All published articles of this journal are available on ScienceDirect.
Long-Term Kidney Outcomes in High BMI Living Kidney Donors: A Narrative Review
Abstract
Background:
The shortage of transplantable organs has led to the expansion of the living kidney donor (LKD) pool to include overweight (body mass index [BMI] 25-30 kg/m2) and obese (BMI ≥30 kg/m2) donors. Higher BMI is a known risk factor for chronic kidney disease and progression to end-stage renal disease.
Methods & Materials:
We focused our review on long-term kidney-related outcomes (kidney function, hypertension, and proteinuria) among higher BMI LKDs. A PubMed search retrieved a total of 27 articles reporting on renal outcomes for obese LKDs. Of these, nine studies with ≥5 years of follow-up were selected for inclusion.
Results:
Five studies reported a decrease in kidney function at follow-up for higher BMI LKDs. Higher BMI was noted as a risk factor for hypertension in six studies. Proteinuria was reported in six studies, and a significant association was noted with higher BMI status in three.
Conclusion:
This review highlights the continued need for long-term documentation of kidney function and related outcomes in higher BMI LKDs.
1. INTRODUCTION
Chronic kidney disease (CKD) is a major public health problem. It is defined by the National Kidney Foundation as the presence of glomerular filtration rate (GFR) ≤60 mL/min for ≥3 months, pathological abnormalities or markers of kidney damage in blood or urine, or by abnormalities on imaging tests. Decreased GFR is linked to increased risk of cardiovascular events and all-cause mortality in the general population [1, 2]. Patients with end-stage renal disease (ESRD) on dialysis also carry an increased risk of cardiovascular and cerebrovascular disease-related morbidity and mortality [3].
Kidney transplantation (deceased and living donor) has paved way for better outcomes and quality of life for individuals with ESRD. It is well known that the wait-time for deceased donor kidneys has greatly increased. This, together with superior techniques and outcomes of living donor transplants, has led to a shift towards live donor transplantation.
Data from the Organ Procurement and Transplantation Network indicates a downward trend in living kidney donors (LKDs) since 2010 [4]. The reason for this change is unclear, but one result has been the expansion of the LKD pool to include categories that potentially confer higher risk on LKDs including overweight (BMI [Body Mass Index] 25 to <30 kg/m2) and obese (BMI ≥30 kg/m2) individuals. Such LKDs have increased by 12% (overweight) and 20% (mildly obese) every 5 years since 1999 [4].
We provide a background for higher BMI status risks, conferring kidney-related outcomes in the general and LKD population.
1.2. Risk Associated with Higher BMI in the General Population
1.2.1. Chronic Kidney Disease
Several studies in the general population have noted that higher BMI is associated with increased risk of developing CKD [5-7]. Overweight and obese individuals have been noted to carry up to 40% and 83% higher CKD risk, respectively [8]. Higher BMI status has also been associated with the faster progression of CKD and the development of ESRD [9-11].
Though the exact mechanism by which obesity can cause or worsen CKD is unknown, it is thought to be multifactorial. It may be mediated through the effects of underlying metabolic syndrome, development of hypertension, proteinuria, diabetes mellitus and possibly by glomerular hyperfiltration [12, 13]. The effects of adiposity that impact the kidneys directly via production of adiponectin, leptin, and resistin are also being considered [14]. In addition, the development of abnormal lipid metabolism, activation of the renin-angiotensin-aldosterone system, and increased production of insulin and insulin resistance are also potential offenders in causing kidney injury [14].
1.2.2. Hypertension
According to the Eighth Joint National Committee guidelines, hypertension is defined as systolic and diastolic blood pressure ≥140 mm Hg (≥150 mm Hg, if greater than 60 years of age) and ≥90 mm Hg, respectively [15]. Higher BMI status has been associated with the development of hypertension in both men and women in the general population [16]. For individuals with BMI>30 kg/m2, the adjusted risk of new hypertension was 2.23 (CI 1.75-2.84) and 2.63 (CI 2.20-3.15) for men and women, respectively [16].
1.2.3. Proteinuria
Higher BMI has been associated with the development of proteinuria or albuminuria. The National Kidney Foundation has defined albumubinuria as urine albumin to creatinine ratio >30mg/g [17]. In a study of AAs with hypertensive nephrosclerosis, Toto et al found that increased BMI was independently associated with increased proteinuria when adjusting for sex, age, blood pressure, creatinine, serum glucose, and uric acid [13].
The occurrence of proteinuria has also been noted in studies evaluating patients undergoing nephrectomy for medical reasons other than donation. Praga et al noted that proteinuria appeared an average of 10.1 years post-nephrectomy for other causes unrelated to kidney donation [5].
The study by Praga et al also found that patients who developed proteinuria post-nephrectomy showed a significantly higher baseline BMI than those that did not (31.6±5.6 kg/m2 vs. 24.3±3.5 kg/m2, p<0.001) [5].
1.3. Risk in Living Kidney Donors
1.3.1. Chronic Kidney Disease
A study by Mjoen et al noted higher rates of ESRD along with an increased risk for all-cause mortality and cardiovascular death for actual LKDs with mainly normal BMI compared to eligible LKDs who did not complete donation [18].
1.3.2. Hypertension
Development of hypertension has been noted among LKDs at long-term follow-up. Although they did not include BMI-specific outcomes, Fehrman-Ekholm et al found that approximately 50% of LKDs had hypertension at follow-up (14±9 years) [12].
1.3.3. Proteinuria
A higher incidence of proteinuria has been noted among LKDs with increased duration post-donation, irrespective of BMI status [12]. In addition, data from published literature in normal BMI LKDs, has shown a higher prevalence of type 2 diabetes mellitus with significant weight gain post-donation [19].
Data is still emerging on the long-term outcomes for LKDs. Given the expansion of the LKD pool to include higher BMI donors, we reviewed the literature to obtain studies in this population in hopes of gaining information that might inform our clinical practice. In general, CKD, hypertension, and proteinuria are chronic problems that require longer follow-up to effectively identify and manage. Therefore, it would be important to evaluate these kidney-related outcomes for several years post-donation to determine any additional underlying risks for higher BMI LKDs. We have focused our narrative review on nine studies evaluating long-term renal outcomes (≥ 5 years post-donation), including kidney function (CKD and ESRD), blood pressure, and proteinuria for higher BMI LKDs.
2. METHODS
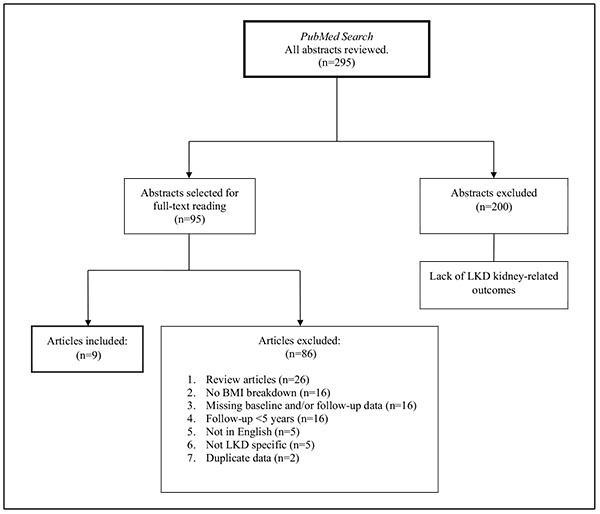
We conducted a PubMed literature search for our narrative review from inception to March 2nd, 2017. Our search strategy is depicted in Fig. (1).

Inclusion criteria:
1. Case-control and observation studies
2. Studies with LKD-specific data
3. Studies with renal outcomes
4. Studies with BMI-specific breakdown
5. Studies including baseline and follow-up data
6. Studies with follow-up ≥5 years
Exclusion criteria:
1. General Reviews
2. Non-English studies
3. Conference abstracts
4. Studies without LKD-specific data
5. Studies without renal outcomes
6. Studies which did not specify BMI
7. Studies without baseline and/or follow-up data
8. Studies with follow-up <5 years
The first author (KM) and second author (HC) individually screened the titles and abstracts. Abstracts were read by both HC and KM, with the authors initially synthesizing data individually and then discussing themes as a team. Articles were selected for full-text reading based on the inclusion and exclusion criteria listed above. Articles selected for full-text review were read in full by HC and KM, and decisions to include in the manuscript were made as a team. We also hand-searched reference lists for additional articles, but did not find any sources not captured by the original PubMed search.
3. RESULTS
Our PubMed literature search yielded 295 articles. From this search we selected 95 articles for further review. Details are included in the flow diagram in Fig. (2).
Out of the 95 studies, 27 reported on kidney-related outcomes for obese and overweight LKDs. Two of these studies were excluded due to duplicate data presented in articles already included in our review. Sixteen studies were excluded due to follow-up periods <5 years (3 days to 4.25 years). Nine studies meeting our inclusion criteria were selected for inclusion. Table 1 provides a description of the studies included in this narrative review.
| Author/ Study Design | Follow-up (years, ±SD) | Group | BMI (kg/m2) | N |
GFR estimation method/ Creatinine Clearance |
|---|---|---|---|---|---|
|
Nogueira [20]/ Prospective |
7.1± 1.6 | Control | <35 | 31 | MDRD |
| Study | ≥35 | 8 | |||
| Nogueira [21]/ Prospective | 6.8 ± 1.5 | Study* | ≥30 | 36 | MDRD |
| Locke [22]/ Retrospective | 10.7 median (26.8 max) | Control | <30 | 58,004 | CKD-epi |
| Study | ≥30 | 20,588 | |||
| Bello [23]/ Cross-sectional | 12.37 ± 7.60 | Control | <30 | 57 | Cystatin-C based formula of Rule |
| Study | ≥30 | 20 | |||
| Ibrahim [24]/ Prospective | 12.2 ± 9.2 | Study† | ≤30 | 180 | Iohexol |
| >30 | 75 | ||||
| Tavakol [25]/ Prospective | 11 ± 7 | Control | <30 | 82 | MDRD |
| Study | ≥30 | 16 | |||
| Taner [26]/ Prospective | 5.8 | Control | ≤35 | 16 | 125I-iothalamate clearance |
| Study | >35 | 11 | |||
| Kerkeni [27]/ Retrospective | 9.28 ± 6.3 | Control | <25 | 87 | Creatinine Clearance |
| Study 1 | 25–<30 | 62 | |||
| Study 2 | ≥30 | 40 | |||
| Gracida [28]/ Prospective | 6.725 ± 2.715 | Control | ≤30 | 422 | Creatinine Clearance |
| Study | >30 | 81 |
†Matched controls from National Health and Nutrition Examination Survey 2003-2004, 2005-2006
Nogueira (2009) et al conducted a prospective study examining the long-term outcomes for 39 African American LKDs [20]. They calculated kidney function using five different equations, including MDRD, Mayo Clinic Quadratic Formula, Body Surface Area Adjusted Cockcroft-Gault for Creatinine Clearance, using actual body weight, lean body weight, and the African-American Study of Hypertension and Kidney Disease Simplified GFR formula. They report their final analysis using the MDRD eGFR estimates. The LKDs were 34% males, with an average age of 37.4±11 years. The average BMI was 28.9±5.9 kg/m2 pre-donation and 31.1±6.3 kg/m2 at follow-up [20].
The study by Nogueira et al (2010) was a prospective study evaluating 36 obese LKDs seven years after donation [21]. They estimated kidney function using the MDRD eGFR equation. They compared the outcomes using the cohort of 255 LKDs from the study by Ibrahim et al. [24] The mean age at baseline was 45.1±10.8 years. LKDs were 22.9% male. The average BMI was 34.2±3.38 kg/m2 at baseline and 35.8±5.3 kg/m2 at follow-up [21].
Locke et al performed a national retrospective study of 119,769 LKDs linked the Centers for Medicare and Medicaid Services data to ascertain the risk of post-donation ESRD based on BMI among LKDs [22]. They calculated kidney function using the CKD-epi equation. Cumulative incidence of ESRD was estimated using Kaplan-Meier methods and compared between obese and non-obese LKDs. Risk of ESRD by obesity status at donation was estimated using Cox proportional hazards models adjusting for LKD age, sex, race, blood pressures, relationship to recipient, and baseline eGFR. The mean BMI at baseline was 32.7±3.2 kg/m2 for obese LKDs and 24.8± 2.9 kg/m2 for non-obese LKDs [22].
Bello et al conducted a cross-sectional study of 77 LKDs evaluating renal function and the presence of risk factors [23]. Two multivariate models were tested to evaluate the risk of CKD and the effect of hypertension. In each model, they evaluated the role of BMI on specific outcomes. The eGFR was calculated using the Cystatin-C based formula of Rule. The average BMI at follow-up was 26.65±5.04 kg/m2 [23].
In the prospective study by Ibrahim et al, hypertension, kidney function, general health status, and quality of life were evaluated in 255 LKDs [24]. GFR was measured by iohexol clearance and estimated by MDRD. LKDs were matched to controls from the National Health and Nutrition Examination Survey 2003-2004 and 2005-2006 [24].
Tavakol et al followed 98 LKDs with matched controls from the National Health and Nutrition Examination Survey 2005-2006 in a prospective study examining kidney-related outcomes [25]. The GFR was calculated using MDRD. They calculated total urine protein and albumin excretion (abnormal when values > 150 and 30 mg/d, respectively). Multivariate logistic regression and linear regression models helped determine the independent association between BMI at donation and kidney-related outcomes [25].
The prospective study of 46 LKDs by Taner et al evaluated the impact of older age, obesity, and hypertension on the compensatory response of the remaining kidney five years post donation [26]. The 11 obese LKDs and 9 hypertensive LKDs were compared to 16 standard LKDs. The mean BMI of obese LKDs was 38.1±2.2 kg/m2 compared to standard LKDs which 22.6±1.2 kg/m2. GFR was measured by 125I-iothalamate clearance [26].
Kerkeni et al retrospectively evaluated 189 LKDs and assessed both post-operative and long-term consequences of nephrectomy based on BMI [27]. Outcomes for overweight and obese LKDs were compared to those with BMI <25 kg/m2. The mean BMI at the time of donation was 26.5±4.8 kg/m2, with 33% of LKDs being overweight and 21% obese. Kidney function was assessed by creatinine clearance [27].
The study by Gracida et al evaluated long-term kidney-related outcomes in 628 LKDs. There were 81 LKDs who were obese prior to donation and 16 with hypertension [28]. They compared outcomes with 422 LKDs without risk factors. Mean BMI was 32.8 kg/m2 in the obese group and 24.7 kg/m2 in the standard group. The kidney function was measured using creatinine clearance and the method of GFR calculation was not mentioned [28]. They did not report hypertension or proteinuria outcomes at follow-up.
3.1. Higher BMI and Kidney Function (CKD and ESRD)
Having shown the data regarding increased risk of chronic kidney disease in the general higher BMI non-donor population, we will review the higher BMI LKD-specific data found in the studies selected for this narrative review. Five studies (range of follow-up, 7-20 years), noted significant decreases in kidney function for higher BMI LKDs.
Ibrahim et al evaluated long-term outcomes and risk of ESRD for LKDs over the course of 12.2±9.2 years [24]. The mean GFR was 84±13.8 ml/min per 1.73 m2 at baseline and 63.7±11.9 ml/min per 1.73 m2 at follow-up. They noted that 29% of LKDs had BMI >30 kg/m2 at follow-up. These LKDs were found to carry significantly higher odds of having GFR< 60 ml/min per 1.73 m2 (OR 1.12, CI 1.02-1.23, p=0.02) compared to lower BMI LKDs [24].
In the study by Nogueira et al (2009), outcomes for 39 African American (AA) LKDs were reported [20]. Pre-donation eGFR was 102.5±20.3 ml/min per 1.73 m2. They found a statistically significant greater absolute (40.1±7.3 vs. 28.3 ±17.1ml/min per 1.73m2) and relative decrement (39.8% vs. 26.2%) in GFR at follow-up for those who were morbidly obese compared to those with BMI<35 kg/m2. There were no BMI-stratified data available to compare outcomes for the normal, overweight, and obese LKDs within the BMI < 35 kg/m2 group [20].
Nogueira et al (2010) evaluated 36 obese LKDs (45.7% AA) [18]. The average eGFR was 91±20.3 ml/min per 1.73m2 at baseline and 63±15.3 ml/min per 1.73m2 at follow-up. They found a statistically significant overall drop in GFR from baseline to follow-up (29.2%) [21]. It is also important to note that the absolute decrement in GFR was similar for LKDs with BMI≥ 35 kg/m2 and obese LKDs with BMI< 35 kg/m2 (31.5±15.6 and 24.7±11.0 mL/min/1.73 m2, P=NS). Nogueira et al also compared certain demographic and outcome data for the obese LKDs in this study and the LKD cohort from the study by Ibrahim et al. [24] They noted that higher percentages of the obese LKDs in this study had GFR values <60 mL/min compared to the Ibrahim cohort (47.2% vs. 39.3%). Additionally, AA obese LKDs had a statistically significant greater absolute drop in GFR compared to non-AA LKDs (33.3±9.6 and 22.7±12.7 mL/min/1.73m2, respectively, p=0.016) [21].
The study by Locke et al reported long-term data examining outcomes and risk of ESRD for LKDs stratified by BMI ≥ 30 kg/m2 (n=20,588) and <30 kg/m2 (n=58,004) [22]. On multivariate analysis, obesity (HR 1.86, CI 1.05-3.30, p=0.04) and AA race (HR 4.62, CI 3.46-6.16, p<0.001) were the two risk factors that were significantly associated with the development of ESRD. The risk for ESRD post-donation increased 7% for every 1 unit increase in baseline BMI >27 kg/m2 [22].
In the study by Bello et al, renal outcomes and risk factors for 77 LKDs were evaluated.20 On multivariate analysis, higher BMI was found to have a statistically significant prevalence ratio for low GFR <60 mL/min at follow-up (PR 1.10, CI 1.05-1.15, p<0.0001) [23].
Four studies found no significant differences in kidney function between obese and non-obese LKDs at follow-up (range, 6-11 years) [25-28].
3.2. Higher BMI and Blood Pressure
The effects of higher BMI on the risk of hypertension in the general population have been reviewed. Hypertension data specific to higher BMI LKDs was available in eight studies in our review. BMI status was found to be associated with the presence of hypertension in six of these studies.
Ibrahim et al reported that higher BMI significantly increased the risk of hypertension (OR 1.12, CI 1.04-1.21, p=0.003) [24].
In the two studies by Nogueira et al (2009, 2010), 41% of LKDs were found to be hypertensive at follow-up [20, 21]. In the 2009 study, mean blood pressures at follow-up were 120.8±14.5 mm Hg and 79.7±9.3 mm Hg for systolic and diastolic, respectively. In the 2010 study, mean blood pressures at follow-up were 122.0±13 mm and Hg 77.3±7.4mm Hg for systolic and diastolic, respectively. Nogueira et al (2010) also noted that, although mean blood pressures were similar at follow-up, a larger percentage of obese LKDs in their study had a diagnosis of hypertension than the LKDs from the Ibrahim study (30.6% vs. 24.7%) [21].
At follow-up, Tavakol et al noted that the mean systolic and diastolic blood pressures were higher for obese LKDs and non-obese LKDs who became obese during the study [25]. A greater number of obese LKDs carried a hypertension diagnosis at follow-up. On multivariate analysis, obesity at donation was found to be a risk factor for the development of hypertension during the study (OR 4.02; CI 1.20- 13.00, p=0.021) [25].
On multivariate analysis, Bello et al found that higher BMI carried a statistically significant prevalence ratio for hypertension at follow-up (PR 1.11, CI 1.04-1.17, p=.0003). At the time of follow-up, 20 LKDs had hypertension, with a mean BMI of 30.41±5.88 kg/m2, compared to 57 without hypertension, with a mean BMI of 25.34±3.99 [23].
At baseline, in the study by Locke et al the mean systolic and diastolic blood pressures were higher for obese LKDs (124.1±13.1 mm Hg and 75.6±9.3 mm Hg, respectively) compared to non-obese LKDs (119.9±13.3 mm Hg and 72.9±9.4 mm Hg, respectively) [22].
Three studies either did not report comparison statistics [28] or did not find any differences in blood pressure relating to BMI status at follow-up [26, 27].
3.3. Higher BMI and Proteinuria
Risk of proteinuria has been discussed in the non-donor higher BMI population in the background section. Among the nine studies included in our current review, six studies presented data on proteinuria for higher BMI LKDs. The presence of proteinuria was noted to be significantly associated with higher BMI in three of these studies.
Tavakol et al found that the 24-hour urine protein was 80±30 mg/d at baseline and 146 ± 62 mg/d at follow-up. A greater percentage of obese LKDs (44%) had abnormal proteinuria compared to non-obese LKDs (18%) (P=0.03). On multivariate analysis, obesity at donation was a significant predictor of abnormal proteinuria at follow-up (OR 8.9, CI 1.1-70.0, p=0.039) [25].
Nogueira et al (2009) found that 18% of the AA LKDs developed proteinuria at follow-up [20].
In the 2010 study by Nogueira et al, 19% of subjects developed microalbuminuria. They noted that development of microalbuminuria appeared to correlate with greater decrements in GFR [21].
The remaining three studies found no association between BMI and the presence of proteinuria [24, 26, 27].
4. DISCUSSION
In our current narrative review evaluating kidney-related outcomes for higher BMI LKDs, we noted significant heterogeneity in the reporting of data among the nine studies, ranging from the study design, follow-up period, control groups used, method of kidney function estimation, to the reporting of proteinuria and hypertension. The strengths of the included studies were that they all had long-term follow-up periods (≥ 5years), reported kidney function based on BMI status, provided data on kidney function outcomes on higher BMI donors, and had a control group either within the study or utilized a reference group from the general population. We recognize that the limitations of our review are based on the heterogeneity of the published literature. There were inconsistencies in assessing and reporting additional outcomes such as hypertension and proteinuria. Among the nine studies included in this narrative review, six studies reported complete data on kidney function measurement, hypertension, and proteinuria.
Despite the heterogeneity in the reporting of different kidney related outcomes, we found five studies demonstrating decreased kidney function including ESRD correlating with higher BMI status. Among the four studies reporting no significant relationship between GFR and BMI, one showed higher incidence of hypertension and proteinuria for overweight and obese LKDs.
Accurate assessment of kidney function in this population both pre- and post-kidney donation is important. Among the nine studies included in this review, several different methods were used to estimate/measure kidney function: MDRD eGFR estimates (3), creatinine clearance (2), CKD-epi (1), Cystatin C (1), exogenous clearance markers (2). Measuring GFR using exogenous markers such as inulin, 125I-iothalamate, or iohexol clearance is considered the gold standard. Inulin clearance is cumbersome to perform; 125I-iothalamate or iohexol clearances can be conducted in a clinical setting, but these markers can be difficult to procure.
Given the fact that overweight and obese individuals are at higher risk for the development of CKD, it is necessary to assess GFR accurately as they are being increasingly considered in the LKD pool. Use of serum creatinine-based GFR estimating equations may not provide the most accurate results in higher BMI individuals due to hyperfiltration [29]. It would be essential to consider methods subject to less variability, such as the use of 24-hour urine creatinine clearance or measurement of the clearance of an exogenous filtration marker. Use of an estimating equation combining both Cystatin-C and creatinine has been proposed to overcome the limitations of equations based solely on serum creatinine [30]. With the ongoing expansion of the LKD pool, it would be ideal to incorporate the consistent use of one of these methods in future studies to report kidney function both at baseline and long-term follow-up.
A large number of the included studies (6 out of 8) evaluating hypertension noted higher BMI to be a significant risk factor. Proteinuria was reported in six studies, with 50% finding a significant association with higher BMI status. Hypertension and proteinuria are well known risk factors for CKD and need to be diagnosed in a timely manner and followed to institute appropriate therapeutic interventions in higher BMI LKDs. In addition, AA race was found to be an independent risk factor for decreased kidney function, highlighting the implications of such non-modifiable risk factors that need to be accounted for in donor selection.
Improvement in cardiovascular outcomes with short-term weight loss has been documented [31, 32]. The potential benefits of pre-donation weight loss need to be assessed in this population of LKDs In addition, the effects of sustained weight loss post-donation for overweight and obese LKDs also need to be evaluated in future studies.
It is also to be noted that out of the sixteen short-term studies evaluating kidney related outcomes in higher BMI LKDs (follow-up ranging from 3 days to 4.25 years) that we did not include in our current narrative review, 50% found a decline in kidney function. We believe there is a need to focus on long-term follow-up to document these outcomes given the expansion of the LKD pool to include higher BMI LKDs.
CONCLUSION
It would be therefore ideal for higher BMI LKDs to be followed by both their transplant centers and primary care physicians. Typically the follow-up protocol for LKDs currently in practice is to be seen at discharge, 6 months, 1 year and 2 years after donation. Given the potential higher risk of CKD in this expanded higher BMI LKD pool, a closer and longer-term follow-up would be essential. In addition, a multidisciplinary approach would allow for the early identification of potential complications, such as the development of metabolic syndrome, diabetes mellitus, hypertension, proteinuria and other risk factors of CKD. This would enable the timely implementation of effective management strategies in this high risk group. Our narrative review also highlights the need to conduct long-term prospective studies to assess and document accurate kidney function and important kidney-related outcomes in this expanded LKD pool of higher BMI LKDs, to enable risk assessment and to establish safe selection methods.
LIST OF ABBREVIATIONS
| LKD | = Living kidney donor |
| BMI | = Body mass index |
| CKD | = Chronic kidney disease |
| GFR | = Glomerular filtration rate |
| ESRD | = End stage renal disease |
| AA | = African American |
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
Kalyani Murthy has no relevant financial disclosures or conflicts of interest.
Hannah Caldwell has no relevant financial disclosures or conflicts of interest.
Mary Ann Simpson has no relevant financial disclosures or conflicts of interest.
ACKNOWLEDGEMENTS
Kalyani Murthy participated in research design, article search and review, and the writing of the manuscript.
Hannah Caldwell participated in article search and review and the writing of the manuscript.
Mary Ann Simpson participated in research design and the writing of the manuscript.


