All published articles of this journal are available on ScienceDirect.
Ischemic Etiology of Incontinence in Women: A Theory and A Review
Abstract
Incontinence in women is a major health problem. In a recent study, incontinence affects more than 10 million Americans, 85% of whom are women. Incontinence can result in an extremely altered life style, and is the main reason that women are admitted to nursing homes. Care for the incontinent patient represents an enormous expense nationally.
The emphasis of this review is on the relevance of estrogen-dependent ischemia to urethral smooth muscle tone and tonic α-adrenergic tension in the maintenance of continence in women. Additionally, the role of ischemia in bladder dysfunction will be explored. In general, the relative contribution of smooth muscle tension is greater in females than in males. It is clear from both animal and human data that during bladder filling there is a progressive increase in sympathetic stimulation of the urethra resulting in increased α-adrenergic tension.
INTRODUCTION
Incontinence in women is a major health problem. In a study prepared by Yankelovich and presented by The Bladder Health Council of the American Foundation for Urologic Disease, it was stated that incontinence affects more than 10 million Americans, 85% of whom are women. It affects 20% of women over the age of 45, the number increasing with age. Nearly 50% of all incontinence patients are dissatisfied with current treatment options, and 32% are very dissatisfied. 60% of women currently undergoing treatment report that they would try any novel therapy developed. Characterized by a high morbidity rate, incontinence is associated with a loss of self esteem, an inability to work, and is a major stress factor. It can result in an extremely altered life style, and is the main reason that women are admitted to nursing homes. Care for the incontinent patient represents an enormous expense nationally [1-4].
The emphasis of this review is on the relevance of estrogen-associated ischemia to urethral smooth muscle(SM) tone and tonic α-adrenergic tension in the maintenance of continence in women. Additionally, the role of ischemia in bladder dysfunction will be explored. Several investigators have estimated that 30-50% of total urethral pressure is contributed by SM tone [5]. In general, the relative contribution of SM tension is greater in females than in males. Abdominal pressure, pelvic organ prolapse and urethral hypermobility, vascular compression, connective tissue and elastic components of the urethra can contribute to changes in urethral closure pressure [6]. It is clear from both animal and human data that during bladder filling there is a progressive increase in sympathetic stimulation of the urethra resulting in increased α-adrenergic tension [7,8], and there is excellent direct evidence that α-adrenergic tension is an important factor in continence. Incontinence in general can result from urethral dysfunction, detrusor instability, detrusor insufficiency (with the resultant inability to empty effectively) and overflow incontinence. Prolapse and other anatomic findings such as urethral hypermobility can contribute to significant detrusor dysfunction and incontinence.
It is clear that in a variety of organ systems vascular insufficiency may lead to tissue changes and dysfunction. The nitric oxide pathway has been shown to allow for relaxation of SM of the corporal tissue, isolated bladder and urethral smooth muscle, and prostate. Impairment of relaxation is observed in the presence of ischemia. In addition, a reduction in circulating estrogen has been associated with urothelial and vaginal atrophy and disorders of the bladder from incontinence to increased urinary tract infections.
CELLULAR MECHANISMS OF URETHRAL SMOOTH MUSCLE TONE
Continence is dependent upon normal urethral physiology and anatomy; specifically, the maintenance of both active smooth muscle tone (i.e. the sustained response to α-adrenergic stimulation) and the integrity of the mucosal urothelium, which forms a water-tight seal during bladder filling [3]. In men, the prostate and the well developed external striated sphincter contribute significantly to total urethral tension and continence. Women, in contrast, do not have a prostate or a well developed external striated sphincter which completely encircles the urethra, and are much more dependent upon sustained (tonic) urethral tension for continence during bladder filling. In women, mucosal atrophy during periods of reduced circulating estrogen decreases the integrity of the urethral seal during bladder filling and results in stress incontinence.
The following physiological characteristics at the level of the urethra should be directly related to an increased incidence of incontinence: a) reduced adrenergic innervation, b) reduced α-adrenergic receptor density, c) reduced tonic response to adrenergic stimulation, d) reduced oxidative energy generation, and e) reduced participation of sarcoplasmic reticulum(SR) Ca+2 release during adrenergic stimulation.
The level of α-adrenergic tone is directly related to the density of adrenergic innervation and the α-adrenergic receptor density. Metabolically, α-adrenergic tone (sustained tension) is supported by oxidative metabolism, and is lost during periods of ischemia or hypoxia [9]. Additionally, α-adrenergic responses to stimulation are directly related to Ca+2 movements (Ca+2 entry into the cell; Ca+2i uptake into the SR, its storage and subsequent release). In this regard, we have evidence that Ca+2i release plays a major role in mediating the urethral response to both neuronal and α-adrenergic stimulation.
Similar to all smooth muscle contraction, bladder SM contraction depends upon a rise in [Ca+2]i [10] resulting from both Ca+2 influx from extracellular spaces and Ca+2 release from intracellular stores (Ca+2-induced Ca+2 release [CICR]) [11]. In the normal bladder, Ca+2 influx is mediated primarily, if not exclusively, through L-type (dihydropyridine-sensitive) Ca+2 channels; no N or T-type channels are believed to participate in bladder smooth muscle response to stimulation [12, 13].
Both extracellular Ca+2 influx and Ca+2 release from intracellular storage sites occur in response to autonomic receptor stimulation of the lower urinary tract [14, 15]. Although most of the studies described in this body of literature relate to bladder body SM, we now have direct evidence that CICR participates to a similar or greater extent in the response of urethral smooth muscle to both field stimulation and direct α-adrenergic stimulation by phenylephrine administration [16, 17]. Ovariectomy has been shown to decrease the contractile responses to field stimulation and phenylephrine, and it is believed to be through the inhibition of calcium storage and release from internal and external sites [18-20].
STUDIES OF SMOOTH MUSCLE TONE
Rats
Animal studies provide evidence that tonic α-adrenergic stimulation is a major factor in the maintenance of continence in women. For example, a reduction in urethral pressure is observed in rats treated with alfuzosin [21]. The importance of α- adrenergic function in the maintenance of urethral tension and continence during bladder filling is supported by this correlation between the clinical studies in women discussed below and basic studies in animals.
Smooth muscle function is altered by hormonal manipulation. Borda and co-workers found a decreased maximal response and sensitivity of contractility to prostaglandins E2 and E1 of bladder body strips from 20 day ovariectomized female rats compared to those from normal females [22]. Longhurst et al. observed decreased contractility of rat bladder strips in response to field stimulation and ATP two months after ovariectomy, and to field stimulation, carbachol, ATP, and KCl 4 months after ovariectomy. The data by Longhurst et al. suggested that strip responsiveness to ATP was more sensitive to loss of sex hormones than was responsiveness to muscarinic agonists. The ovariectomy-induced changes were prevented by estradiol replacement [23]. Ovariectomy and estrogen replacement studies show conflicting results, likely due to varying doses and preparations of estrogen, different time-courses and different species.
Rabbits
Autonomic Receptors
There are many studies examining changes in autonomic receptors in the lower urinary tract after ovariectomy and/or estrogen treatment; like the contractility data, results are conflicting. However, all agree that affinity of autonomic ligands for their receptors does not change. Polyestradiol treatment of mature female rabbits causes increased alpha-receptor density in the urethra [24], but estradiol treatment of immature females has no effect on alpha-, beta-, or muscarinic receptor density in the rabbit bladder base [25]. Similarly, Shapiro found that neither ovariectomy nor estradiol treatment affects muscarinic receptor density in the female rabbit urethra/base compared to mature, intact controls [26]. Three separate studies on autonomic receptors in the rabbit bladder body present conflicting results. Levin et al. found that estradiol treatment of immature female rabbits caused increases in bladder body alpha- and muscarinic receptor densities, but no change in beta-receptor density [27]. Shapiro found no change in bladder muscarinic receptor density after ovariectomy, and a decrease after ovariectomy + estradiol treatment [26]. Batra and Andersson ovariectomized rabbits and then, after 2 weeks, treated them with estradiol polyphosphate [28]. They observed a decreased muscarinic receptor density after ovariectomy, which was not reversed by estradiol treatment. These conflicting data may be the result of different concentrations and timings of hormonal manipulations.
Urethral Mass
Studies showing that estradiol treatment increases bladder and urethra mass [29, 30] support clinical findings of increased urethral mucosa thickness in postmenopausal women after estrogen therapy [16, 31, 32].
Detrusor Contractility
Studies examining the effects of hormone treatment on bladder contractile function have produced conflicting results. Short-term (4 days) high-dose treatment of immature female rabbits with estradiol caused increased contractile responses of bladder body strips to epinephrine, ATP, methoxamine, and bethanechol compared to immature intact controls [27, 30]. However, Batra and Andersson found no change in sensitivity or maximal contractile response to field stimulation or carbachol of bladder strips from female rabbits treated with estradiol polyphosphate for up to 8 weeks compared to ovariectomized controls [28].
Blood Flow
In recent studies in our lab we have demonstrated that ovariectomy results in a significant decrease in blood flow to the bladder mucosa and detrusor using both quantitative measurement via fluorescent microspheres, and using real-time laser Doppler methodologies. The decreased blood flow is correlated with mucosal hypoxia, mucosal apoptosis, and mucosal and detrusor smooth muscle atrophy, and increased mucosal permeability. Estrogen administration to ovariectomized rabbits resulted in the relief of hypoxia, mucosal hyperplasia and restoration of the mucosal permeability barrier [33-39]. Specifically, in our chronic studies [33, 34, 36], female rabbits were divided into three groups. The rabbits in group 1 received sham operations (Shm), group 2 received ovariectomies (Ovx) for two weeks, and group 3 received ovariectomies + estrogen therapy at 1mg/kg/week via Alzet pump (Ovx +E) beginning 1 week following ovariectomy and continuing for 5 weeks. At 6 weeks following ovariectomy various studies were performed on 6-8 rabbits per group. The results can be summarized as follows: Ovx resulted in: 1) a significant decrease in bladder weight 2) significant decrease in blood flow to the bladder mucosa and smooth muscle (Fig. 1A); corresponding with a decreased vascular density (Fig. 1B); 3) a thinning of the bladder mucosa. 4) a decrease in the smooth muscle / connective tissue ratio resulting in a decreased compliance. 5) decreased contractile responses to field stimulation, carbachol, and KCl, whereas estrogen administration resulted in a significant increase in contractile responses. 6) Ovx induced a generalized hypoxia of the mucosa and submucosal elements, which were reversed by estrogen administration (Fig. 2). 7) Ovx stimulated mucosal and submucosal apoptosis, and increased mucosal permeability. 8) Ovx resulted in atrophy of the bladder smooth muscle, whereas estrogen administration resulted in significant hypertrophy of the smooth muscle (Fig. 3) and mucosal hyperplasia. 9) Ovx resulted in an increased incidence of uninhibited contractions observed during cystometry (personal observation).
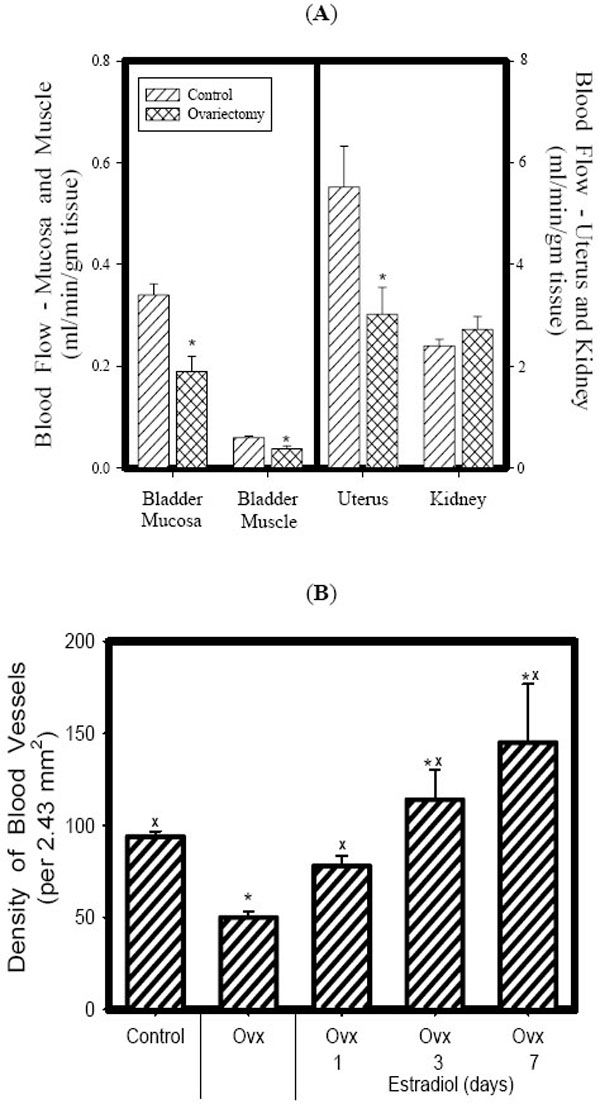
Effect of Ovx on blood flow (A). Estrogen supplementation restored both blood flow and vascularity to control levels within 7 days (B). Each bar is the mean +/- SEM of 4-6 individual rabbits. * = significantly different from control; x = significantly different from Ovx.
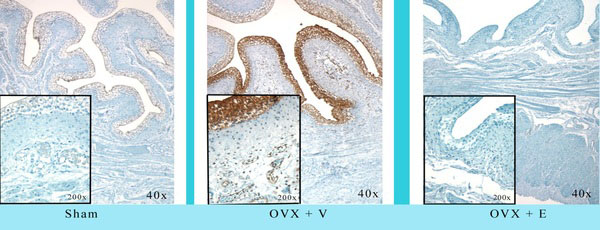
Effect of Ovx and Ovx + estrogen on hypoxia using hypoxyprobe-1 immunohistochemistry. Ovx resulted in diffuse hypoxia within the mucosa and submucosa; estrogen reversed the hypoxia completely. Reduced from 40X.
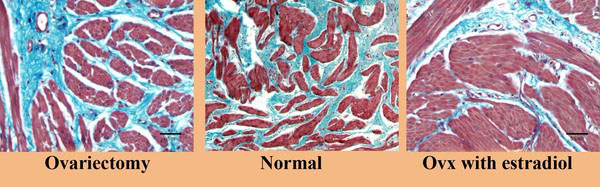
Effect of Ovx and ovx + estrogen on bladder structure. Ovariectomy resulted in atrophy of the smooth muscle compartment whereas estrogen administration resulted in significant hypertrophy of the smooth muscle. Reduced from 200X.
In a study recently published, we looked at the effects of cycling estrogen from low to high by first performing a bilateral ovariectomy; at two weeks we began estrogen administration via the implantation of a slow release tablet (1 mg/kg/day). At the end of two weeks estrogen administration we removed the tablet to create a second period of low estrogen. At the end of two weeks, the estrogen tablet was restored for a second period of estrogen administration. The results demonstrated a cyclical effect on bladder weight and urethral thickness (Fig. 4). Histologically, in both muscle and mucosal compartments, there was a cyclical shift from atrophy (during low estrogen periods) to hypertrophy (muscle) and hyperplasia (mucosa) during high estrogen periods [36]. Similar results were observed for vascular density (Fig. 5) for the bladder body; however, the vascular density for the urethra did not change. Similar results were shown for the smooth muscle / collagen ratio. That is, for the bladder body there was a clear cycling; the ratio significantly decreased during low estrogen periods and significantly increased during periods of high estrogen (Fig. 6).

Effect of cyclical estrogen on bladder mass and urethral cross sectional area. Each bar is the mean +/- SEM of 4-6 individual rabbits. x = significantly different from control; xx = significantly different from ovx + E; **= significantly different from ovx + E – E. p < 0.05.
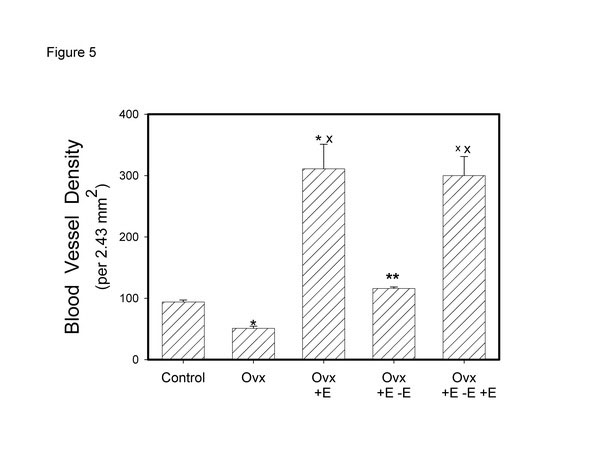
Effect of cyclical estrogen on vascular density. Each bar is the mean +/- SEM of 4-6 individual rabbits. * = significantly different from control; x = significantly different from ovx; ** = significantly different from ovx + E; xx = significantly different from ovx + E – E; p < 0.05.
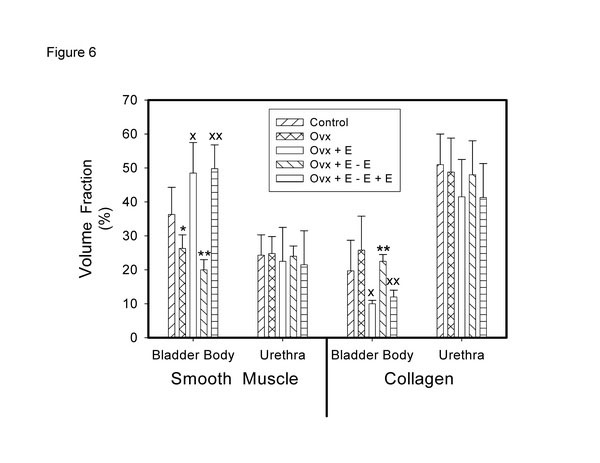
Effect of cyclical estrogen on smooth muscle – collagen ratio. Each bar is the mean +/- SEM of 4-6 individual rabbits. * = significantly different from control; x = significantly different from ovx; ** = significantly different from ovx + E; xx = significantly different from ovx + E – E; p < 0.05.
Our studies demonstrate clearly that ovariectomy (reduced circulating estrogen) resulted in decreased blood flow to the mucosa and to a lesser extent to the smooth muscle, hypoxia of the mucosa, submucosa, and vascular endothelium, apoptosis, atrophy of the smooth muscle compartment and increased collagen deposition, decreased compliance, increased permeability of the urothelium, contractile dysfunction and an increased incidence of uninhibited contractions.
CLINICAL STUDIES
Autonomic Receptors
Varied human clinical studies support the role of smooth muscle tone and function in incontinence. There is a considerable base of clinical evidence in men and women which demonstrates that α-adrenergic stimulation (especially using chronic slow-release therapy) increases both maximal urethral pressure (MUP) and maximal urethral closure pressure (MUCP), significantly reducing both stress and sphincteric incontinence. Specifically, Diokno and Taub reported that ephedrine sulfate treatment was effective for sphincteric incontinence in 27 out of 38 patients [40]. Awad et al. demonstrated that norepinephrine administration resulted in a significant increase in both MUP and MUCP [41]. Several clinical studies have demonstrated that the α-agonist, phenylpropanolamine, is an effective therapeutic agent for stress and sphincteric incontinence [42, 43]. Alternately, there is considerable evidence that anti-hypertensive therapy with α-adrenergic antagonists, such as prazosin, can induce moderate to severe stress incontinence [42-45]. In normal healthy women, prazosin administration resulted in a significant reduction in urethral closure pressure [46]. Terazosin produces lower urethral closure pressure in human females [47].
Estrogen and Progesterone
It is clear that estrogen plays a role in both the lower urinary tracts of animal models and human models. Estrogen and progesterone receptors have been shown to be present in the lower urinary tract, vagina, and pelvic musculature. The female genital and lower urinary tracts both arise from the urogenital sinus and are both sensitive to the effects of sex steroid hormones. Deficiency of estrogen as found in menopause leads to atrophy of the urethra, bladder, etc. [48]. This has been found to be associated with urinary frequency, nocturia and infection, although the exact role is unclear. The effects of estrogen therapy vary according to the route of administration, systemic therapy improves incontinence episodes and first sensation to void, however it appeared to worsen nocturia. As opposed to local estrogen therapy, which seems improves frequency, urgency, urge incontinence, first sensation to void, and bladder capacity [48, 49].
Both androgen and progesterone receptors are expressed in the lower urinary tract. Progesterone receptors have been reported in the bladder, trigone, and vagina, although their presence may depend on estrogen status. Androgen receptors are present in both the bladder and urethra. The role of both these receptors has not yet been elucidated. The mechanism of estrogen may include the following: (1) increasing urethral resistance, (2) raising the sensory threshold of the bladder, (3) increasing α-adrenoreceptor sensitivity in the urethral smooth muscle (although animal data exists to the contrary above), and (4) promoting β3adrenoceptor–mediated relaxation of the detrusor muscle [48]. In one study, it was found that in women estrogen treatment influences collagen synthesis and directly affects collagen metabolism in the lower genital tract. The composition of collagen composition also changes [49].
If manipulating hormone levels in animals alters lower urinary tract blood flow and function, one wonders whether physiologic cycling of estrogen contribute to voiding dysfunction over the years. In other organ systems it is clear that alterations in the level of circulating hormones that occur normally in women can alter mucosal and smooth muscle function. For example, asthma is significantly more prevalent among women and women are affected more severely by asthma than are men [50-52]. Asthma has been shown to be significantly more severe immediately prior to menses and less severe immediately after menses. In a recent study by Skobeloff, severity of asthma was directly compared with plasma levels of estrogen and progesterone [53]. The results demonstrate that the symptoms of asthma were least severe during the sustained high estrogen levels observed during the post-ovulatory period and substantially more severe during the low-estrogen period immediately pre-ovulatory period. Although the etiology of asthma is not fully understood, there is a definite relationship between asthma and inflammatory stimulation of the bronchial mucosal surface.
Mucosal cell structure and metabolism are under direct hormonal control (estrogen and progesterone). The normal alterations in women's hormonal levels could potentially increase mucosal sensitivity to stresses such as distension, ischemia, and hypoxia. Experimentally, it has been long recognized that alterations in sex hormones can have profound effects on smooth muscle and mucosal structure and function, in that female factors lead to variable responses in otherwise well controlled animal experiments [54-56]. The basic strategy used to "normalize" these hormone-induced alterations has been simply to exclude females from scientific studies, both at the experimental (animal) and clinical levels. As mentioned above, recent studies in our lab have confirmed the marked effects of alterations in estrogen on bladder mucosal structure and properties in rabbits [33-39]. The following is a brief discussion of the effects of hormonal alterations on lower urinary tract function.
In nulliparous premenopausal women, it has been found that there is a mid-cycle and early luteal-phase increase in the functional urethral length as measured by urethral pressure profile [57]. This occurs in tandem with increased plasma estradiol. The vascular supply to the lower urinary tract plays a significant role in providing urinary continence to women. As mentioned previously, the major target for ischemia and hypoxia is cellular metabolism. Blood flow to the lower urinary tract, as well as to the vagina and uterus, can be affected significantly by alterations in estrogen and progesterone [58, 59]. Estrogen has been shown to increase blood flow to the bladder and urethra whereas progesterone can block this effect [60, 61]. The demonstration that blood flow can be modulated by alterations in sex hormones, together with the finding by Irwin and Galloway that blood perfusion is reduced during distension in IC patients [62], support the importance of reduced blood flow in this gender-related disorder. In a study of 17 awake male patients who underwent saline cystoscopy while undergoing analysis of bladder blood flow by laser doppler flow of the posterior bladder wall. It was found that at capacity bladder blood flow is significantly decreased. After bladder drainage, there is rebound blood flow and reperfusion, suggesting bladder ischemia leads to architectural changes of the bladder wall [63]. Also, in a study by Tsai et al., it was found that women who were on transdermal hormone replacement therapy were found to have decreased vascular impedance on Duplex Doppler Ultrasonography of the uterine, internal carotid, ophthalmic and bladder wall arteries [64]. In a study on young women with recurrent urinary tract infections (UTI), application of local estrogen cream lead to decreased resistive indices and increased end diastolic blood flow velocity in 2 bladder arteries as compared to before starting the treatment. These patients were also found to have an increase of the infection-free period between UTIs. In a follow-up period of 11 months after treatment, 24/30 patients reported no symptoms of cystitis [65].
Estrogen reduction during menopause induces mucosal atrophy and other degenerative changes affecting bladder and urethral function, which can be at least partially reversed by estrogen therapy [16, 31, 32]. Functionally, mucosal atrophy might increase the susceptibility of post-menopausal women to urinary tract infections due to disruption of the mucosal barrier [66]. In addition, yeast and bacterial adherence to buccal and other mucosal surfaces have been shown to be significantly altered during the menstrual cycle [60, 61, 67].
In a meta analysis by Cardozo et al., 11 randomized controlled studies on over active bladder and the effect of either systemic or local estrogens were analyzed. Overall, estrogens (local and systemic therapy) were associated with alleviating symptoms of overactive bladder. Systemic estrogen therapy only had a significant improvement in nocturnal frequency but is associated with worsening nocturia as opposed to vaginal estrogen which shows benefit in all symptoms analyzed [49].
Maintenance of the integrity of the mucosa is dependent upon cellular metabolism. Although there are no studies on the effects of hormonal alterations on bladder mucosal metabolism, there is information on the effects of female sex hormones on both gastrointestinal and buccal mucosal metabolism and cellular function. Gastric enzyme synthesis and secretions, and buccal enzyme synthesis and activities are modulated by alterations in estrogen and progesterone levels during menses [68-71]. The demonstration that alterations in estrogen and progesterone can significantly affect mucosal cellular metabolism, enzyme synthesis and secretion, and metabolic function is relevant to the hypothesis that urothelial metabolism and cellular function can be significantly altered by altered circulating levels of these hormones. Azpiroz et al. described dysuria in Irritable Bowel Syndrome (IBS) patients and Cukier et al. found patient with IBS were more likely to experience nocturia, urgency, and some forms of urinary urge incontinence [72,73]. The ultimate effect on lower urinary tract function is yet to be characterized.
Ageing
As mentioned previously, incontinence associated with ageing is a major health problem [1, 2]. In a study by Lepor et al. in 1993, they found that the AUA symptom score of both aging men and women, ages 59 – 79, worsened as they aged [74]. This may be related to pelvic ischemia. We hypothesize that incontinence (in relation to ageing) is in part related to a generalized decrease in the metabolic energy available to sustain urethral tone, in addition to the post-menopausal hormonal alterations which occur in women. Reduced energy supply causes a progressive decrease in the tonic response of the urethra, the loss of α-adrenergic tonic tension, and a decreased urethral pressure, resulting in incontinence. The increased smooth muscle "fatigue" is concurrent with changes in the detrusor's ability to empty the bladder completely; the resultant progressively increasing residual volume is also associated with the effects of ischemia on lower urinary tract function. As women age, flow rate, voided volume, and bladder capacity decrease and post void residual urine increases. This is suggestive of increasing fibrosis of the bladder wall with aging. Maximum isometric detrusor pressure and maximum pressure at peak flow are found to be preserved. Female urethral pressures are also found to decrease with age [75]. Supporting this supposition are the studies by Lin et al. who demonstrated that in ageing rats there was a progressive decrease in the contractile responses to repetitive autonomic stimulation (increase in fatigue) which was directly associated with both a decrease in the metabolic energy available to support the contractile responses, and a decrease in mitochondrial enzyme activities [76, 77]. Thus, in association with ageing, there is a progressive reduction in the rate of high energy phosphate generation (during periods of stimulation) that significantly increases the rate of fatigue of the bladder smooth muscle. Previously, we demonstrated a significantly reduced ability of isolated whole bladders from aged rabbits (as compared to those from young rabbits) to generate pressure and empty efficiently. The rate of power generation, the percent emptying, and the amount of work performed by bladders from old rabbits were significantly less than bladders from young rabbits. In addition, there was a marked reduction in the ability of the bladders isolated from old rabbits to respond to an increase in outlet resistance [78, 79]. Our studies support the theory that ageing is associated with a progressive reduction in the ability of the bladder to sustain increased tension.
CONCLUSIONS
The overall conclusion from the studies reviewed is that modulation of circulating female sex hormones (especially estrogen) can have profound effects on bladder and urethral structure, mass, blood flow, tissue oxygenation, autonomic receptor density, distribution, and responses. The evidence is growing that ovariectomy induces hypoxia and ischemia and that hypoxic/ischemic damage to bladder nerves, smooth muscle, and the mucosa can result in significant bladder dysfunction [80]. We further hypothesize that physiologic cycling of estrogen can lead to cyclic ischemia, leading to lower urinary tract dysfunction and incontinence. Acute ischemia from childbirth, chronic ischemia related to vascular disease or hormonal withdrawal in menopause can also play a role.
ACKNOWLEDGEMENTS
This material is based upon work supported in part by the Office of Research and Development Medical Research Service, Department of Veteran’s Affairs, and in part by NIH grant RO-1-DK 067114, and in part by Capital Region Medical Research Foundation.


