All published articles of this journal are available on ScienceDirect.
Aminophylline as an Adjunct to Ureteroscopic Lithotripsy: A Systematic Review of Local and Intravenous Use
Abstract
Introduction
Aminophylline, a non-selective phosphodiesterase inhibitor with smooth muscle relaxant effects, is established in pulmonary medicine but has not been systematically reviewed as an adjunct in ureteroscopic lithotripsy. This review assesses its efficacy and safety when administered either intravesically or intravenously.
Methods
PubMed, Cochrane Library, EBSCO, and Google Scholar were searched to June 2025. Eligible studies were Randomized Controlled Trials (RCTs) or semi-experimental trials assessing aminophylline during ureteroscopic lithotripsy. Outcomes included Stone-Free Rate (SFR), operative time, ureteral access, stent requirement, pain, hematuria, Catheter-Related Bladder Discomfort (CRBD), and adverse events. Risk of bias was assessed with RoB 2.0 for RCTs and ROBINS-I for non-RCTs.
Results
Eight studies (596 patients) were included. Intravesical aminophylline consistently improved ureteral access and reduced operative time; Lubana et al. showed higher success (82% vs 44%, p = 0.002) and less stenting (38% vs 68%, p = 0.011). Shabayek and Saafan reported reductions in intraureteral pressure, hematuria, and pain. Intravenous aminophylline showed mixed results: Barzegarnezhad found higher SFR (97.6% vs 84%), Ghadyan reported lower procedural success but less stenting, while Rehab et al. observed reduced CRBD (26% vs 59%, p = 0.025) and analgesic use. Adverse events were mild and transient.
Discussion
Evidence is limited by small samples, heterogeneous designs, and variable definitions of “success.” Effect sizes may vary with stone size, location, and lithotripsy modality. Sensitivity analysis was not feasible, lowering certainty.
Conclusion
Aminophylline may be a safe, low-cost adjunct to ureteroscopic lithotripsy, enhancing access, reducing operative time, and lowering morbidity. However, evidence remains low-certainty and hypothesis-generating, warranting larger multicenter RCTs.
1. INTRODUCTION
Urolithiasis is a prevalent condition in urological practice, with global prevalence ranging between 1% and 20% depending on geographic region [1-3]. In Southeast Asia, the incidence continues to rise, and men are more frequently affected than women [1]. When stones migrate into the ureter, they can cause acute obstruction and renal colic, necessitating intervention. Available management options include shock wave lithotripsy (SWL), percutaneous approaches, open ureterolithotomy, and ureteroscopy (URS) [3].
URS has become one of the most commonly performed procedures for both upper and lower ureteral stones. However, it is associated with complications in 9–25% of cases, including ureteral injury, bleeding, infection, and access failure from ureteral spasm or a narrow intramural ureter [4, 5]. Primary access failure occurs in 1–37% of unstented patients, and while pre-stenting can overcome this problem, it requires an additional procedure and may cause stent-related morbidity in up to 80% of patients [5, 6]. These limitations highlight the need for safe and effective pharmacologic adjuvants.
Alpha-blockers are commonly prescribed to facilitate ureteral access and improve outcomes of URS, but their therapeutic effect requires several days [7-9]. In contrast, aminophylline, a non-selective adenosine receptor antagonist, induces rapid smooth muscle relaxation within minutes by increasing intracellular cAMP/cGMP and reducing calcium influx [10-12]. It has long been used for renal colic, and intravesical instillation relaxes the distal ureter within 5 minutes, whereas alpha-blockers may take up to one week to achieve similar relaxation [7, 13, 14].
Despite its established role in pulmonology, the use of aminophylline in urolithiasis remains underexplored. Preliminary trials suggest potential benefits in reducing operative time, perioperative pain, hematuria, and stent requirement while improving stone-free rates. However, existing evidence is limited to small randomized and semi-experimental studies, with heterogeneous designs and outcome definitions. To date, no systematic review has synthesized this body of evidence.
The aim of this review was therefore to evaluate the efficacy and safety of aminophylline, administered locally or intravenously, as an adjunct to ureteroscopic lithotripsy in patients with ureteral stones.
2. MATERIAL AND METHOD
2.1. Protocol Registration and Literature Search
This review was prospectively registered in PROSPERO (CRD42024583664). The methodology adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines [15]. A comprehensive literature search was performed across PubMed, Cochrane Library, EBSCO, and Google Scholar, from database inception up to June 2025. The search strategy combined Medical Subject Headings (MeSH) and free-text keywords, including: (“Aminophylline” OR “aminophyllin” OR “xanthine”) AND (“ureteral stones” OR “ureteral calculi” OR “ureteral calculus” OR “renal colic” OR “renal colic pain”) AND (“ureteroscopy” OR “ureteroscopic” OR “ureteroscopic lithotripsy” OR “transureteral lithotripsy”).
Reference lists of eligible studies and relevant reviews were also screened to identify additional publications. No language restrictions were applied. Articles published in non-English languages were included after translation into English using a two-step process: preliminary translation by software (Google Translate/DeepL), followed by verification and correction by two independent reviewers with medical backgrounds to ensure accuracy of scientific and clinical terms.
2.2. Eligibility Criteria
Studies were eligible if they: (1) included patients with ureterolithiasis undergoing ureteroscopic lithotripsy (URS/TUL), (2) evaluated aminophylline administered locally or intravenously, and (3) were randomized controlled trials or observational studies. Exclusion criteria were case reports, editorials, studies with insufficient outcome data, and articles without accessible full text.
2.3. Study Selection
Two reviewers independently screened all titles and abstracts, with full texts retrieved for potentially eligible articles. Disagreements were resolved by consensus or third-party adjudication. For duplicate or overlapping populations, the most recent or complete report was included. The study selection process is shown in the PRISMA flow diagram (Fig. 1).
2.4. Data Extraction
Data extraction was performed independently by two reviewers using a standardized form. Extracted variables included study characteristics (author, year, country, design, sample size), participant demographics, exclusion criteria, intervention (dose, route, timing), comparator, stone size and location, type of lithotripsy, and reported outcomes. Outcomes of interest were stone-free rate (SFR) or procedural success, operative time, need for ureteral stenting, hematuria, postoperative pain, catheter-related bladder discomfort (CRBD), and adverse events.
All included procedures were performed under either spinal or general anesthesia, consistent with standard urologic surgical protocols. Intravesical aminophylline was commonly administered as 250 mg (10 mL) diluted in 150 mL of normal saline, instilled 5 minutes before ureteroscopy, while intravenous doses ranged from 250 mg in 100–250 mL saline to 4 mg/kg infused pre- or intraoperatively.
Procedural success was defined as complete stone removal or stone-free status achieved intraoperatively, as reported by each study. For consistency, outcomes were grouped by domain (stone-free rate, operative time, stent requirement, postoperative pain, hematuria, and adverse events).
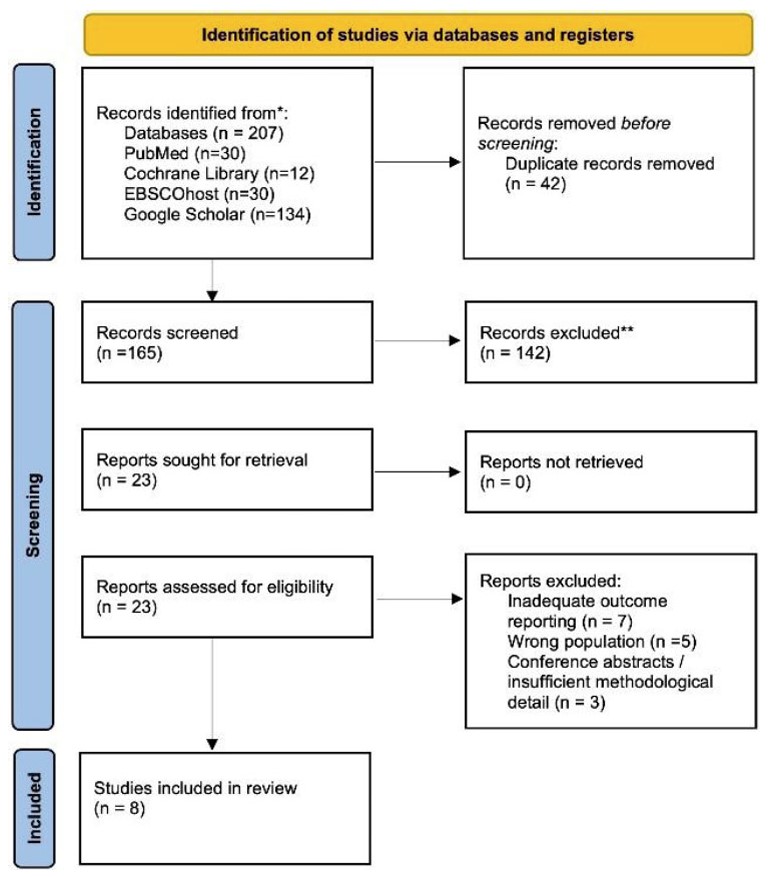
PRISMA 2020 flowchart.
2.5. Quality Assessment
The Cochrane Risk of Bias 2.0 tool was applied for randomized controlled trials, assessing domains such as randomization process, deviations from intended interventions, missing data, outcome measurement, and selective reporting [16]. Non-randomized studies were evaluated using the ROBINS-I tool. Assessments were conducted independently by two reviewers, with consensus reached by discussion [17].
2.6. Data Synthesis
Because of variations in study design, interventions, and outcome reporting, a meta-analysis was not performed. Instead, results were summarized narratively and structured according to the route of aminophylline administration (local vs intravenous). Outcomes were grouped by domain, and quantitative effect sizes with p-values were presented as reported in the original studies.
The complete electronic search strings and standardized data-extraction form are provided in the Supplementary Appendix to enhance transparency and reproducibility.
3. RESULTS
3.1. Study Selection
A total of 207 records were retrieved from four major databases: PubMed (n=30), Cochrane Library (n=13), EBSCOhost (n=30), and Google Scholar (n=134). After removing duplicates, 165 articles remained for screening. Of these, 120 were excluded based on title and abstract review due to irrelevance to aminophylline or ureteroscopic lithotripsy. The remaining 45 full-text articles were assessed for eligibility.
Following detailed evaluation, 38 articles were excluded for the following reasons: studies involving the wrong population (n=15), irrelevant intervention (n=11), and insufficient outcome reporting (n=12). Ultimately, eight studies fulfilled the inclusion criteria and were included in this systematic review. The selection process is illustrated in Fig. (1) (PRISMA flow diagram).
3.2. Study Characteristics
Eight studies with a total of 596 patients were included, consisting of five RCTs, two double-blind RCTs, and one semi-experimental trial from Iran, Pakistan, Egypt, and India. Aminophylline was administered either locally (intravesical/instillation) or intravenously. Definitions of procedural success varied across studies, ranging from stone-free rate (SFR) on imaging, intraoperative clearance, to ease of ureteral access (Table 1).
The characteristics and outcomes of the included studies are summarized in Table 1.
3.3. Local Aminophylline Instillation
Barzegarnezhad et al. [18-20] defined success as ureteroscope passage and complete lithotripsy, reporting higher rates in the aminophylline group (95% vs 71.6%, p=0.071). Khan et al. [21] defined success as complete stone removal, finding significantly higher success (88.9% vs 71.1%, p=0.03) and shorter operative time (39.9 vs 48.7 minutes, p<0.001). Saafan et al. [22] compared intravesical aminophylline with balloon dilatation. While SFR was similar (87.8% vs 88.1%), aminophylline significantly reduced intraureteral pressure (12.2 → 7.8 mmHg, p<0.001), postoperative pain, and hematuria (p<0.05). Shabayek et al. [14] also showed comparable SFR between groups (87.8% vs 88.1%, p=0.968), but lower hematuria (19.5% vs 42.9%, p=0.022) and pain (9.8% vs 28.6%, p=0.030) with aminophylline.
Lubana et al. [13] found significant improvement in access and success (82% vs 44%, p=0.002), shorter operative time (33.5 vs 54.6 minutes, p<0.001), and reduced DJ stent requirement (38% vs 68%, p=0.011).
3.4. Intravenous Aminophylline Administration
Barzegarnezhad et al. [19] assessed SFR after intravenous aminophylline, reporting higher rates (97.6% vs 84%) and shorter operative times (p<0.05). Ghadian et al. [20], in a semi-experimental trial, defined procedural success as stone removal without auxiliary intervention. They found success only in 38% of aminophylline patients, with longer operative time (36.2 vs 18.4 minutes, p<0.001), but reduced DJ stenting (36% vs 74%). Rehab et al. (2024) focused on catheter-related bladder discomfort (CRBD) rather than SFR, showing reduced CRBD (26% vs 59%, p=0.025), lower VAS scores in the first six postoperative hours (p<0.05), and decreased opioid/ketorolac requirements. Mild adverse events such as bradycardia, headache, shivering, and postoperative nausea and vomiting (PONV) were reported, but no severe complications occurred.
3.5. Safety Profile
Across studies, local aminophylline instillation was consistently safe, with no systemic complications. Intravenous aminophylline occasionally caused mild cardiovascular changes (tachycardia, bradycardia) or minor postoperative symptoms, but no life-threatening adverse events.
3.6. Risk of Bias Assessment
The methodological quality of the included studies was assessed using the Cochrane Risk of Bias 2.0 (RoB 2) tool for randomized controlled trials and the ROBINS-I tool for the non-randomized studies.
Among the randomized controlled trials, most trials demonstrated a low to moderate risk of bias as illustrated in Fig. (2). Randomization procedures were generally described, although allocation concealment was sometimes unclear. For example, Barzegarnezhad et al. [18] and Khan et al. [21] had some concerns regarding randomization and blinding of participants. Barzegarnezhad et al. [19] and Rehab et al. [23] were judged to have a low overall risk of bias, as randomization, blinding, and outcome reporting were adequately addressed. In contrast, Saafan et al. [22] was considered at high risk of bias, particularly due to inadequate allocation concealment and selective outcome reporting. The more recent studies by Shabayek et al. [14] and Lubana et al. [13] were classified as having some concerns, primarily due to unclear blinding of outcome assessors and potential reporting bias, although their randomization process was appropriate.
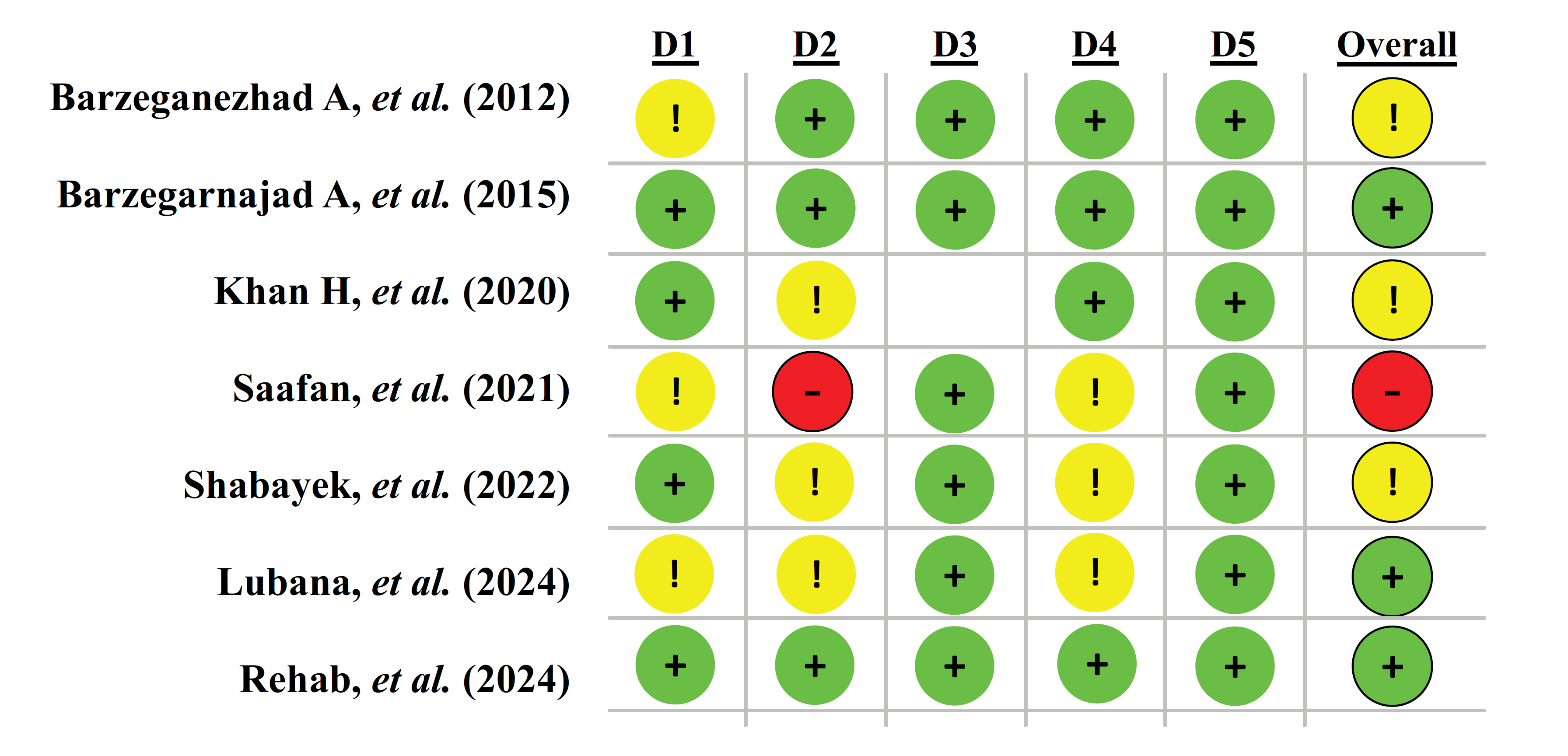
Cochrane Risk of Bias 2.0 (RoB 2).
| No. | Author (Year) / Refs. | Country | Design | N (I/C) | Intervention | Anesthesia Type | Duration | Age (mean ± SD) | Stone Size & Location | Lithotripsy Type | Definition of Success Rate | Main Findings (n, %, mean ± SD, p-value) | Complications/Adverse Events |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Barzegarnezhad A, et al. (2012) [18] | Iran | RCT | 120 (60/60) | Local aminophylline (250 mg aminophylline in 10 ml, mixed with 150 ml NS) vs NS | Not specified (likely spinal/GA) | 5 min | 34.8 ± 13.2 vs 35.4 ± 12.7 | <20 mm, distal ureter | Pneumatic Swiss lithoclast | Passage of the ureteroscope and complete lithotripsy with stone clearance | Success: 95% vs 71.6% (p=0.071); DJ stent: 13.3% vs 18.3% (p=0.071) | Not reported |
| 2 | Barzegarnezhad A, et al. (2015) [19] | Iran | RCT (double-blind) | 44 (21/22; 1→ dropout) | IV aminophylline (250 mg/100 ml NS, 1h preop) vs NS | Not specified | 30 min infusion | 32.9 ± 13.9 vs 32.2 ± 15.3 | Mean stone ~10 mm | Swiss pneumatic lithoclast | Stone-free rate (SFR) confirmed after URS | Success: 97.6% vs 84% (NS); surgery time shorter with aminophylline (p<0.05) | Not reported |
| 3 | Ghadyan, et al. (2017) [20] | Iran | Semi-experimental | 64 (32/32) | IV aminophylline (250 mg/250 ml NS) intraop | Spinal | Intraop | Mean 35.1 y (15–51) | Mostly distal ureter | URS spinal, Swiss pneumatic lithoclast | Procedural success (stone removal during URS without auxiliary SWL/stenting) | Success: 38% vs 59% failure; surgery time: 36.2 vs 18.4 (p<0.001); DJ stent: 36% vs 74% (p<0.001) | Mild hematuria; Tachycardia (n=8) |
| 4 | Khan, et al. (2020) [21] | Pakistan | RCT | 90 (45/45) | Local aminophylline (250 mg aminophylline in 10 ml, mixed with 150 ml NS) vs NS | Not specified | 5 min | 47.6 ± 18.1 | >4 mm, ureter | NA | Complete stone removal (stone-free at the end of URS) | Success: 88.9% vs 71.1% (p=0.03); surgery time: 39.9 ± 7.0 vs 48.7 ± 7.0 (p<0.001) | Not reported |
| 5 | Saafan, et al. (2021) [22] | Egypt | RCT | 50 (25/25) | Intravesical aminophylline (250 mg aminophylline in 10 ml, mixed with 150 ml NS) vs Balloon dilator | Spinal | 5 min | NA | Lower ureteral stones | Semi-rigid URS | Ureteral access after dilation; intraureteral pressure drop; SFR (residual fragments <4 mm) | No diff in SFR, op time, complications, stenting. Balloon group had ↑ pain & hematuria (p<0.05). Intraureteral pressure ↓ from 12.2 ± 1.85 → 7.8 ± 1.71 (p<0.001) | Balloon group: ↑ pain & hematuria; aminophylline safer |
| 6 | Shabayek, et al. (2022) [14] | Egypt | RCT | 83 (41/42) | Local aminophylline (250 mg aminophylline in 10 ml, mixed with 150 ml NS) vs balloon dilation | Spinal | 5 min | 37.8 ± 8.9 vs 38.4 ± 8.8 | <20 mm, juxtavesical/lower ureter | Pneumatic (lower), Holmium laser (upper) | Stone-free rate (SFR) = no residual stones <4 mm on postop NCCT | Success: 87.8% vs 88.1% (p=0.968); surgery time: 38.1 ± 7.0 vs 35.6 ± 7.3 (p=0.126) | Hematuria: 19.5% vs 42.9% (p=0.022); Pain: 9.8% vs 28.6% (p=0.030) |
| 7 | Lubana, et al. (2024) [13] | India | RCT | 100 (50/50 | Local aminophylline (250 mg aminophylline in 10 ml, mixed with 150 ml NS) vs NS | Spinal | 5 min | 38.5 ± 9.5 vs 40.5 ± 9.5 | Ureteric calculi <20 mm | Pneumatic ± laser | Ease of ureteral access + complete lithotripsy and extraction without auxiliary procedures | Success: 82% vs 44% (p=0.002); surgery time: 33.5 ± 4.3 vs 54.6 ± 15.9 (p<0.001); DJ stent: 38% vs 68% (p=0.011) | Not reported |
| 8 | Rehab, et al. (2024) [23] | Egypt | RCT (double-blind) | 45 (23/22) | IV aminophylline (4 mg/kg) vs NS | General Anesthesia | 20 min infusion intraop | 40.8 ± 6.3 vs 39.1 ± 6.8 | Ureteric stones (all stented) | URS under GA | Not defined as SFR; primary outcome was reduction in catheter-related bladder discomfort (CRBD) | CRBD: 26% vs 59% (p=0.025); VAS ↓ 0–6h (p<0.05); opioid & ketorolac ↓ (p<0.05) | PONV (3 vs 2, drug-related); Bradycardia (n=1, drug-related); Headache (n=1, drug-related); Shivering (n=1, drug-related) |
Abbreviations: I=Intervention; C=Control; CRBD = Catheter-Related Bladder Discomfort; DJ stent = Double J stent; IV = Intravenous; NS = Normal Saline; RCT = Randomized Controlled Trial; SFR = Stone-Free Rate; SWL = Shock Wave Lithotripsy; URS = Ureteroscopy; URSL = Ureteroscopic Lithotripsy; VAS = Visual Analog Scale.
| Study (Author, Year) / Refs. | Bias due to Confounding | Bias in the Selection of Participants | Bias in the Classification of Interventions | Bias due to Deviations from Intended Interventions | Bias due to Missing Data | Bias in the Measurement of Outcomes | Bias in the Selection of the Reported Result | Overall Risk of Bias |
|---|---|---|---|---|---|---|---|---|
| Ghadyan, et al. (2017, Iran) [20] | Serious (no randomization, potential baseline imbalance) | Moderate (patients selected from a single center, semi-experimental design) | Low (intervention clearly defined: IV aminophylline vs NS) | Moderate (no blinding, surgeon aware of group) | Low (complete follow-up reported) | Moderate (some outcomes subjective, e.g., hematuria) | Moderate (no protocol, selective reporting possible) | Serious |
The single non-randomized study by Ghadian et al. [20] was assessed using the ROBINS-I tool, and the results are presented in Table 2. This study was judged to have a serious overall risk of bias, mainly due to its semi-experimental design and absence of randomization, raising concerns about confounding and participant selection. Although the intervention was clearly defined (intravenous aminophylline vs. saline), the lack of blinding increased the risk of bias in deviations from intended interventions and outcome measurement. Missing data were minimal, but selective reporting could not be excluded.
In summary, the majority of the evidence comes from randomized controlled trials of moderate to good quality, while one non-randomized study carried a serious risk of bias. These methodological limitations should be considered when interpreting the overall findings.
3.7. Summary of Study Outcomes and Key Findings
Table 3 provides a comparative summary of the primary outcomes reported across the included studies, categorized by route of aminophylline administration. Most trials demonstrated improved ureteral access, reduced operative time, and lower rates of postoperative complications in the aminophylline groups compared to controls. Local or intravesical administration showed consistent benefits in reducing intraureteral pressure, hematuria, and pain scores, while intravenous use produced mixed effects on stone-free rates but was associated with reduced catheter-related bladder discomfort (CRBD) and analgesic requirements.
Overall, the data suggest that aminophylline, when used as an adjunct to ureteroscopic lithotripsy, enhances procedural efficiency and safety with minimal adverse events.
3.8. Summary of Evidence Certainty (GRADE Assessment)
The overall certainty of evidence was evaluated using the GRADE framework, considering study design, risk of bias, consistency, directness, precision, and potential publication bias. Table 4 presents the summarized assessment of key outcomes, including operative time, stone-free rate, need for ureteral stenting, postoperative pain and hematuria, and safety.
| Route of Administration | Author (Year) / Refs. | Main Outcome(s) | Effect Size / Result Summary | p-value (as reported) |
|---|---|---|---|---|
| Local / Intravesical | Barzegarnajad et al., 2012 [18] | Stone-free rate (SFR); DJ stent use | ↑ Success (95% vs 71.6%); ↓ DJ stent use (13.3% vs 18.3%) | 0.071 (SFR); 0.071 (stent) |
| Khan et al., 2020 [21] | SFR; operative time | ↑ Success (88.9% vs 71.1%); ↓ operative time (39.9 ± 7.0 vs 48.7 ± 7.0 min) | 0.03 (SFR); < 0.001 (time) | |
| Saafan et al., 2021 [22] | Intraureteral pressure; pain & hematuria | ↓ Pressure (12.2 → 7.8 mmHg); ↓ pain & hematuria vs balloon dilator | < 0.001 (pressure); < 0.05 (symptoms) | |
| Shabayek et al., 2022 [14] | SFR; pain; hematuria | No SFR diff (87.8% vs 88.1%); ↓ hematuria (19.5% vs 42.9%); ↓ pain (9.8% vs 28.6%) | 0.968 (SFR); 0.022 (H); 0.030 (P) | |
| Lubana et al., 2024 [13] | SFR; operative time; stent requirement | ↑ Success (82% vs 44%); ↓ operative time (33.5 vs 54.6 min); ↓ DJ stent (38% vs 68%) | 0.002; < 0.001; 0.011 | |
| Intravenous (IV) | Barzegarnajad et al., 2015 | SFR; operative time | ↑ SFR (97.6% vs 84%); ↓ operative time with aminophylline | < 0.05 |
| Ghadyan et al., 2017 [20] | Procedural success; operative time; stent use | ↓ Success (38%); ↑ operative time (36.2 vs 18.4 min); ↓ stent (36% vs 74%) | < 0.001 (time & stent) | |
| Rehab et al., 2024 [23] | CRBD; pain; analgesic need | ↓ CRBD (26% vs 59%); ↓ VAS pain (0–6 h); ↓ opioid/ketorolac use | 0.025 (CRBD); < 0.05 (others) |
| Outcome | No. of Studies (Design) | Consistency of Results | Risk of Bias | Imprecision | Indirectness | Publication Bias | Certainty of Evidence |
|---|---|---|---|---|---|---|---|
| Operative time reduction | 4 RCTs (Khan 2020, Saafan 2021, Shabayek 2022, Lubana 2024) | Consistent reduction in operative time with aminophylline across studies | Some concerns (small sample size, unclear allocation) | Moderate (n = 45–100 per study) | Direct evidence | Unlikely | Moderate |
| Stone-free rate (SFR) / procedural success | 6 studies (5 RCTs + 1 semi-experimental) | Variable: some trials showed higher SFR, others showed non-significant | Some risk (heterogeneous outcome definition, unclear blinding) | Serious (small trials, wide variance) | Direct | Possible (favoring positive findings) | Low |
| Need for a DJ stent / ureteral access | 4 RCTs | Consistent reduction in stent requirement and improved ureteral access | Low to moderate | Moderate | Direct | Unlikely | Moderate |
| Pain and hematuria | 3 RCTs | Consistent reduction in postoperative pain and hematuria vs control | Low to moderate | Moderate | Direct | Unlikely | Moderate |
| Adverse events | 3 RCTs + 1 semi-experimental | Mild and transient only (tachycardia, headache, PONV) | Low | Moderate | Direct | Unlikely | Moderate–High |
| Catheter-related bladder discomfort (CRBD) | 1 RCT (IV route) | Single study, consistent internal results | Low | Serious (single small trial) | Direct | Unclear | Low |
Across the included studies, the certainty of evidence ranged from low to moderate. Evidence supporting reduced operative time, improved ureteral access, and decreased stent requirement was judged as moderate certainty, supported by consistent findings across multiple randomized trials. In contrast, outcomes such as stone-free rate and catheter-related bladder discomfort were graded as low certainty due to variability in study design, outcome definitions, and limited sample sizes. Adverse events were mild and transient across all studies, supporting a moderate-to-high level of confidence regarding the safety of aminophylline as an adjunct during ureteroscopic lithotripsy.
4. DISCUSSION
Urolithiasis is a common condition worldwide, with a steadily rising prevalence in recent decades [1]. Treatment for urinary stones should be administered promptly, guided by stone size, shape, location, and ureteral anatomy [24]. Stones < 3 Mm usually pass spontaneously and can be managed conservatively, whereas stones 7–10 mm rarely pass without intervention. In such cases, medical expulsive therapy (MET) with alpha-blockers, calcium channel blockers, PDE5 inhibitors, or corticosteroids can facilitate passage and reduce colic attacks [8, 9].
Ureteroscopy (URS) is now widely recognized as an effective and safe treatment for ureteral and renal stones, with procedure rates increasing by more than 251.8% between 1996 and 2016 [25]. URS is considered the standard treatment for stones >10 mm, regardless of location within the ureter [26-28]. However, complications such as ureteral avulsion, mucosal injury, bleeding, and urinary tract infections remain potential concerns [8]. In 1–37% of unstented patients, primary access failure occurs due to a narrow ureteral orifice or intramural segment [16]. The common solution is pre-stenting, which passively dilates the ureter but carries drawbacks, including the need for a second procedure and the potential for stent-related symptoms, reported in up to 80% of cases [8, 9].
To improve URS outcomes, pharmacologic adjuvants have been evaluated. Alpha-blockers are frequently prescribed, but their therapeutic effect requires several days [7, 22]. In contrast, aminophylline is inexpensive, safe, and has a rapid onset of smooth muscle relaxation within 5 minutes of instillation [8-10, 19]. Local aminophylline reduced the ureteropelvic junction and infundibular spasm, facilitating access to staghorn stones [28]. Significant decrease in intraureteral pressure from 12.3 ± 1.9 mmHg to 8.5 ± 1.9 mmHg after administration [14].
Evidence from five randomized controlled trials (RCTs) supports the benefit of aminophylline (local or intravesical) (Table 1). Barzegarnezhad et al. (2012) [18] observed a higher stone-free rate (95% vs 71.6%), though the difference was not statistically significant. Khan et al. [21] reported higher success (88.9% vs 71.1%, p=0.03) and shorter operative time (39.9 vs 48.7 minutes, p<0.001). Saafan et al. [22] compared intravesical aminophylline with balloon dilatation, finding no significant difference in SFR, operative time, or stenting, but balloon dilatation was associated with significantly higher hematuria and postoperative pain, while aminophylline markedly reduced intraureteral pressure (from 12.2 ± 1.85 to 7.8 ± 1.71 mmHg, p<0.001). Shabayek et al. [14] found similar stone-free rates but significantly lower hematuria (19.5% vs 42.9%, p=0.022) and pain scores (9.8% vs 28.6%, p=0.030). Lubana et al. [13] confirmed higher success (82% vs 44%, p=0.002), shorter operative time (33.5 vs 54.6 minutes, p<0.001), and reduced stent requirement (38% vs 68%, p=0.011). Importantly, none of these studies reported systemic toxicity from local aminophylline.
Intravenous aminophylline has also been investigated. Rehab et al. [23] showed that IV aminophylline significantly reduced catheter-related bladder discomfort (26% vs 59%, p=0.025), lowered VAS pain scores in the first 6 hours, and reduced opioid/ketorolac requirements. Barzegarnezhad et al. [19] found a higher stone-free rate (97.6% vs 84%) and significantly shorter operative time with IV aminophylline. Ghadian et al. [20] reported lower procedural success (38%) and higher need for stenting/auxiliary procedures, although intraoperative ureteral spasm relief was observed; mild tachycardia occurred in 8 patients.
The results presented in Table 1 reveal notable variability among the included studies. This variability encompasses several important factors, such as the average age of the participants, the size of the stones, the location of the stones, and the specific types of ureteroscopy lithotripsy employed. These differences can significantly influence the outcomes and success rates of each study regarding the administration of aminophylline instillation. Another important consideration is that the definition of “success” was not uniform across trials. Some studies defined success as the stone-free rate (SFR), others as procedural success or ease of ureteral access [18, 20-22]. This heterogeneity limits comparability and may contribute to the observed differences in outcomes. The average age of participants may affect treatment response, although results were not statistically significant in the study by Morgan et al [29]. The location of stones in the proximal ureter can reduce the success rate of ureteroscopy [30]. Stone size also affects the success rate of ureteroscopy, with larger stones requiring longer operative times [31]. Furthermore, the type of lithotripsy employed may vary in efficacy, thereby affecting overall outcomes [31]. Effect sizes likely vary according to stone size, location, and ureteroscopic modality. Future trials should stratify and adjust for these variables.
As highlighted by our risk-of-bias assessments (Fig. 2 for RoB 2 and Table 2 for ROBINS-I), several trials had methodological limitations, including unclear blinding, potential baseline imbalances, and selective reporting, which further temper the certainty of evidence.
Currently, no studies directly compare aminophylline with alpha-blockers; therefore, conclusions about relative efficacy cannot be drawn. However, based on the onset of action, aminophylline provides smooth muscle relaxation within 5 minutes, compared to several days with alpha-blockers [7, 8].
Beyond its use in urolithiasis, aminophylline is also known for its diuretic and reno-protective effects, mediated by adenosine receptor antagonism and improved urine output in acute kidney injury [10, 30]. Mechanistically, aminophylline increases intracellular cAMP/cGMP, reduces calcium influx, and promotes smooth muscle relaxation; theophylline also inhibits adenosine-induced mediator release [10, 12, 32].
In the context of ureteroscopy, smooth muscle relaxation is crucial for facilitating easier instrument insertion and reducing intraoperative resistance, thereby improving both procedural success and patient comfort [18]. The ability of aminophylline to shorten operative time was highlighted by Khan et al. [21], who observed a 9-minute reduction compared with controls. Lubana et al. [13] similarly reported a significantly shorter operative time in the aminophylline group.
5. STUDY LIMITATIONS
This review has limitations. The relatively small sample sizes of the included trials limit generalizability. In addition, there was marked heterogeneity in the definition of “success,” with some studies using stone-free rate (SFR), others procedural success, or ease of access, reducing comparability across trials. Study populations were heterogeneous, and outcome measures were variably reported. Finally, no quantitative meta-analysis was feasible due to differences in study design, interventions, and outcomes. Sensitivity analysis was not feasible given the small number of studies; therefore, the findings should be interpreted cautiously.
In clinical practice, aminophylline may be considered as a low-cost adjunct in centers where alpha-blockers or advanced dilatation devices are not readily available, particularly in Southeast Asia. In resource-limited centers, particularly across Asia, aminophylline may provide a pragmatic alternative to alpha-blockers or balloon dilators. Larger multicenter RCTs with standardized definitions of outcomes are still needed before aminophylline can be routinely recommended in clinical guidelines.
CONCLUSION
Aminophylline, whether given locally or intravenously, appears to facilitate ureteroscopic lithotripsy by improving access, increasing success rates, reducing operative time, and lowering morbidity, with no major adverse effects reported. Its rapid action and safety profile make it a promising adjunct in ureteroscopy; However, because of small sample sizes, heterogeneous study designs, and low overall certainty of evidence, its use should be considered investigational until larger, high-quality multicenter trials are conducted.
AUTHORS’ CONTRIBUTIONS
The contributions of the authors are as follows: N.A.S.: Responsible for conceptualization, literature search, data extraction, formal analysis, writing the original draft, and visualization; F.S.: Provided supervision, contributed to data extraction, conducted the risk of bias assessment, validation, writing of the review, editing, critical revision, and project administration. Both authors read and approved the final manuscript and agree to be accountable for all aspects of the work.
LIST OF ABBREVIATIONS
| CRBD | = Catheter-related Bladder Discomfort |
| SFR | = Stone-free Rate |
| PONV | = Postoperative Nausea and Vomiting |
| URS | = Ureteroscopy |
| SWL | = Shock Wave Lithotripsy |
AVAILABILITY OF DATA AND MATERIALS
All the data and supporting material are available within the article.
ACNOWLEDGEMENTS
Declared none.
AI DISCLOSURE STATEMENT
No AI-assisted tools were used for data analysis, image generation, or manuscript writing. The Rayyan web-based platform was used only to support blinded screening and organization of titles and abstracts during the study selection process. All screening decisions, data extraction, and interpretation were conducted manually and independently by the authors.


