All published articles of this journal are available on ScienceDirect.
Prevalent Practices in Male Anterior Urethral Stricture Management: A Survey
Abstract
Objective:
This article aimed to study the prevalent practices in the treatment and follow-up of urethral strictures. Moreover, the article aimed to investigate the present opinion among urologists as the treatment practices for urethral stricture disease are mostly dependent on the treating surgeon’s expertise and preference in the absence of widely followed standard guidelines.
Methods:
A questionnaire was formulated based on the responses from experts. From October to December 2020, all members of the urological society of India received a mailed questionnaire on a web-based survey platform. Practicing urologists from across the country are among its members.
Results:
A total of 2554 urologists were contacted by mail, and 282 (11%) urologists provided a response. The majority were doing 5 to 15 urethroplasties a year. A newly diagnosed short segment bulbar urethral stricture would be a candidate for a visualized internal urethrotomy (VIU), according to 69.5% of urologists. Recurrence after one attempt at VIU prompted 80% to switch to urethroplasty as the next choice. During preoperative evaluation, 100% of respondents wanted a retrograde urethrogram, 74% deemed micturating cystourethrogram necessary, and 60% wanted a urethroscopy. At discharge, the majority preferred silicone Foley catheters, and the preferred size was 16F (54%). The patients were discharged with both suprapubic catheter and Foley in situ by most urologists. During follow-up, 60.99% wanted uroflowmetry, and the duration of follow-up varied.
Conclusion:
This survey on urethral stricture management amongst urologists shows that there is wide variation in the management of stricture urethra. This survey underscores the need for large-scale, long-term studies to formulate a guideline for the management of urethral stricture disease, thereby bringing uniformity in the care provided.
1. INTRODUCTION
Urethral stricture is a common disease that has an estimated incidence of about 200–1,200 cases per 100,000 male population, with the incidence being much higher in people aged ≥55 years [1]. Strictures can be located either anterior or posterior to the urethra. They differ both in their location as well as in the pathogenesis. The vast majority of urethral strictures are anterior (92.2%), with most of these occurring in the bulbar urethra (46.9%) [2].
Until around a half-century ago, treatment of urethral strictures involved dilatation or urethrotomy. Both were blind procedures and needed a high degree of skill on the part of the treating surgeon, and the results were mostly suboptimal and short-lived. [3] Present-day urethral strictures can, however, be treated by multiple different methods, ranging from endoscopic incision to open reconstruction, with the aim of all surgeries being to achieve a patent urethra to facilitate normal voiding. Treatment selection is not completely standardized, with the ultimate choice depending on the cause of the stricture, length and location of the stricture, and most importantly, the surgeon’s preference and experience. Not only is the primary treatment not well standardized, but there also appears to be no consensus on how to follow up these patients, especially because one major problem with stricture treatment is recurrence [4]. Most of these patients will be followed up with a combination of questionnaires, uroflowmetry (UF), and invasive procedures such as retrograde urethrogram (RGU) and cystoscopy. The follow-up instruments that are actually useful are, by and large, decided by the treating surgeon based on his/her personal experience [5]. Another major barrier in developing standardized practices is that the very definition of failure or success after urethroplasty does not have consensus [6,7].
Therefore, in this study, we attempt to analyze the prevalent practices in treatment and follow-up of urethral strictures in an attempt to understand what the present opinion is among urologists. Moreover, we try to highlight the areas where ambiguity exists so that future studies can focus on solving them.
2. STUDY METHODS
A pilot survey was conducted in which five renowned urethral reconstructive surgeons were asked about the clinical practices of treating urethral strictures that they think vary widely among urologists. A questionnaire with 30 questions was formulated based on the responses from these experts. Conflict among the experts on questions to be included was dissolved by giving higher weightage to the opinion of the urologist with greater experience. Then a nationwide survey was conducted among urologists registered with the Urological Society of India. The survey was conducted from October 2020 to December 2020 as a mailed questionnaire on a web-based survey platform.
Personalized e-mails were sent to the urologists, with a description of the objective and implications of the study. Instructions and a link to participate in the survey were included. All e-mails detailed the purpose of research, its academic nature, and assurance of anonymity and confidentiality. Participation was voluntary, and the participants had 15 days to answer the online survey. Those willing to participate in the study were required to answer all the questions compulsorily, picking the most appropriate response for each question.
SPSS software version 23 (SPSS Inc., Chicago, Illinois, USA) was used for data entry and analysis
3. RESULTS
A total of 2554 urologists were contacted by mail, out of which 282 (11%) urologists provided a response. The demographic characteristics of these respondents have been summarised in Table 1. Overall, 138 of the responding urologists were involved in private practice. A further 128 were practicing in teaching institutions, among whom 57 were urology trainees. Concerning urology practice, the majority were relatively less knowledgeable as 104 respondents had less than five years of experience. A further 54 had between 5 and 10 years of experience. Seventy urologists had greater than 20 years of experience in urology practice (Table 1).
| Workplace | Respondents | Percentage |
|---|---|---|
| Private Sector Healthcare | 138 | 48.9% |
| Government (Public Sector) | 12 | 4.2% |
| Medical College (Teaching Institute) | 128 | 54.4% |
| Other | 4 | 1.4% |
| Position | ||
| Consultant Urologist | 130 | 46% |
| Consultant urologist but not the lead urologist in the team | 30 | 10.6% |
| Faculty in a teaching institute | 65 | 23.1% |
| Resident | 57 | 20.2% |
| Experience | ||
| <5 | 104 | 36.9% |
| 5 to 10 | 54 | 19.2% |
| 11 to 15 | 29 | 10.3% |
| 16 to 20 | 25 | 8.9% |
| >20 | 70 | 24.8% |
| Stricture Caseload per Month | ||
| <5 | 107 | 37.9% |
| 5 to 10 | 122 | 43.3% |
| 11 to 20 | 35 | 12.4% |
| >20 | 18 | 6.4% |
| Urethroplasties done per year | ||
| None | 10 | 3.6% |
| <5 | 64 | 22.7% |
| 5 to 15 | 108 | 38.3% |
| 15 to 25 | 47 | 16.7% |
| >25 | 53 | 18.8% |
One hundred and seven reported to be treating <5 cases of stricture a month, and 122 were handling 5-10 cases a month on an average. Eighteen (6.4%) urologists had a large stricture caseload of more than 20 cases a month. A number of urethroplasties were performed during the last year by these respondents; 10 had performed none, and 64 had fewer than 5 cases. The largest group of respondents, i.e., 108, performed 5 to 15 urethroplasties a year, whereas 100 respondents performed more than 15 cases in the last year (Table 1).
A newly diagnosed short segment bulbar urethral stricture would be a candidate for a visualized internal urethrotomy (VIU), according to 196 (69.5%) urologists. Moreover, 63 (22.3%) respondents were in favor of urethroplasty, and 12 responded by saying that urethroplasty would be preferred if the stricture were in the penile urethra, but VIU would still be the first option in case of bulbar urethral strictures. Of note here is that VIU remained the preferred surgery, with above 65% of respondents opting for it across all demographic subgroups irrespective of experience and stricture case burden in their practice (Fig. 1).
Recurrence after one attempt at VIU prompted 158 (80%) respondents to switch to urethroplasty as the next choice. However, 26 (13.3%) of them would want to attempt a second VIU. Those opting for an open procedure in the primary sitting itself were not as unanimous about managing their failure cases, with 28 respondents wanting to do a VIU and 22 preferring a redo urethroplasty (Fig. 1).
Failed VIU was followed by an unsuccessful urethroplasty. Fifty-two of them wanted to do VIU; a similar number of respondents, i.e., 58, preferred a redo urethroplasty, and 56 preferred referring the patient to a center of excellence for further management (Fig. 1).
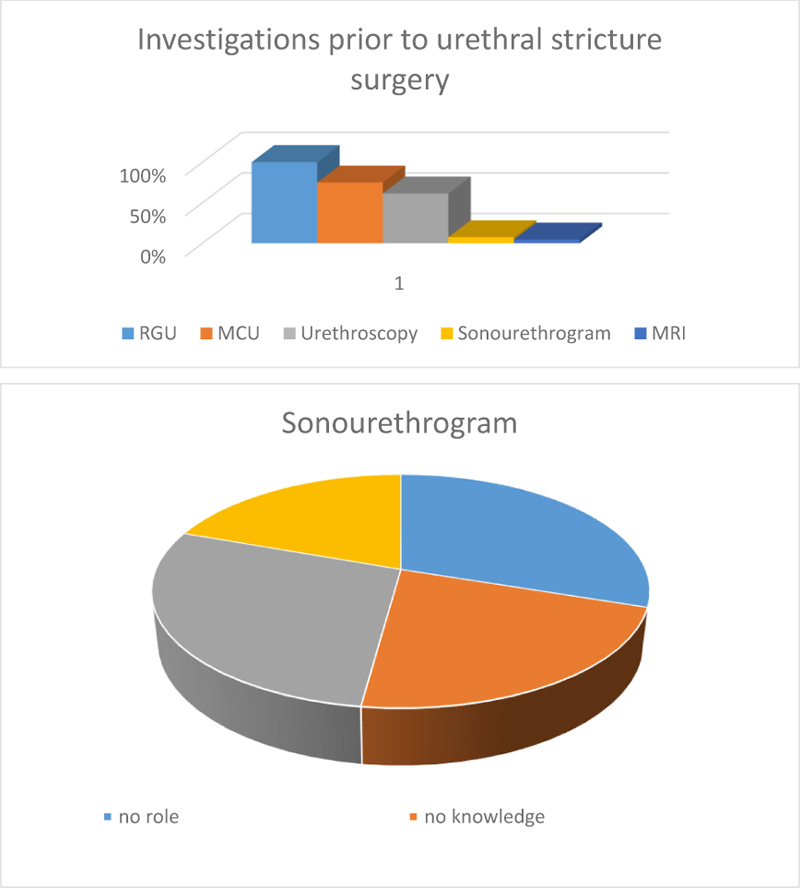
When asked about the preoperative investigations that are necessary before treating urethral strictures, all respondents (100%) wanted a retrograde urethrogram, 74% deemed micturating cystourethrogram necessary, and 60% wanted a urethroscopy. MRI was considered necessary by only 13 respondents (Fig. 2).
Twenty-one respondents wanted a sonourethrogram. However, 30% claimed that sonourethrogram had no role, and 62 said they had no knowledge regarding the use of ultrasound in urethral strictures. Furthermore, 80 respondents (28.4%) believed that sonourethrogram was useful in predicting the success of VIU (Fig. 2).
Regarding VIU, 180 (63.8%) believed that the success rate was low, yet they preferred giving it an attempt as it was less invasive. However, 60 respondents were of the opinion that VIU made future urethroplasty difficult. In contrast, 50% of the respondents preferred to have a Foley catheter in place for 1-week post-procedure, while 96 (34%) believed that 2-3 days of catheterization was sufficient. Forty-four respondents believed in maintaining the catheter for two weeks or more.
While following up with these patients, 60.99% wanted uroflowmetry as the investigation of choice. Also, a similar number of respondents (58.87%) believed that self-calibration by the patient for some time post-procedure improved the results. Regarding the duration of follow-up needed, the opinion varied widely; 88 (31.2%) believed in a 1-year follow-up, but 66 (23.4%) urologists said that a lifelong follow-up is needed.
Regarding augmentation urethroplasty, 200 respondents said it was a good procedure with a very high success rate, but regarding its feasibility, almost an equal number said that it is complex and should be left to experts (83 responses). In addition, 95 believed that it is a skill easy to master.

The practices during urethroplasties also had several disparities. Although the majority, i.e., 256 (90.8%), preferred silicone Foley catheters, the preferred size was 14F among 100 (35.5%) respondents, 16F among 153 (54%), and 18F among 29 (10.6%).
The patients were discharged with both suprapubic catheter and Foley in situ by most urologists (232 respondents, 82%), while 17.7%, i.e., 50 respondents, preferred removing the suprapubic catheter before sending them home. Time for urethral catheter removal post urethroplasty also varied widely as 38 respondents (13.5%) were in favor of removal after 2 weeks, 61% (173) thought 3 weeks was optimum, 61 (22%) wanted to wait till 4 weeks before catheter removal, and a small percentage (4%) wanted to keep the catheter in place till 6 weeks. Before removal, while the majority would do a pericatheter urethrogram in complicated cases (112), a similar number did not believe in doing peri-catheter RGU at all. Also, there were 58 responders who did a peri-catheter RGU in all cases. If a leak was found on such an RGU, deferring catheter removal was the consensus. Only 36%, i.e., 102 responders, believed in doing a second pericatheter RGU before urethral catheter removal (Table 2).
| Post Urethroplasty | Post VIU | |
|---|---|---|
| Catheter size | ||
| 14F | 100 | |
| 16F | 153 | |
| 18F | 29 | |
| Preferred catheters | ||
| SPC and per urethral catheter | 232 | |
| urethral catheter only | 50 | |
| Time of catheter removal | ||
| 2-3 days | NA | 96 |
| 1 week | NA | 142 |
| 2 weeks | 38 | 44 |
| 3 weeks | 173 | |
| 4 weeks | 61 | |
| 6 weeks | 10 | |
| Peri-catheter urethrogram | ||
| All cases | 58 | |
| Complicated cases | 112 | |
| Never | 112 | |
| Follow-up investigations | ||
| Uroflowmetry only | 136 | 172 |
| ASU | 68 | 37 |
| Urethroscopy | 52 | 65 |
| Urethral self-calibration | 24 | 166 |
| Duration of follow-up | ||
| 1 year | 56 | 88 |
| 2 years | 60 | 53 |
| 5 years | 63 | 75 |
| Lifelong | 74 | 66 |
Follow-up of urethroplasty patients was with only uroflowmetry in 136 responses (48.2%), 68 responses were in favor of including MCU and ASU along with uroflowmetry, and another 52 responses included performing urethroscopy for visual assessment of the repaired area (Table 2).
Urethral self-calibration was another area where the views differed. Only 24 respondents advised it routinely, and 95 believed that it had no role whatsoever. Moreover, 70 (25%) believed that it was beneficial in patients complaining of poor stream post urethroplasty, and 33% (93) recommended it in patients with a demonstrable narrowing on urethroscopy post-surgery.
The required follow-up time of the patients varied, with all options being almost equally preferred. Fifty-six responders advised 1-year follow-up, with 2 years by 60, 5 years by 63, and lifelong follow-ups advocated by 74.
Even when it came to defining failure after surgery, there was a difference in opinion with patient symptoms (62.5%), flow rate (49%), imaging studies (51.8%), and patient-reported outcome measures (53.19%) being important parameters among the respondents.
Concerning perineal urethrostomy as a treatment option, the majority believed that it was the best offered to patients with long strictures and multiple coexisting morbidities (181 respondents, i.e., 64%)
4. DISCUSSION
From our results, it is apparent that most urethral reconstructive surgeries are performed by a minority of practicing urologists. The purpose of our study was to determine the actual practice patterns and definitions used to define stricture recurrence by urologists across the nation (members of Urological Society of India).
4.1. Current Scenario of Urethroplasty and other Urethral Surgeries
Despite recent guidelines from prominent urology organizations around the world, such as the American Urology Association (AUA) and the European Association of Urology (EAU), randomized trials to provide irrefutable data on the appropriate management practices of urethral strictures are lacking [8-10]. As noted by Kulkarni et al. while formulating guidelines for urethral stricture management for the Urological Society of India in January 2021 [8], the present guidelines are not standardized, and those available are based mostly on limited data and personal experiences of experts. In the present survey, 90% of the respondents also believe that the treatment strategy is either not standardized or not well-followed.
4.2. Exposure to Urethral Reconstructive Procedures
Mundy stated that a minimum of 15 urethroplasties per year are needed for a center to maintain good quality [11]. In a survey of practicing members of the AUA, 93% of the responders perform <5 urethroplasties per year, whereas 58% do not perform this surgery, and 46% experts perform 20-100 urethroplasties annually [12]. According to our survey demographics, only 6.4% of urologists perform more than twenty cases per year, while 38.2% perform only 5-15 cases annually. Through these results, it is apparent that most urethral reconstructive surgeries are performed by a minority of practicing urologists in India.
4.3. Anterior Urethral Stricture Management Practices
As per literature, RGU and sonourethrography are equally efficacious in the detection of anterior urethral strictures. Further characterization of strictures in terms of length, diameter, and periurethral pathologies, like spongiofibrosis and false tracts, is done with greater sensitivity using sonourethrography as compared with RGU, with the added benefit of lower incidence of complications [13]. Preoperative RGU is often omitted, although estimation of the exact stricture length by endoscopy is unreliable [6]. However, our survey demographics showed that all urologists practicing urethral surgeries preferred to have a preoperative RGU, and 75% considered MCU a part of workup. However, their knowledge and use of sonourethrogram as a preoperative modality were limited.
Open urethroplasty has long been regarded as the gold standard treatment for anterior urethral strictures. In the hands of an experienced urologist with proper procedure selection, one can expect success rates of up to 90% to 95% at 15-year follow-up and complication rates as low as 7% to 14%. Currently, urethral dilation and VIU are the most common procedures used for primary short segment bulbar urethral strictures [14-17]. Two-thirds of the respondents believed that the results of VIU are inferior to urethroplasty; however, they still preferred it to be the first choice for treatment as it is less invasive. This is also in line with what is recommended by AUA and EUA guidelines. Only 22.3% opted for urethroplasty as the primary treatment modality in short-segment anterior urethral strictures.
4.4. Post-Procedure Catheter Practices and Follow up
The best interval period for catheterization after urethroplasty and VIU is unknown, and most published recommendations represent expert opinion only. Moreover, even the published guidelines are not clear in this regard. For anastomotic urethroplasty, suggested catheterization periods range from 7 to 21 days, but only two reports mention the rate of extravasation when this timing is followed, while for VIU, all guidelines uniformly recommend 72 hours [18]. Prolonged catheterization is consistently reported by the patients as the most troublesome part of their surgery experience [19, 20]; hence, keeping the catheter for the minimum possible period without compromising the outcome is very important. In our survey, 50% of the respondents preferred to have a Foley catheter in place for 1-week post-VIU, while 34% were of the opinion that 2-3 days of catheterization were sufficient. Post-urethroplasty, more than 60% preferred to keep the catheter for at least 3 weeks probably because they performed peri-catheter urethrogram before catheter removal and, in the presence of leak, kept the catheter for one more week. The respondents in our survey preferred self-calibration in patients with a poor stream or narrowing on follow-up, which was mostly assessed by uroflowmetry.
4.5. Defining Recurrence or Failure of Urethroplasty
The success rate of urethroplasty surgery is dependent upon how a failure is defined. There is no clear consensus on defining the recurrence or failure of the procedure [7]. Similar views were reflected in our survey, with the majority of respondents considering multiple factors before establishing the diagnosis of a failed procedure. The most common parameters used were patient symptoms (62.5%) and patient-related outcome measures (53.19%).
4.6. Management of Recurrent/ Failed Procedure
As per literature, successive urethrotomy negatively impacts the overall success of subsequent open urethroplasty. Roehrborn and McConnell found that in patients previously treated with multiple dilations, the urethroplasty failure rate doubled from 14.3% to 27.6% [21, 22]. This knowledge was also reflected in our survey as 80% of urologists preferred to perform urethroplasty on previously failed VIU cases, while 13.3% still preferred to opt for a second VIU. However, the disparity between the practices and published reports was highlighted when nearly 20% preferred to perform VIU for the third time after the second recurrence in spite of the results being poor, probably reflecting the poor urethroplasty skills among practicing urologists.
4.7. Sexual Dysfunction Post Urethroplasty
Anterior urethroplasty has a probability of causing transient erectile dysfunction (ED) in as much as 20% of patients. The type of urethroplasty, age, and stricture length have no significant effect on ED [23]. Bulbar urethroplasty poses a greater risk of injury to cavernosal nerves and the vascular supply, thus having higher chances of postoperative ED. Problems, such as impaired erection and ejaculation, are more frequent after bulbar urethroplasty, whereas penile curvature and cosmetic appearance are the primary concerns after penile urethroplasty [24-26]. Our survey showed similar results, with more than 40% of urologists reporting sexual dysfunction in their patients, with reduced rigidity (35.5%) and erectile dysfunction (33.3%) being the most common problems.
CONCLUSION
This survey on urethral stricture management amongst urologists shows that there is wide variation in the practice of preoperative evaluation, type of surgery done for stricture in various locations, post-op management, handling of recurrence of stricture, and follow-up. This survey underscores the need for large-scale, long-term studies to formulate a guideline for the management of urethral stricture disease. This guideline should bring about a uniformity in pre-op evaluation, choice of surgery, duration of post-op catheterization, definition, and management of recurrence of urethral strictures.
AUTHORS’ CONTRIBUTION
Suyog Shetty conceptualized the study and contributed to data acquisition and manuscript writing. Suresh Bhat conceptualized the study and reviewed the manuscript. Anupam Choudhary developed the design and edited the manuscript. Zeeshan Hameed contributed to data acquisition. Mammuleni Sitharam performed a literature search as well as a statistical analysis. Milap Shah wrote the manuscript. Suraj Jayadeva Reddy edited the manuscript.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The institutional ethics committee approved this study (Kasturba Medical College and Kasturba Hospital Institutional Ethics Committee; Reg no: ECR/146/inst/KA/2013/RR-16), and the study registration number is IEC - 725/2020.
HUMAN AND ANIMAL RIGHTS
No animals were used for studies that are the basis of this research. All the humans used were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013 (http://ethics.iit.edu/ecodes/node/3931).
CONSENT FOR PUBLICATION
Participation was voluntary.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We would like to acknowledge Dr. Pankaj Joshi (Kulkarni Endourology Institute) for his critical reviews during the questionnaire formulation process and constant encouragement. We also acknowledge the Urological Society of India for helping us reach out to all the urologists across the nation and abroad.


