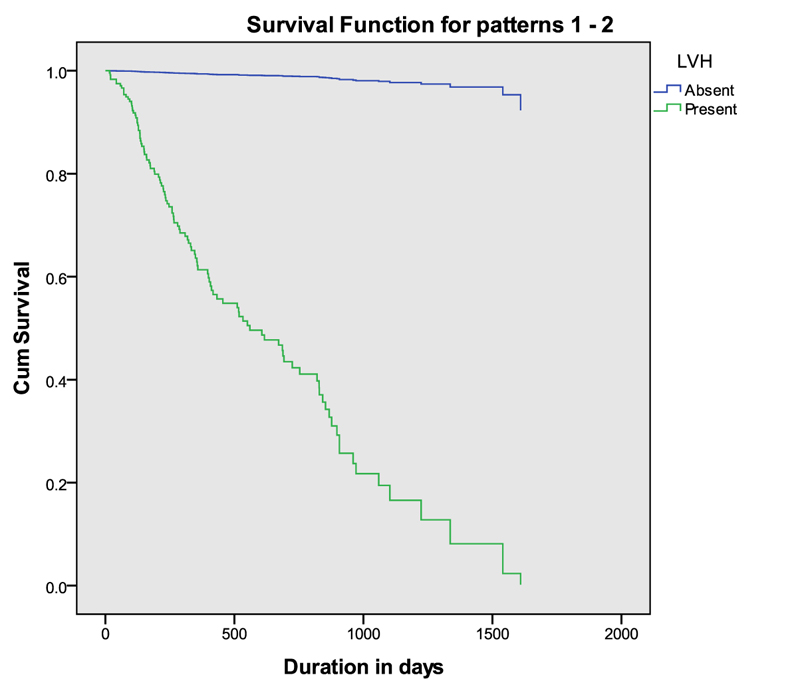
The Kaplan – Meir survival analysis showed that the estimated median survival was 819 days (95% CI: 750 – 1043) for the HD patients followed for five years. Unadjusted hazard ratios for death were estimated by Cox regression analysis using a proportional model. It showed that the presence of a comorbid condition and LVH, higher serum phosphorus, lower serum hemoglobin, prolonged QTC interval, higher blood urea, higher URR and KTv values, and lower serum albumin were statistically associated with the death of the HD patients. Adjusted Cox proportional model, including those variables that were statistically significant in the unadjusted model showed the presence of LVH [HR: 78.1 (95% CI: 10.8 – 565)], single unit increase in serum phosphorus [HR:1.26 (95% CI: 1.1 – 1.45)], single unit decrease in haemoglobin value [HR: 0.78 (95% CI: 0.69 – 0.87)], and single unit decrease in serum albumin [HR:0.52 (95% CI: 0.37 – 0.72)] were significantly associated with death during HD (Table 3). The survival curve separated by the status of left ventricular hypertrophy among patients undergoing chronic haemodialysis after adjusting for covariates is shown in Fig. (1).
| Characteristics | Survival Status, n (%) | P Value | |||
|---|---|---|---|---|---|
| Expired (n=112) |
Alive (n=117) |
Total n (%#) |
|||
| Gender | Male | 87 (49.4) | 89 (50.6) | 176 (76.9) | 0.77 |
| Female | 25 (47.2) | 28 (52.8) | 53 (23.1) | ||
| Co-morbidity | Unknown | 21 (35) | 39 (65) | 60 (26.2) | 0.04* |
| Hypertension and/or DM | 77 (52) | 71 (48) | 148 (64.6) | ||
| Other kidney pathology-(Chronic GN, Pyelonephritis,ADPKD) | 13 (65) | 7 (35) | 20 (8.7) | ||
| Snake bite cortical necrosis | - | 0 | 1 (0.5) | ||
| Left Ventricular Hypertrophy | Yes | 111 (79.9) | 28 (20.1) | 139 (60.7) | <0.001* |
| No | 1 (1.1) | 89 (98.9) | 90 (39.3) | ||
| Characteristics | Survival Status, Mean (SD) | P Value | |
|---|---|---|---|
| Expired (n=112) | Alive (n=117) | ||
| Age in years | 51.2 (12.9) | 50.9 (12.1) | 0.85 |
| Serum Phosphorus | 4.4 (1.3) | 4.0 (0.7) | 0.003* |
| Serum Calcium | 8.8 (0.8) | 8.7 (0.8) | 0.85 |
| QTC interval | 462.4 (35.2) | 401.1 (20.5) | <0.001* |
| Haemoglobin in g% | 6.6 (1.9) | 7.9 (1.6) | <0.001* |
| Serum Sodium | 134.5 (6.2) | 136.3 (7) | 0.05 |
| Serum Potassium | 4.9 (0.8) | 4.8 (0.9) | 0.21 |
| Blood Urea | 108.1 (34.1) | 98.8 (20.5) | 0.01* |
| Serum Creatinine | 6.7 (2.20 | 6.9 (1.9) | 0.47 |
| Urea removal rate | 56.0 (14.2) | 68.5 (6.9) | <0.001* |
| KT/v | 1 (0.4) | 1.3 (0.2) | <0.001* |
| Serum Albumin | 3.6 (0.6) | 4 (0.5) | <0.001* |
| Covariates | Coefficient | Standard Error | Hazard Ratio | P Value | 95% CI of HR | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | |||||||||
| Unadjusted Cox regression analysis | ||||||||||
| Gender | Male | 0.004 | 0.228 | 1.0 | 0.98 | 0.64 | 1.56 | |||
| Female | 1 (Reference) | |||||||||
| Co-morbidity | Unknown | 1 (Reference) | ||||||||
| Hypertension and/or DM | 0.535 | 0.248 | 1.7 | 0.03* | 1.1 | 2.8 | ||||
| Other kidney pathology –(Chronic GN, Pyelonephritis,ADPKD) | 0.934 | 0.356 | 2.5 | 0.009* | 1.3 | 5.1 | ||||
| LVH | No | 1 (Reference) | ||||||||
| Yes | 4.766 | 1.005 | 117 | <0.001* | 16.3 | 841 | ||||
| Age in years | 0.001 | 0.008 | 1.0 | 0.88 | 0.98 | 1.01 | ||||
| Serum Phosphorus | 0.354 | 0.081 | 1.4 | <0.001* | 1.2 | 1.7 | ||||
| Serum Calcium | 0.021 | 0.115 | 1.0 | 0.85 | 0.82 | 1.3 | ||||
| QTC interval | 0.020 | 0.002 | 1.02 | <0.001* | 1.02 | 1.02 | ||||
| Haemoglobin in g% | -0.335 | 0.054 | 0.71 | <0.001* | 0.64 | 0.79 | ||||
| Serum Sodium | -0.021 | 0.015 | 0.97 | 0.16 | 0.95 | 1.1 | ||||
| Serum Potassium | 0.102 | 0.101 | 1.12 | 0.31 | 0.91 | 1.4 | ||||
| Blood Urea | 0.008 | 0.003 | 1.01 | 0.02* | 1.01 | 1.01 | ||||
| Serum Creatinine | -0.053 | 0.046 | 0.95 | 0.25 | 0.86 | 1.1 | ||||
| Urea removal rate | -0.050 | 0.007 | 0.95 | <0.001* | 0.94 | 0.96 | ||||
| KTv | -1.951 | 0.256 | 0.14 | <0.001* | 0.09 | 0.23 | ||||
| Serum Albumin | -1.023 | 0.157 | 0.36 | <0.001* | 0.26 | 0.49 | ||||
| Adjusted Cox Regression Analysis | ||||||||||
|
Co- morbidity |
Unknown | 1 (Reference) | ||||||||
| Hypertension and/or DM | 0.096 | 0.262 | 1.1 | 0.71 | 0.66 | 1.83 | ||||
| Other kidney pathology (Chronic GN, Pyelonephritis,ADPKD) | 0.486 | 0.366 | 1.6 | 0.18 | 0.79 | 3.32 | ||||
| LVH | No | 1 (Reference) | ||||||||
| Yes | 4.359 | 1.009 | 78.1 | <0.001* | 10.8 | 565 | ||||
| Serum Phosphorus | 0.235 | 0.071 | 1.26 | 0.001* | 1.10 | 1.45 | ||||
| Haemoglobin in g% | -0.250 | 0.058 | 0.78 | <0.001* | 0.69 | 0.87 | ||||
| Urea removal rate | 0.013 | 0.043 | 1.01 | 0.75 | 0.93 | 1.10 | ||||
| KTv | -1.115 | 1.671 | 0.33 | 0.51 | 0.01 | 8.67 | ||||
| Serum Albumin | -0.659 | 0.174 | 0.52 | <0.001* | 0.37 | 0.72 | ||||
4. DISCUSSION
In the present study, 229 patients on HD for various chronic kidney diseases were followed retrospectively for five years and half of them died during the study period. Cardiac pathology accounted for one fourth of those deaths and one third were sudden deaths during HD. The Kaplan – Meir survival analysis showed that their median survival was 819 days. The median survival time is actually the median time on dialysis until death. Three-fourths of them were male and around 60% of them had hypertension and or diabetes mellitus as a comorbidity. Adjusted Cox proportional model showed the presence of LVH, a decrease in haemoglobin and serum albumin and an increase in serum phosphorus were significantly associated with death during HD.
Median survival was 819 days in our study, whereas a study in Ghana showed it as 90 days and in Ethiopia it was 263 days [12, 13]. There could be various factors associated with the five year survival of HD patients. Those factors are further discussed below. It was found that gender was not significantly associated with survival in our study, that was similar to a study done in the UK [14]. However, few previous studies showed that the mortality was higher in males [15-17]. Hypertension and diabetes mellitus were associated with poor prognosis. However, after adjusting for confounders, they became statistically insignificant in the current study. Studies in other countries also found that diabetic patients do more badly on HD [18]. On the contrary, an Indian study reported no significant survival difference between non-diabetics and diabetics.
Cardiac diseases account for major cause of mortality in our study and that was similar to the finding of a previous study [19]. Patients with LVH had a significantly higher probability of death and the same finding was reported in many other studies as well [20, 21]. LVH has been described as one of the major factors which lead to sudden cardiac deaths in the general population and more so in the dialysis population, especially in the presence of cardiac disease [22]. In hemodialysis patients, ACE inhibitors play a key role in the regression of LVH, independent of blood pressure changes. Hence screening for LVH and treating them with ACE inhibitors may actually help to improve survival amongst HD patients.
Serum Hb%, albumin and phosphate levels were an independent predictor of mortality in the current study. In alignment with our study, previous studies also revealed that higher serum phosphorus values were a bad prognostic marker [23-25]. In order to improve survival among HD patients, close attention needs to be paid to these relevant factors also, when they come for follow-up. In an earlier French study, where they looked at aortic pulse wave velocity and aortic stiffness and survival in hemodialysis patients, it was demonstrated that ACE inhibitors play a key role in the regression of LVH and had a favorable effect on the survival of patients in hemodialysis independent of blood pressure changes [26]. This is an important observation supporting the role of ACE Inhibitors in hemodialysis patients to cause regression of LVH in light of these results, as shown in our study.
Infections are an important cause of mortality on dialysis in most dialysis units and our study also showed that infection was the second commonest cause of mortality and cardiovascular causes are the commonest, unlike US studies which showed that infection was the commonest cause of death [27]. This is important as our study shows the significance of serum albumin levels, as lower serum albumin levels are directly linked to immunity and mortality.
A survival analysis conducted in Indonesia showed a significant difference in survival on dialysis based on the presence of insurance status compared to non-insured groups (p-value<0.05 [28]. This factor became redundant in our study as almost all patients were covered by insurance given by the government or other private insurance providers. This goes to show that it is not merely a patient factor that is impacting the survival of these patients but also the characteristics of the physician’s treatment modalities and those of the institutions concerned.
A Canadian study has demonstrated that there is a lower risk of mortality in hemodialysis care in private, not-for-profit centers when compared with care in private for-profit centers. When using a random-effects model, the pooled estimate showed that there was a higher risk of dying associated with private, for-profit dialysis center (relative risk, 1.08; 95% confidence interval, 1.04-1.13; P<.001) [29]. Our rural hemodialysis centre offers a purely non-profit model with free dialysis and free food being offered to our patients using government schemes.
A study from Macedonia indicated that death actually is the most severe result of inadequate dialysis and may in fact be used as an indicator of dialysis adequacy [30]. Hence, measures to improve the treatment factors may help in improving survival. Yet another study from Philippines discussed developing a probability risk factor of death in ESRD patients generated by Cox regression and Weibull distribution at a certain age to help us to improve the quality of medications and methodology to help lessen the risk of death in ESRD patients [31].
4.1. Strength and Limitations
Our study had more strength and few limitations. Firstly the cohort study design was appropriate to identify the factors associated with five year survival of HD patients. Secondly, the robust Cox regression analysis was applied for the same. Thirdly, the sample size was fairly large enough to fulfill the assumptions of statistical tests and the statistical power. Fourthly, the study variables were clinically relevant based on the review of literature. Finally, unlike the previous studies, we captured even the early mortality events and hence was a better index of actual mortality rate. Retrospective nature of the design was adopted to save time and resources, however, there was missing information in the CCR, which resulted in a reduction in the sample size. As there were many comorbidities, we grouped them into fewer categories and hence the exact quantification of the hazard of each one of them could not be estimated. The limitations of this study are that too many variables could not be considered in the Cox proportional hazard model applied and some of these covariates were dependent and modifiable. The issue of under dialysis impacting on survival is another relevant factor. This is an important factor impinging on the survival of hemodialysis patients as most of our patients undergo twice weekly dialysis sessions. This problem has been addressed at length in an Iranian study where the authors clearly infer higher mortality with dialysis sessions if carried out less than thrice a week [32]. The results of our study are generalizable to other hemodialysis patients in and around rural Pondicherry. As this is a highy selective group of patients, it is not possible to generalize the results of this study to other groups of selected patients. As no children were included in this study (below 18 years), the results are not applicable to children in general.
5. THE FUTURE
As there were sudden deaths captured in the data gathered, we need to address the causative factors for the same. More focus also needs to be placed on acquired Long QT syndrome / arrhythmogenicity and channelopathies in future studies [ 33-38].
There has been a concept that the physician–case load ratio impacts directly on survival, as shown in urban dialysis centers, implying that the nephrologist with the lowest case load had lower mortality rates, offered higher dialysis doses, had been giving longer dialysis sessions and received more kidney transplants [39]. This needs to be looked into further.
Quantization of comorbidities and approximation to survival in hemodialysis studies [40] especially using modern computational models using Artificial Intelligence, are presently lacking in the field of nephrology and need to be studied in depth urgently.
About the issue of the set of patients who discontinued the treatment at their center, not much information is not available for their outcomes or even if they entered into another dialysis programme. It seems that about 10% of deaths on chronic hemodialysis programmes are preceded by stopping dialysis [41].
The NECOSAD study group-Netherlands co-operative study has developed a prognostic model tool that helps in picking up those dialysis patients who would be more likely to have poor health risk and combines survival, quality of life and morbidity [42].
CONCLUSION AND RECOMMENDATIONS
The median year of survival of HD patients was 2.3 years and LVH, lesser haemoglobin levels, lower serum albumin and higher phosphorus were the independent, harmful prognostic factors identified. Screening for LVH, anemia, hypoalbuminemia and hyperphosphatemia amongst HD patients and managing them to prevent these bad prognostic factors has the potential to improve their survival. Future research on the predictors of sudden death, quantification of comorbidities and the utility of acquired long QT syndrome to improve survival are required.
AUTHORS' CONTRIBUTIONS
PRK was responsible for developing the concept, planning, collecting the data, maintaining database, writing the paper, proofreading, revising and review of literature. VV was responsible for writing of the paper,analysis of the data,revising the paper and proofreading. AD was responsible for planning, data analysis, revising the paper and proof reading. PD was responsible for the planning, data analysis, revising the paper and proofreading of the paper.
LIST OF ABBREVIATIONS
| HD | = Hemodialysis |
| CKD | = Chronic Kidney Disease |
| CCR | = Comprehensive Case Record |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the SMVMCH Research Committee and Institutional Ethics Committee (Ref / IEC/: 36/2017).
HUMAN AND ANIMAL RIGHTS
No animals were used for studies that are the basis of this research. This research was conducted on humans and are in accordance with the Helsinki Declaration of 1975, as revised in 2013 (http://ethics.iit.edu/ecodes/node/3931).
CONSENT FOR PUBLICATION
Informed consent was obtained from all participants.
STANDARDS FOR REPORTING
STROBE guidelines and methodology were followed while writing this manuscript.
AVAILABILITY OF DATA AND MATERIALS
All available data can be obtained by contacting the corresponding author [P.K]. All individual participant data collected during the study is available after deidentification, beginning 3 months and ending 5 years following article publication.
FUNDING
None.
CONFLICT OF INTEREST
The authors do not have any other conflicts of interest to declare.
ACKNOWLEDGEMENTS
We would like to thank the management of SMVMCH for allowing us to conduct the study in the hemodialysis unit of SMVCH.


