All published articles of this journal are available on ScienceDirect.
Autonomic Nervous System Activity in Patients with Lower Urinary Tract Symptoms Secondary to Benign Prostatic Hyperplasia Estimated by Heart Rate Variability
Abstract
Introduction:
Aging induces autonomic nervous system (ANS) dysfunction with increased sympathetic drive. Benign Prostatic Hyperplasia (BPH) is responsible for lower urinary tract symptoms (LUTS), and its pathogenesis is complex. Exemplified by previous studies, the probably cause of BPH and secondary LUTS is due to the overly active sympathetic ANS resulting from aging.
Objectives:
The aim of our study was to estimate the ANS activity in BPH patients with LUTS using frequency domain analysis parameters of heart rate variability (HRV). Additionally, the relationship of ANS activity to the subjective measures of LUTS, and the objective measures of BPH, as well as the biochemical and biometrical variables, were investigated.
Material & methods:
The study was performed on 30 men with LUTS secondary to BPH. The cohort of patients was asked to complete IPSS and quality of life questionnaires. We performed biometrical, biochemical measurements and urological estimations. Additionally, a serum sample was obtained for Prostate Specific Antigen and its derivatives analysis. ANS activity was assessed by HRV measurements in resting conditions, after simulation with deep breathing and by the tilt up test. In the HRV recording, frequency domain analysis parameters were calculated according to fast Fourier transformation and the correlation for ANS activity parameters vs BPH variables were analyzed. The following HRV parameters were taken into consideration: VLF (power of very low frequency), LF (power of low frequency), LFnu (LF power in normalized units), HF (power of high frequency), HFnu (HF power in normalized), LF/HFnu (normalized ratio of LF power to HF power), PSD (total power of spectrum of the RR variability).
Results:
All participants (mean age 65 ± 7 years) presented moderate LUTS (IPSS = 14 pts.; QofL = 3 pts.) with Qave.= 7,4 ml/sec. and PVR = 48 ± 45 ml. Normalized values of LF and HF were 60.86 ± 18.96 [%] and 39.14 ± 18.96 [%], respectively. VLF - 238.75 ± 214.96 [ms2]. LF/HF ratio and its normalized value were 2.97 ± 3.04 [1] and 1.57 ± 1.40 [1], respectively. In response to deep breathing, significant increases of LF, LFnu, LF/HF, LF/HFnu and total power of HRV spectrum and a decrease of HFnu were observed. The E/I ratio was 1,12 ± 0,08. During the tilt up test, VLF, LFnu, LF/HF, and LF/HFnu were increased, while HFnu decreased. The 30/15 ratio was 0,98 ± 0,05. The observed strong correlations are as follows: 1) between prostate enlargement and HFnu and LFnu power; 2) between total PSA level and LFnu, HF, HFnu; 3) between free/total PSA ratio and LF/HFnu; 4) between PSA density of the transition zone and HF; 5) between plasma noradrenalin level and HF; 6) between age and LFnu, HFnu, LF/HF and LF/HFnu; 7) between plasma adrenaline level and prostate enlargement: prostate length and transition zone height.
Conclusions:
These results demonstrate the sympathetic overactivity of ANS at rest in patients with BPH and LUTS. It is also suggested that in the pathophysiology of BPH, the heighten activity of the sympathetic ANS, and parasympathetic drive are important.
INTRODUCTION
Disturbances of the autonomic nervous system (ANS) function play a pivotal role in some cardiological and gastrointestinal diseases (ventricular arrhythmias, motility disorders, etc.) [1, 2], however, little is known about ANS activity in patients with benign prostatic hyperplasia (BPH) and other urological states like an overactive bladder (OAB). Animal models and human research suggest that ANS is involved inthe pathogenesis of BPH, and OAB [3, 4]. The aetiology of BPH is complex. The incidence of BPH increases with age. Aging also induces ANS dysfunction with increased sympathetic drive. Recent studies show that ANS dysfunction may be strongly involved in the pathogenesis of BPH and secondary lower urinary tract symptoms (LUTS), however, its pathophysiological mechanisms are still under investigation.
Heart rate variability (HRV) analysis is widely used to estimate the activity of the cardiovascular autonomic regulatory system, reflecting both sympathetic and parasympathetic components. Power spectrum analysis of HRV, from short-term ECG recordings, provides a tool for assessing quantitatively the effects of the different divisions of the ANS on the heart [5, 6]. The spectrum of HRV is usually divided into four different frequency components: total, HF, LF, VLF power. The HF power reflects the modulation of efferent parasympathetic (vagal) activity by ventilation. The LF component reflects both modulation of efferent parasympathetic (vagal) and efferent sympathetic activity by baroreflex activity. VLF constitutes most of the TP in the HRV and in part reflects: thermoregulatory mechanisms, fluctuation in activity of the renin - angiotensin system and the function of peripheral chemoreceptors. The unique phenomenon of sympathetic activity is the apparent autonomous rhythm (T-rhythm; 0.4 -1,2 Hz) associated with fear and arousal [7], however, the role of VLF component is still unclear. Various methods to measure HRV have been used for both clinical and research purposes in cardiology [6], but only a few studies have investigated HRV in patients with BPH [4, 8].
AIM
The aim of our study is to estimate the ANS activity in BPH patients with LUTS using frequency domain analysis parameters of HRV. Additionally, the relationship of ANS activity to subjective measures of LUTS, objective measures of BPH, as well biochemical and biometrical variables were investigated.
PATIENTS AND METHODS
The study was performed on thirty men, who complained of moderate LUTS, secondary to BPH (mean age 65 ± 7 years; IPSS = 14 pts.; QofL = 3 pts.; Qave.= 7,4 ml/sec.). All participants accepted the study protocol and provided written consent. The study was approved by the Ethical Committee of Jagiellonian University (Cracow, Poland). Patients who met any of the criteria listed below, were excluded from the experiment [9].
Excluding Criteria: 1) an episode of unstable angina pectoris, myocardial infarction, transient ischemic attack or a cerebrovascular accident within the last 6 months; 2) untreated ischemic disease, arterial hypertension, diabetes; 3) any known primary neurological conditions, such as Multiple Sclerosis, Parkinson’s disease or any other neurological diseases known to affect bladder function; 4) any prior medical or surgical intervention for BPH; 5) any prior experimental medical or surgical intervention for prostatic disease; 6) use of α1-blocker or β-blocker drugs for hypertension within the previous year; 7) phenylephrine, pseudoephedrine, imipramine or an anticholinergic or cholinergic medications within 4 weeks of the screening visit; 8) estrogens, androgens or any drug producing androgen suppression, including finasteride or anabolic steroids at any time during life; 9) any clinically significant renal or hepatic impairment; 10) serum prostate specific antigen (PSA) level greater than 10 ng/ml; 11) any requirement for the daily use of a pad or device for incontinence; 12) history or current evidence of: prostate or bladder carcinoma, pelvic radiation or surgery, urethral stricture or bladder neck obstruction; 13) active urinary tract disease (eg. urinary tract infections of any type) or a history of cystoscopy or biopsy of the prostate within 2 weeks prior to the screening visit or an imminent need for surgery; 14) documented bacterial prostatitis within the last year; 15) history of neoplasmatic disease; 16) history of complete urinary retention [9].
Study Protocol: All included patients were asked to complete IPSS (International Prostate Symptom Score) and quality of life (QofL) questionnaires to estimate the severity of LUTS. The score range of IPSS was from 0 to 35 pts. The level of LUTS severity: 0-7 pts. (mild), 8-19 pts. (moderate), 20-35 pts. (severe). QofL scale ranged 0 – 6 pts. Then the biometrical, biochemical and urological estimations were performed on all participants, as follows:
Biometrical Measurements: Abdominal obesitywas estimated using waist, hip circumference and waist-to-hip circumference ratio, as well as body mass index (BMI) and body area surface (BAS) measurements.
Biochemical Measurements: Serum catecholamine levels (adrenaline and noradrenalin) were measured in resting conditions before HRV recordings in all participants.
Urological Measurements: Patient medical history and physical examination, including digital rectal examination and urinalysis were obtained. In the transrectal ultrasound, the size of the prostate and transition zone of the gland were measured using width (W), height (H) and length (L). Width was identified on a transverse image, on the contrary length and height was identified on a perpendicular longitudinal plane. Prostate and transition zone volume were calculated using the formula W x H x L x 0,523. All participants performed uroflowmetry to estimate their average urinary flow rate (Qave.), and were subjected to trans-abdominal ultrasound to measure the post void residual volume (PVR). Additionally, 1 serum sample was obtained for Prostate Specific Antigen - PSA (total, free, free/total ratio) and PSA derivatives (PSA density, PSA density of transition zone) analysis.
Participates returned for a second visit to measure the autonomic nervous system activity, by the heart rate variability assessment (CNSystem – Task Force Monitor). Each subject underwent 5-minutes HRV recording at rest, during deep breathing (DB) test (6 breaths/min.) and during the tilt up test (TUT). Autonomic tests were performed according to procedures described by Ewing [10].
HRV: 5 minute ECG monitoring was performed. Then the ECG signal was subjected to frequency analysis (based on fast Fourier transformations). The following HRV parameters were taken into consideration [1]: VLF - power of very low frequency (0,0033 – 0,04 Hz), LF - power of low frequency (0,04 – 0,15 Hz), LFnu – LF power in normalized units LF/(TP – VLF) x 100, HF - power of high frequency (0,15 – 0,4 Hz), HFnu - HF power in normalized units HF/(TP – VLF) x 100, LF/HF – ratio of LF power to HF power, LF/HFnu – normalized ratio of LF power to HF power, PSD (TP) – total power of spectrum of the RR variability. Normalized units are used to emphasize the reciprocal action of the parasympathetic and sympathetic limbs of ANS and to minimize the effect of changes in total power on the values of LF and HF components.
Deep Breathing Test: 5 minute HRV recordings were obtained during regular deep breathing at a rate of 6 breaths per minute (5 seconds inspiration and 5 second expiration in each breath). The following parameters of the test were estimated: mean ratio of the longest RR interval during expiration to the shortest RR interval during inspiration calculated from 6 consecutive cycles (E/I ratio) and alterations of HRV parameters during deep breathing.
Tilt Up Test: The subjects were placed on a tilt table, tilted at 60° for a period of 15 seconds and it was maintained at that position for 5 minutes with constant HRV recording. The following parameters were obtained: the ratio between the highest RRI around the 30th heart beat and the lowest RRI around the 15th heart beat after standing-up (30/15 ratio) and alterations of HRV parameters during the test.
Statistical Analysis
The data obtained was expressed as mean ± standard deviations (SD). A one-way Wilcoxon test was used to estimate the differences within the evaluated HRV parameters. Also linear r-Pearson correlations were done for the ANS parameters vs the urological, biochemical and biometrical variables. Statistical significance was set at p ≤ 0.05 for all tests.
RESULTS
All participants (mean age 65 ± 7 years) presented with moderate LUTS (IPSS = 14 pts.; QofL = 3 pts.) secondary to bladder outlet obstruction caused by prostate enlargement, and it was confirmed in uroflowmetry (Qave.= 7,4 ml/sec., PVR = 48 ± 45 ml) and ultrasound (Table 2). Biometrical description and PSA levels estimations are shown in Tables 1 and 2, respectively.
Biometrical Character of Examined Men
| Parameter | Mean | SD |
|---|---|---|
| Age [years] | 66 | 7 |
| Height [cm] | 175 | 6 |
| Weight [kg] | 82 | 12 |
| Body Mass Index – BMI [kg/m2] | 26.92 | 3.82 |
| Waist [cm] | 101 | 9 |
| Hip [cm] | 104 | 6 |
| Waist-Hip ratio - WHR [1] | 0.97 | 0.05 |
| Body Surface Area - BSA [m2] | 1.97 | 0.15 |
Urological Profile of Examined Men
| Parameter | Mean | SD |
|---|---|---|
| Prostate size | ||
| Prostate width - W [mm] | 48.80 | 5.90 |
| Prostate height - H [mm] | 38.03 | 8.38 |
| Prostate length - L [mm] | 37.03 | 7.60 |
| Prostate volume - Prostate Vol. [ml] | 37.73 | 17.83 |
| Transition zone of prostate size | ||
| Transition zone width - W-TZ [mm] | 36.17 | 6.67 |
| Transition zone height - H-TZ [mm] | 30.40 | 6.70 |
| Transition zone length - L-TZ [mm] | 27.93 | 7.29 |
| Transition zone volume - TZ-Vol. [ml] | 18.10 | 9.60 |
| Lower urinary tract symptoms estimation | ||
| IPSS [pts.] | 14 | 8 |
| Quality of life (QofL) [pts.] | 3 | 1 |
| Uroflowmetry (UFM) | ||
| Average urinary flow rate - Qave. [ml/sec.] | 7.4 | 3.1 |
| Post void residual volume - PVR [ml] | 48 | 45 |
| Prostate specific antigen (PSA) and PSA derivatives estimation | ||
| PSA total - tPSA [ng/ml] | 3.15 | 2.78 |
| PSA free - fPSA [ng/ml] | 0.66 | 0.56 |
| PSA free/total ratio - f/tPSA [1] | 0.24 | 0.13 |
| PSA density - PSAD [ng/ml/cc] | 0.07 | 0.04 |
| PSA density of transition zone - PSADT [ng/ml/cc] | 0.17 | 0.11 |
Hormonal evaluation obtained increased levels of plasma catecholamine: adrenaline = 164,20 ± 70,84 pg/ml [norm: < 125 pg/ml] and noradrenalin = 2137,73 ± 1400,58 pg/ml [norm: < 600 pg/ml].
ANS Measurements: Frequency analysis of HRV at rest obtained power values: LF and HF - 246.63 ± 276.23 [ms2] and 212.69 ± 311.84 [ms2], respectively. Normalized values of LF and HF - 60.86 ± 18.96 [%] and 39.14 ± 18.96 [%], respectively. VLF - 238.75 ± 214.96 [ms2]. Total power of the HRV spectrum was 698.07 ± 729.10 [ms2]. LF/HF ratio and its normalized value was 2.97 ± 3.04 [1] and 1.57 ± 1.40 [1], respectively (Table 3).
HRV Parameters at Rest and During Autonomic Tests
| HRV Parameters | n | HRV At Rest | HRV Deep Breathing Test | p | HRV Tilt Up Test | p | |||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | ||||
| VLF [ms2] | 30 | 238.75 | 214.96 | 2328.62 | 8341.61 | 0.136 | 960.85 | 1929.18 | 0.004 |
| LF [ms2] | 30 | 246.63 | 276.23 | 1519.54 | 1640.09 | 0.001 | 374.18 | 457.50 | 0.228 |
| LFnu [%] | 30 | 60.86 | 18.96 | 77.26 | 15.79 | 0.001 | 65.99 | 14.75 | 0.024 |
| HF [ms2] | 30 | 212.69 | 311.84 | 296.47 | 411.66 | 0.214 | 203.97 | 394.06 | 0.393 |
| HFnu [%] | 30 | 39.14 | 18.96 | 22.74 | 15.79 | 0.001 | 34.01 | 14.75 | 0.024 |
| PSD (TP) [ms2] | 30 | 698.07 | 729.10 | 4144.63 | 8184.18 | 0.001 | 1539.01 | 2114.48 | 0.015 |
| LF/HF [1] | 30 | 2.97 | 3.04 | 15.14 | 23.18 | 0.001 | 3.44 | 3.34 | 0.047 |
| LF/HF(nu) [1] | 30 | 1.57 | 1.40 | 9.98 | 15.58 | 0.001 | 2.20 | 2.49 | 0.016 |
In response to DB, a significant increase of LF, LFnu, LF/HF, LF/HFnu and total power of HRV spectrum and a significant decrease of HFnu was observed. The E/I ratio was 1,12 ± 0,08 [normal value: ( 1,21; abnormal value: ≤ 1,21] (Table 3).
During the TUT, VLF, LFnu, LF/HF, LF/HFnu, and total power of HRV spectrum significantly increased, while HFnu significantly decreased. The 30/15 ratio was 0,98 ± 0,05 [normal value: ≥ 1,04; borderline value: 1,01 – 1,03; abnormal value: ≤ 1,] (Table 3).
Additionally, heart rate was faster during deep breathing and during the tilt up test compared to resting conditions. Diastolic and mean blood pressure significantly increased during the tilt up test, when compared to resting (Table 4).
Beat Mean Characteristic at Rest and During Autonomic Tests at Rest and During Autonomic Tests
| Beat Mean Parameters | n | At Rest | Deep Breathing Test | p | Tilt Up Test | p | |||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | ||||
| R-R intervals [ms] | 30 | 995 | 141 | 951 | 162 | 0.001 | 889 | 146 | 0.001 |
| Heart rate - HR [bpm] | 30 | 62 | 9 | 65 | 11 | 0.001 | 70 | 11 | 0.001 |
| Systolic blood pressure -sBP [mmHg] | 30 | 129 | 18 | 128 | 19 | 0.092 | 131 | 24 | 0.291 |
| Diastolic blood pressure - dBP [mmHg] | 30 | 83 | 12 | 82 | 12 | 0.092 | 91 | 13 | 0.001 |
| Mean blood pressure - mBP [mmHg] | 30 | 95 | 12 | 94 | 13 | 0.079 | 101 | 15 | 0.037 |
This investigation of the relationship of ANS activity to the subjective measures of LUTS, the objective measures of BPH, as well as the biochemical and biometrical variables obtained some strong correlations, which are as follows: 1) between prostate enlargement and HFnu and LFnu power (Fig. 1); 2) between total PSA level and LFnu, HF, HFnu; 3) between free/total PSA ratio and LF/HFnu; 4) between PSA density of transition zone and HF; 5) between plasma noradrenalin level and HF; 6) between age and LFnu, HFnu, LF/HF and LF/HFnu (Table 5).
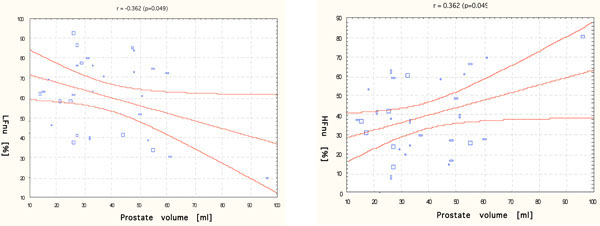
r - Pearson correlation between prostate volume and LFnu (left) and HFnu (right).
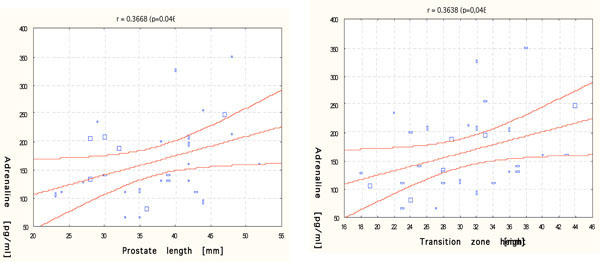
r - Pearson correlation between serum adrenaline level and prostate length (left) and transition zone height (right).
Correlation Between HRV Parameters and Age, Biochemical as Well as Urological Measurements
| R - Pearson Correlation | LFnu [%] | HF [ms] | HFnu [%] | LF/HF [1] | LF/HF (nu) [1] |
|---|---|---|---|---|---|
| Prostate volume [ml] | -0.362 | 0.278 | 0.362 | -0.191 | -0.172 |
| p=0,049 | p=0,136 | p=0,049 | p=0,311 | p=0,364 | |
| tPSA [ng/ml] | -0.351 | 0.383 | 0.351 | -0.244 | -0.247 |
| p=0,050 | p=0,037 | p=0,050 | p=0,193 | p=0,189 | |
| F/T PSA ratio [1] | -0.039 | -0.175 | 0.039 | -0.243 | -0.353 |
| p=0,838 | p=0,354 | p=0,838 | p=0,196 | p=0,050 | |
| PSADT [ng/ml/cc] | -0.244 | 0.426 | 0.244 | -0.171 | -0.198 |
| p=0,194 | p=0,019 | p=0,194 | p=0,367 | p=0,296 | |
| Noradrenalin [pg/ml] | -0.180 | 0.398 | 0.180 | -0.207 | -0.040 |
| p=0,342 | p=0,030 | p=0,342 | p=0,273 | p=0,836 | |
| Age [years] | -0.446 | 0.001 | 0.446 | -0.439 | -0.518 |
| p=0,014 | p=0,995 | p=0,014 | p=0,015 | p=0,003 |
Additionally, our results obtained a positive correlation between the serum adrenaline level and prostate enlargement: prostate length and transition zone height (Fig. 2).
DISCUSSION
These results show a significant importance of ANS activity in the pathogenesis of BPH. The HRV assessment generally demonstrates the over-stimulation of the sympathetic component of ANS at rest in patients with BPH and LUTS, when compared to healthy humans. Autonomic tests confirmed ANS dysfunction in this group of patients. In DB and in the TUT, impaired parasympathetic response of ANS to stimulation was observed.
Only a few previous animal studies have focused on ANS activity in rats with BPH. These studies revealed the significance of autonomic innervation of the prostate gland in the pathogenesis of prostate enlargement. McVary et al. [11] observed that autonomic denervation of the prostate leads to ipsilateral atrophy and contralateral hyperplasia of the ventral lobe in rats. Also, Wang et al. [12] studies on rats, showed a drop in the DNA and protein content of the ventral lobe of prostate, as a result for complete bilateral prostate denervation. It has been proven that spontaneously hypertensive rats have increased autonomic drive when compared to normotensive control rats, thus leading to BPH in young age and secondary LUTS (frequency) [11]. Many epidemiological studies show strong correlations between BPH and increased ANS drive. Meigs et al. [13] demonstrated that in patients with BPH, the incidence of obesity, increased insulin level and risk of diabetes and arterial hypertension are significantly higher than in healthy men. Sandfeldt et al. [4] observed that the risk factors for cardiovascular disease become more common when the prostate is very large. Additionally, prostatic stroma, but not secretory acini, receives rich noradrenergic innervation. Stimulation of these nerves causes contractions of prostate smooth muscle that are inhibited by α1-adrenoceptor antagonists that act at the (1L-adrenoceptor. Such actions underlie the clinical use of α1-adrenoceptor antagonists in BPH [14]. Additionally, the incidence of BPH increases with age. It is well known, that aging induces the ANS dysfunction with increased sympathetic drive, which is clearly observed in this correlation analysis of age and HRV parameters - LFnu, HFnu, LF/HF and LF/HFnu. Moreover, in this selective study, the patient’s higher systolic blood pressure is strongly related to prostate enlargement and biometrical features. Sandfeldt et al. [4] also demonstrated that men with a marked enlargement of the prostate had higher arterial pressure and blood glucose concentrations. These facts may confirm one of the accompanied patho-mechanisms of BPH pathophysiology based on the sympathetic over-activity.
To our knowledge, this present study is the first to assess HRV in patients with BPH during autonomic tests. The performed tests revealed not only sympathetic over-stimulation of ANS, but also impaired parasympathetic response of ANS. Due to a dense network of cholinergic fibres, which supplies both prostate epithelium and stroma, the dysfunction of parasympathetic ANS drive appears to be another important pathomechanism of BPH. The muscarinic receptor subtype mediating muscarinic agonist-induced smooth muscle contraction or enhancement of contractions evoked by nerve stimulation differs in different species. In humans, there is evidence for M1 receptors on the epithelium, M2 receptors on the stroma, and M2 and M3 receptors in some prostate cancer cell lines. Also, several recent investigations indicate that muscarinic receptors may also mediate or modulate normal, benign and malignant prostate growth [15].
The obtained results of HRV measurements and increased level of plasma catecholamine (adrenaline and noradrenalin) confirmed the sympathetic over-activity in patients with BPH and a positive correlation was concluded between adrenaline plasma level, and prostate length and transition zone height. McVary et al. [9] also observed a positive correlation between urinary noradrenalin level and SF36P (physical reaction to tilting up). Contrary to the results, no correlation with transition zone and volume of the prostate were observed.
Based on analysis of correlation between prostate volume (secondary tPSA level) and normalized values of LF and HF power spectra, it was postulated that prostate enlargement may be the result of dramatic changes in the autonomic profile. In patients with larger prostatic volume, the parasympathetic drive was intensified (decrease of LFnu and increase of HFnu values). Moreover, patients with a higher level of tPSA and PSA density of transition zone, have an increased parasympathetic ANS activity - increment of HF power value. This relationship is suspected to be a consequence of increasing cholinergic activity and its modulating effect on prostatic growth. This main finding which suggests that not only the sympathetic ANS over-activity, but also parasympathetic arm changes are important in pathophysiology of BPH.
These findings, followed by further quantitative analysis of the ANS dysfunction, may help in explaining the role of sympathetic over-stimulation and parasympathetic dysfunction in the pathogenesis of BPH as well as help in finding new treatment options for men with BPH. Understanding the ANS profile in BPH pathophysiology could be a necessary condition for the proper selection of patients with LUTS, who can benefit from pharmacological modulation of sympathetic or parasympathetic components of ANS activity.


