All published articles of this journal are available on ScienceDirect.
The Diagnosis and Treatment of Vesicoureteral Reflux: An Update
Abstract
Vesicoureteral reflux [VUR] remains a common problem seen by pediatric providers. Despite a great deal of research, the debate regarding how to screen and treat patients reremains tense and controversial. This review seeks to summarize the management of VUR with emphasis on recent published findings in the literature and how they contribute to this debate. The goals of managing VUR include preventing future febrile urinary tract infections [FUTI], renal scarring, reflux nephropathy and hypertension. The topdown approach with upper tract imaging and selective vesicocystourethrogram [VCUG] is an emerging alternative approach in the evaluation of children after their first FUTI. The elimination of bladder and bowel dysfunction [BBD] is an important management strategy to prevent further FUTIs, regardless of treatment choice. Antibiotic prophylaxis is a safe and effective modality to sterilize the urinary tract. Endoscopic treatment of VUR is an attractive modality in select patients, although some concerns remain regarding its effectiveness and durability as compared to to the gold standard of open or laparoscopic ureteroneocystostomy. Lastly, further research is required to determine the most effective algorithm to evaluate the pediatric patient after the first febrile UTI.
1. INTRODUCTION
Galen, the ancient anatomist first described the role of the vescioureteric valve in the one directional flow of urine in the first century AD [1] (Fig. 1). However, it was not until Dr. Hutch described a causal relationship between vesicoureteral reflux [VUR] and pyelonephritis in the paraplegic population that the medical community [urologists, in particular] began to devote more attention towards the relevance of VUR [2]. Another landmark reported by Ransley and Risdon strengthened the significance of VUR by finding renal scarring in a porcine model with the combination of VUR, elevated intravesical pressures and infected urine in the bladder [3]. In 1973, Dr. Bailey described “reflux nephropathy” as “the coarse renal scarring that results in form of urinary tract infection [UTI] in the setting of VUR” [4]. After these seminal reports on VUR, VUR has become one of the most studied topics in all of pediatric urology. Our current understanding of the interplay between UTI, VUR, and renal scarring was heavily influenced by the clinical research of Smellie in 1991 that pointed out the importance and role of antibiotic prophylaxis in promoting kidney health [5]. Unfortunately, despite the frequency of VUR within the population and the extensive history of study, the proper evaluation and treatment of the patient with VUR remains a contentious issue.

Caption: Normal ureterovesical junction and trigone. A. Cross-section of the ureterovesical junction. B. Coronal section of the ureterovesical junction. From Tanagho EA, Pugh RCB. The anatomy and function of the ureterovesical junction. Br J Urol. 1963; 35:151-65, with permission.
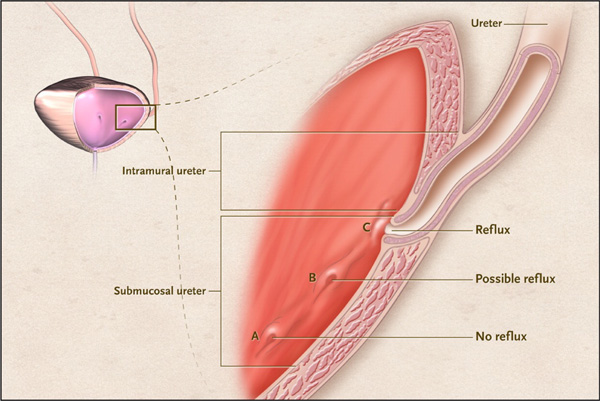
Anatomy of the Ureterovesical Junction. From: Diamond DA, Mattoo TK. Endoscopic treatment of primary vesicoureteral reflux. N Engl J Med 2012; 366:1218-26, with permission.

Forest plots depicting the negative influence of BBD on the resolution rate of VUR. Patients with BBD have a lower resolution rate (a) compared to patients without BBD (b). Elder JS, Diaz M. Vesicoureteral reflux—the role of bladder and bowel dysfunction. Nat Rev Urol. doi:10.1038/nrurol.2013.221, with permission.
In the United States, both the American Urological Association [AUA] and the American Academy of Pediatrics [AAP] have provided guidelines in the hope of helping pediatricians and pediatric urologists provide appropriate screening and treatment for patients with both UTIs and VUR. In Europe, the European Association of Urology [EAU] and the National Institute of Health and Clinical Excellence [NICE] have also attempted to lay out guidelines on the management of VUR in the pediatric population. In the guidelines, bladder and bowel dysfunction [BBD] has become more recognized as a major determinant in the potential of future urinary tract infections and resolution of VUR [with or without treatment]. This article not only recounts current research and guidelines, but also discusses the increasing role of BBD and addresses recent landmark studies in the screening and management of VUR.
2. ETIOLOGY
The flow of urine from the ureter to the bladder is normally one directional across the vesicoureteral junction. Normally, VUR does not occur because there is compression of the tunneled, submucosal ureter as the bladder fills with urine. VUR is a developmental anomaly created by an inadequate length of the intravesical submucosal tunnel of the ureter in relation to the pressures experienced by the patient’s bladder (Fig. 2). Today VUR is classified as either primary or secondary VUR. VUR is primary if it is thought to be an isolated, fundamental deficiency in the above-described anatomic ureteral tunnel. Paquin was the first to describe the normal anatomical and physiological tunnel’s dimensions as 5 [length] to 1 [diameter]. Secondary VUR refers to VUR that is due to abnormal bladder dynamics and/or elevated voiding pressures. These abnormalities are seen with lower urinary tract dysfunction stemming from bladder outlet obstruction [posterior urethral valves or ureterocele], neurogenic bladder, or dysfunctional voiding. In secondary VUR, the vesicoureteral tunnel may or may not be of normal length, however, due to elevated intravesical voiding pressures, the vesicoureteral tunnel is “insufficient” to prevent VUR. Older literature did not appreciate this distinction of VUR and it was common to see reports of patients with ‘primary reflux’ with a predominance of bladder dysfunction. These same reports would describe spontaneous resolution of VUR following anticholinergic therapy to address the bladder dysfunction [6].
3. EPIDEMIOLOGY
In 1966, Baker et al. stated that VUR may be present in up to 70% of infants who present with a UTI [7]. Within the general population, VUR is estimated to be present in 1-2% of all children [8]. Interestingly, there are racial and gender differences. The incidence of VUR in black children is one third to white children [9]. Females are twice as likely as males to have VUR in the general population. However, in the infant population, VUR is more prevalent in boys. Unfortunately, most of our knowledge regarding the diagnosis of VUR is confounded by selection bias with the population that is screened; those with a previous UTI or hydronephrosis [HN] are more likely to be evaluated. Gender predispositions of VUR may be related to the prevalence of UTI and HN within the general population; hence, a significant proportion of VUR may remain undiagnosed.
There is a familial relationship of VUR and approximately 30% of siblings of index patients will have VUR from screening studies. Primary VUR is found with a significantly higher prevalence in close relatives of those with reflux. Kaefer et al. found that the prevalence of VUR was 80% between identical twins and that the prevalence was 35% in nonidentical or fraternal twins [10]. Scott et al. over a 3-year period studied familial VUR in patients that were seen in antenatal clinics [11]. Newborns with parents or other family members with definite [cystography] or probable VUR were enrolled in the study. Of 211 with confirmed family history, there was a prevalence of VUR in 31% of patients.
3.1. Sibling Screening
Today, routine sibling screening for VUR is no longer practiced. For those with a brother/sister with VUR, the AUA Guidelines would suggest consideration for a voiding cystourethrogram [VCUG] only if there is evidence of renal scarring on imaging or a history of a significant UTI [12]. Similarly, the EAU Guidelines suggest a VCUG if there is evidence of renal scarring on ultrasonography or a history of urinary tract infection. In older, asymptomatic children, screening for VUR is not recommended [13]. In general, sibling screening has diminished in practice as we selectively evaluate children based on a risk stratification profile rather than uniformly screen all siblings.
4. VUR DIAGNOSIS
Traditionally, an acute episode of pyelonephritis prompted a diagnostic work-up that included a VCUG, and this remains the gold standard imaging test for the diagnosis of VUR. The rationale for VCUG testing in the setting of pyelonephritis is based on the higher prevalence of VUR diagnosed after a significant UTI [often febrile]. A meta-analysis by Faust et al. determined VUR to be the primary risk factor predisposing children to acquired renal cortical scarring [approximately 3 fold], possibly by increasing the risk of recurrent UTIs [14].
With the widespread adoption of prenatal ultrasound for maternal-fetal care, another population now targeted for diagnostic evaluation includes those infants with persistent abnormalities on a postnatal ultrasound. In these patients, the AUA recommends VCUG for those with HN [SFU grade 3 or 4], hydroureter or an abnormal bladder ultrasound on either late prenatal or postnatal ultrasound. Patients with lower grade HN are given the options of VCUG or observation [12]. The EAU acknowledges that the degree of HN is not a reliable indicator of VUR. However, VCUG is recommended for those with bilateral high-grade HN, duplex kidneys with HN, ureterocele, ureteral dilation or abnormal appearing bladders [13]. The 2011 revised AAP Guidelines on the management of UTI no longer recommend automatic VCUG testing following a UTI. Instead, the AAP recommends a VCUG following recurrent febrile UTIs or a renal/bladder abnormality on ultrasound showing hydronephrosis, hydroureter or evidence of renal scarring [14,15].
Once the diagnosis of VUR is established on VCUG, VUR is categorized or graded in severity by the degree of ureteral dilation on VCUG. The grading system for VUR that is globally accepted is based upon the radiologic grading system established by the International Reflux Study Committee in 1981 [16]. Grade I shows reflux into a non-dilated ureter; Grade II, into pelvis and calyces without dilatation; Grade III, mild to moderate dilatation of ureter, renal pelvis and calyces with minimal blunting of fornices; Grade IV, moderate ureteral tortuosity and dilatation of pelvis and calyces; and Grade V, gross dilatation of ureter, pelvis and calyces, loss of papillary impressions and ureteral tortuosity.
5. DIAGNOSTIC AND SCREENING PROTOCOLS
The challenge in the evaluation and management of VUR is primarily due to the lack of consensus on which patients to screen and which patients require treatment. As previously described, most clinicians use renal and bladder ultrasound [RBUS] as a barometer in deciding who should have a VCUG. RBUS is an ideal screening tool as it is inexpensive, non-invasive and uses no radiation. Unfortunately, some studies have found RBUS findings to be hampered by a poor sensitivity and negative predictive value for determining VUR [17]. In a study comparing ultrasound and VCUG imaging within a population of newborns with a family history of VUR, there was low sensitivity and a poor predictive value for detecting VUR when evaluating prenatal and postnatal renal pelvic diameters [18]. The limitations of RBUS are highlighted by reports of normal RBUS findings in up to 60% of cases of VUR despite missing 50% of renal abnormalities noted on dimercaptosuccinic acid renal scan [DMSA] [19-21]. Drawbacks to VCUG testing are the invasive nature, potential radiation exposure and the risk of potential iatrogenic UTI with urethral and bladder catheterization. The radiation concern has been lessened with limited dosimetry and spot films and the invasive nature has been tempered with sedation. Nevertheless, VCUG compared to RBUS is clearly provokes more anxiety for patients, and families.
5.1. Top-Down Approach
As an alternative to the traditional approach of early VCUG testing in the diagnostic evaluation, some clinicians advocate a “top-down” approach. The rationale for the top-down approach is that only VUR that results in pyelonephritis is clinically relevant. With the knowledge that only pyelonephritis leads to renal scarring, this approach employs a DMSA soon following a confirmed, febrile UTI. Those with a negative exam would require no further evaluation unless recurrent UTI was noted. However, the VCUG would be performed upon those with confirmed, focal photopenia on DMSA. In a retrospective evaluation of the top-down approach, 303 children under 2 years old had a DSMA and VCUG performed following their first UTI [22]. Despite an 82% rate of FUTIs, VUR was found in only 26%. Of those with FUTI and VUR on VCUG, 66% had an abnormal DMSA. In this cohort of patients with abnormal DMSA scans, the top down approach would have only identified 66% of those with VUR and risk of future scarring and excluded 40% of the cohort without VUR from a VCUG. Of seven patients with dilating, grade III VUR and a normal DMSA, all except one had an initial FUTI. Furthermore, these same 7 patients with normal DMSA scans were found on follow up to have complete resolution of VUR [5 patients] or downgraded to grade I VUR [2 patients].
Tseng et al. conducted a 10-year retrospective review of 142 children seen at their tertiary care hospital following their first UTI [23]. All had DMSA renal scans and VCUG performed. Seventy percent of the patients had evidence on
DMSA of pyelonephritis, and 1.4% had evidence of scarring on DMSA. VCUG found 30% of the patients had VUR. The PPV and NPV of abnormalities on DMSA for predicting the presence of VUR on subsequent VCUG were 37 and 88%, respectively. Five patients with a normal DMSA were noted to have VUR on VCUG and they were of low grade.
The advantage of the top-down approach is that the patients with abnormal DMSA findings would be selectively evaluated, as the majority of those with VUR and a febrile UTI do not develop renal scarring. By targeting an at risk cohort for a VCUG, a segment of children with FUTI would be shielded from the morbidity of further screening and potential treatment. Unfortunately, increasing use of the DMSA scan has limitations. These limitations include limited availability of the DMSA isotope, requirement of an intravenous line, frequent sedation requirements, radiation exposure [albeit small], and higher financial costs. Lastly, most patients found to have renal scarring with abnormal DMSA scans have a negative VCUG and the persistence of renal parenchymal lesions is independent of VUR [24]. Whether these lesions appeared prior to the presenting UTI and whether they necessitate antibiotic prophylaxis remains uncertain.
Interestingly, DMSA scan abnormalities in conjunction with high grade VUR [e.g. grades 3-5] are associated with a greater chance for breakthrough UTI [60%] despite antibiotic prophylaxis when compared to those with a normal DMSA and a similar VUR grade [8%] [25]. Furthermore, only 5% of those with an abnormal DMSA showed VUR improvement compared to 46% that resolved in the normal DMSA group. When referencing the AUA guidelines, DMSA imaging is recommended in the setting of an abnormal renal ultrasound that suggests scarring or an elevated serum keratinize [12]. The EAU Guidelines mention the alternative top-down approach and describe the potential to miss 5-27% cases of VUR, with questionable significance [13].
6. BLADDER AND BOWEL DYSFUNCTION
6.1. VUR, UTIs, and BBD
There is a known relationship and co-morbidity between VUR and bladder and bowel dysfunction [BBD]. In 1979, Koff et al. studied 53 neurologically intact children with UTI and BBD and found that 50% had VUR [26]. Recently, this relationship has been further elucidated by the Swedish reflux trial in children where they found a total of 20% of patients enrolled with high grade reflux had bladder dysfunction characterized by a high bladder capacity and incomplete bladder emptying associated with an increased post-void residual [27]. Dilating reflux directly correlated with bladder dysfunction and successful treatment of bladder dysfunction was associated with improvement of reflux grade. Furthermore, renal damage at study entry and follow-up was significantly associated with bladder dysfunction at 2 years.
The latest AUA VUR guidelines recognize this concomitant relationship between VUR, UTIs, and BBD [12]. BBD has a negative correlation with the spontaneous VUR resolution rate and the success rate following endoscopic surgery (Fig. 3). Additionally, there is a higher incidence of postoperative UTI in those children with both VUR and BBD [12]. Due to this comorbidity, the AUA recommends screening for symptoms indicative of BBD upon initial evaluation. These symptoms include urinary frequency, urgency, prolonged voiding intervals, daytime wetting, perineal/penile pain, holding maneuvers and constipation/encopresis. The guidelines recommend treatment of BBD prior to any surgical treatment of VUR. Lastly, continuous antibiotic prophylaxis [CAP] is recommended for those with concomitant BBD and VUR due to an increased risk of UTI. The EAU guidelines also acknowledge the association between lower urinary tract dysfunction [LUTD] and VUR [13]. They also state a video-urodynamic study may be warranted if a child with LUTD and a history of FUTIs is refractory to standard bladder urotherapy [13].
In contrast to endoscopic correction, open ureteral reimplantation can be quite successful in the treatment of VUR in children, regardless of the presence of BBD [28]. This is attributed to the more durable continence mechanism created by ureteral reimplantation. The continence mechanism for endoscopic ureteral injection relies on principles of Poiseuille’s Law by narrowing the radius of the ureteral lumen with a bulking agent and thus increases resistance of retrograde urine flow. This increased resistance, though, is more susceptible to persistence of VUR in the setting high pressure voiding with BBD. In contrast, the continence mechanism established by ureteral reimplantation is based on a flap-valve mechanism which allows coaptation of the tunneled ureteral segment during bladder filling and is significantly more reliable in treatment of VUR for those with BBD.
A critical driving force in the management of VUR is the presence of UTI and in particular FUTI. With BBD, there is a significant elevated risk and incidence of UTIs. The association of UTIs with BBD is due to several characteristics of the lower urinary tract with BBD that provides an ideal environment for the propagation of uropathic bacteria. Children with BBD often void infrequently or have incomplete emptying which results in a prolonged dwelling time of urine and incubation time for bacterial growth.
6.2. BBD Assessment and Management
After identifying LUTD, it is recommended to screen for the presence of concomitant bowel dysfunction given their frequent interdependence. The coexistence of bladder and bowel dysfunction is termed BBD [29] and the importance of BBD is that the physiologic regulation of the bladder and bowel are shared and thus dysfunction in one organ system is seen in the other organ. Multiple studies have demonstrated that treatment of bowel dysfunction first can lead to resolution of bladder dysfunction [and a significantly lower rate of UTI], and vice versa [30-32]. The assessment of BBD should consist of a thorough history, physical exam, and several noninvasive studies. Recommended testing for BBD includes urinalysis, +/- urine culture, elimination diaries, symptom questionnaires, uroflowmetry with or without EMG and pelvic ultrasound to evaluate the functional and anatomical characteristics of the bladder and bowel [30, 34].
Management of BBD initially consists of behavioral modification [termed standard urotherapy] that entails education regarding the anatomy and function of the bladder, bowel and pelvic floor musculature. Details regarding timed voiding, proper voiding posture, double voiding, and pelvic floor relaxation exercises are all mentioned in the literature as valid treatments of LUTD [33]. More innovative and interactive treatments are reserved for children that do not respond to standard urotherapy [34]. A recent study by Ladi-Seyedian et al. assessed the efficacy of animated biofeedback and pelvic floor muscle exercises in children with non-neuropathic underactive bladder [35]. In terms of UTI relapse rates, postvoid residual volume, voiding time, and maximum urine flowrate, patients receiving biofeedback therapy fared significantly better than the controls after six months of treatment. Pharmacologic therapy [e.g. alpha blockers and Botulinum-A toxin] is also selectively used to improve bladder emptying despite their off-label and/or investigational nature [36-38]. Finally, intermittent urethral catheterization of the bladder is a rare requirement to empty the bladder in patients with refractory BBD or LUTD and recurrent UTIs.
7. TREATMENT OPTIONS
7.1. Continuous Antibiotic Prophylaxis [CAP]
For years, CAP’s efficacy has been questioned because of its potential risks [selecting resistant organisms, Stevens-Johnson syndrome] and uncertain benefit [39-41]. Later studies provided clear evidence of the efficacy of CAP as compared to observation. The most notable examples included Craig et al. with the PRIVENT study and the Swedish reflux trial in children [42,43]. These studies, however, demonstrated a small benefit from CAP. Some argued that the results were statistically, but not clinically significant. Others argued that these studies only included a subset of patients with high grade VUR, and were therefore not generalizable to whole VUR population. The recently published RIVUR [Randomized Intervention for Children with Vesico-Ureteral Reflux] trial is now the largest, randomized, placebo-controlled, double-blinded, multicenter study comparing CAP [trimethoprim/sulfamethoxazole] to placebo [44]. The RIVUR trial addressed limitations in the PRIVENT and Swedish reflux trial studies by using a placebo control, examining a large sample size of children with VUR and employing strict UTI criteria [catheterized specimens for all non-toilet-trained patients enrolled]. The RIVUR study included 607 patients aged 2-72 months with grade I-IV VUR and a first or second symptomatic UTI. The majority of patients were female [92%] and 86% of the patients had an index FUTI including 56% of the 126 toilet-trained children with BBD. Although no significant difference was seen in the incidence of renal scarring, the RIVUR study significantly found a 50% relative decrease in the incidence of UTIs in those treated with CAP. There was no significant difference seen in the study cohorts’ resistance patterns of fecal Escherichia coli. However, the first symptomatic UTI with resistant E. coli was statistically more likely to occur among those on prophylaxis [63% vs 19% of placebo]. Interestingly, the benefit of CAP was most pronounced in those with bladder dysfunction as well as those who initially presented with a febrile UTI.
In the AUA VUR Clinical Guidelines, the benefit of CAP was based on the recommended use of screening criteria to determine a risk profile of future UTIs [12]. High-risk groups who would benefit from CAP included those <1years of age, dilating VUR [grades 3-5] and/or a history of febrile UTI. For those > 1year of age, CAP is recommended in those with BBD. Otherwise, CAP is considered optional for those >1 year of age. The EAU VUR guidelines suggest that regardless of the grade of reflux or presence of renal scars, all patients diagnosed at age < 1 year can be treated initially with CAP [13]. Definitive surgical or endoscopic correction is preferred for those with frequent breakthrough infections. In all children presenting at age 1-5 years, CAP is the preferred, initial option. However, those with lower grades of reflux and no symptoms are considered potential candidates for surveillance without CAP.
7.2. Surgery
The recommendation for surgical treatment is a shared-decision-making process involving the parents/guardians and physician. The decision is influenced by multiple factors including the risk-benefit of intervention, presence of breakthrough UTIs and also the anxiety from VCUG testing with persistent VUR.
Endoscopic therapy is an attractive, minimally invasive, outpatient, surgical option in the treatment of VUR. The injection of polytetrafluoroetheylene [Teflon] to treat VUR was used in the 1980’s and 1990’s until reports of safety concerns and adverse reactions associated with Teflon migration into other organs negated its usage. Endoscopic therapy has re-emerged in the surgical treatment for VUR since the Federal Drug Administration approved dextranomer hyaluronic acid copolymer [Deflux®] in 2001 for endoscopic correction of grades II-IV VUR [45, 46]. The success rate of endoscopic treatment is approximately 60-90% and is more dependent and influenced by the grade of VUR and the absence of BBD. Modification of endoscopic techniques by using multiple submucosal injection sites at the ureteral orifice has been reported to improve success rates [47, 48]. A meta-analysis by Elder et al., however, suggests the most significant prognostic factor in the success of endoscopic treatment is the pre-operative VUR grade, not the particular technique [49].
Open neoureterocystotomy or ureteral reimplantation is the “gold standard” for surgical correction of VUR. The success rate of open ureteral reimplantation is approximately 95% regardless of VUR grade or presence of BBD. Ureteral reimplantation corrects an inadequate ureteral tunnel length by establishing a flap-valve mechanism that allows passive compression of the submucosal ureter during bladder filling. The key of surgical characteristic in the construction of the reimplanted ureter is establishing a 5:1 ratio in the tunneled length-to-ureteral diameter [50]. There are multiple surgical variations of open ureteral reimplantation that involve intravesical [IV], extravesical [EV] or combined intra- and extravesical dissections [51-56].
The first of the IV approaches is the Politano-Leadbetter reimplant. Refluxing ureters are commonly located too lateral within the bladder and this approach involves the creation of a neo-hiatus insertion site into the posterior bladder wall superior and medial to the original hiatus [51]. The ureter is passed from the new hiatus through a newly created, submucosal tunnel toward the bladder neck. The Glenn-Anderson technique involves the same IV mobilization and ureteral tunneling found in the Politano-Leadbetter technique, however, there is no translocation to a new hiatus and the original hiatus is maintained [52]. The decision-making for a Glenn-Anderson technique is dependent upon the anatomy of the patient’s trigonal and bladder anatomy and whether the ureter is located too inferiorly preventing a 5:1 length:diameter ratio for the tunneled advancement.
The cross-trigonal, Cohen ureteral reimplant is an IV mobilization of the ureter that maintains the original insertion hiatus into the bladder wall [53]. Unlike the Glenn-Anderson technique, the ureter is reimplanted across the trigone toward the contralateral ureter. This surgical technique is particularly advantageous in the creation of a long tunnel for reimplanted dilated ureters. Some argue, however, that this technique affects the anatomy by making endoscopic access to the ureters more difficult if needed in the future [e.g. for urolithiasis], but this does not seem to be an issue longitudinally.
Both Lich and Gregoir simultaneously developed the EV technique of ureteral reimplantation [54]. The technique involves EV dissection of the ureter toward the ureteral orifice, followed by an incision through the seromuscular layer but not through the mucosal layer to create a bed for the EV ureter, followed by a closure of the detrusor muscle defect upon the ureter. This technique was originally rejected in the United States amid fears of poor results, but this changed over the last few decades and the longitudinal results are equivocal to IV techniques [55]. A notable morbidity resulting from EV ureteral reimplantation is the development of transient urinary retention in approximately 16% of patients. This is attributed to a transient neuropraxia [56,57]. Fortunately, the incidence of urinary retention is significantly reduced in patients undergoing unilateral EV ureteral reimplantation. Patients with unilateral VUR are thus an ideal selection group for EV reimplantation. Persistent advocates of bilateral, EV ureteral reimplantation report low incidence of urinary retention with modifications of the surgical dissection including limited ureteral dissection and mobilization, no ligation of the obliterated umbilical artery, no dissection in proximity to the obliterated umbilical artery and use of a low-coagulation-current needle-tip Bovie [58].
Laparoscopic iterations of these EV techniques have been subsequently developed in the hope of achieving a shorter period of convalescence and less postoperative pain [59-61]. Laparoscopic IV iterations of the open, Cohen ureteral reimplantation approach have also been developed [62-64]. Another recent laparoscopic variation includes robot-assisted laparoscopy using the Da Vinci platform for EV, laparoscopic ureteral reimplantation [65].
The 2010 AUA Clinical Guidelines consider surgical intervention as an “option” in the management of VUR [12]. The International Reflux Study in Children compared children with <11 years of age with dilating VUR randomly assigned to surgical intervention or CAP. This study showed no difference in “urographic" renal scars, UTI recurrence rate, or renal growth. However, there was an increase in febrile UTIs in the CAP group found after ten years of follow up [66]. Subsequently, they recommend that “patients receiving CAP with a febrile breakthrough-UTI be considered for open surgical ureteral reimplantation or endoscopic injection, with curative intent.”
CONCLUSION
The evaluation and management of VUR has changed significantly over the last two decades. The increased knowledge of the natural history of VUR and the identification of high-risk groups has tailored clinical decision-making and testing for VUR. RBUS and DMSA imaging are now frequently used in lieu of an immediate VCUG. We know that UTI is an important driving factor in the management of VUR and that infection control for UTI supersedes the significance of VUR. Another significant concept that is better understood in the management of VUR is the role of BBD. BBD is a significant risk factor for the prevalence of UTIs and development of secondary VUR. The evaluation for BBD in toilet-trained patients with VUR and UTIs should routinely be performed, as successful BBD management promotes downgrading or elimination of VUR.
New findings from the RIVUR trial show that CAP is associated with a substantially reduced risk of UTI recurrence among children with VUR but not of renal scarring. Lastly, surgical correction of VUR in selected patients remains a definitive treatment choice and there is a wide array of surgical techniques that can be tailored for the treatment of VUR.
ABBREVIATIONS
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
Declared none.


