All published articles of this journal are available on ScienceDirect.
Acute Intermittent Peritoneal Dialysis in Critically Ill COVID-19 Patients with Renal Failure: Saviour or Succourer
Abstract
Background:
The COVID-19 pandemic has led to an increase in critically ill patients with renal failure, with many requiring renal replacement therapies. Unfortunately, many of them are too critically ill to tolerate intermittent hemodialysis. In the setting of limited resources, we did bedside acute intermittent peritoneal dialysis for critically ill COVID-19 patients with hemodynamic instability with or without ventilator support admitted to our intensive care unit.
Objective:
The aim of the study was to determine the outcome of intermittent peritoneal dialysis in critically ill COVID-19 patients.
Methods:
Our retrospective observational study included 91 patients with critically ill SARS-CoV2 infection and renal failure admitted to the intensive care unit of our hospital from July 2020 to September 2021, who underwent acute intermittent peritoneal dialysis.
Results:
The demographic, laboratory, and treatment parameters were compared between survivors and non-survivors. Variables, like increased mean age (49.88 vs. 59.07 years), presence of diabetes mellitus (36.4% vs. 63.8%), increased lung involvement (57.3% vs. 75.0%), mechanical ventilation (15.2% vs. 70.7%), systolic (84.3 vs. 77.5 mm of Hg) and diastolic (59.09 vs. 42.93 mm of Hg) blood pressures, were associated with poor outcomes. The use of hypertonic PD (63.6% vs. 37.9%), better urea reduction ratios (44.33 vs. 39.84), and increased PD cycles (66.52 vs. 44.26) were associated with a better outcome. Complications, like haemorrhage and peritonitis, occurred in 10.9%.
Conclusion:
PD is a cost-effective bedside RRT that can be considered an effective option in critically ill COVID-19 patients. Good urea clearance, hemodynamic stability, and minimal resource requirements are also the features that favour peritoneal dialysis.
1. INTRODUCTION
1.1. Global Burden
A novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was identified as the putative factor for the cluster of pneumonia cases in Wuhan, China, in December 2019. This outbreak has led to a global pandemic within a short period. As of October 1, 2021, there were more than 2.35 billion confirmed SARS-CoV-2 cases with 4.8 million deaths globally, according to the World Health Organization [1]. Although the vast majority of infected individuals developed mild or no respiratory symptoms, a significant proportion of patients progressed to severe lung damage characterized by acute hypoxic respiratory failure (AHRF), needing intensive care support [2]. Lung involvement and acute respiratory failure are the characteristics of coronavirus disease 2019 infection (COVID-19), but other organs, such as the heart, kidneys, liver, and central nervous system, are also involved.
Our hospital faced a massive burden of COVID-19 cases during the first and second pandemic waves. Till October 1, 2021, we had treated nearly 32,000 COVID-19 cases with either RTPCR positive or radiological evidence of COVID-19 or both. Since we are a tertiary care referral centre for nearly ten surrounding districts in southern Tamil Nādu in India, we received more and more critically ill patients. The incidence of renal failure among our COVID-19 patients was nearly 25%, and dialysis dependency was about 5% of total admission.
1.2. COVID-19 and Kidney: A Critically ill Disease
Acute kidney injury is one of the most critical complications of COVID-19, occurring in 0.5–7% of cases and 2.9–23% of intensive care unit (ICU) patients [3-5]. In previous reports by Cheng Y et al., acute kidney injury (AKI) develops in 5% to 15% cases and carries a high (60% - 90%) mortality rate in SARS and MERS-CoV infections [5]. A study by Fanelli V et al. involving > 2000 inpatients with COVID-19 reported an AKI incidence of 27.8% [6]. Renal failure is an independent risk factor linked to increased in-hospital mortality in patients with severe sepsis regardless of etiology [7, 8]. In a multicentre study involving 3099 COVID-19 ICU patients from the United States of America, around 20% had acute kidney injury (AKI) requiring renal replacement (RRT) [9]. Similarly, in the United Kingdom, during the first pandemic wave, a quarter of ICU COVID-19 patients needed renal replacement therapy [10]. Furthermore, an ongoing requirement for renal support and prolonged hospitalization poses a significant health care resource burden.
Patients with pre-existing ESKD appear to be at high risk for COVID-19 and its complications, as most of them are aged, with comorbidities, and some of them might be on immunosuppressant medication to treat an autoimmune disease or a graft rejection [11]. Also, those patients already having CKD are at a higher rate of becoming dialysis-dependent and critically ill. Hemodialysis patients have additional risk factors, including chronic immune dysregulation, the need to go to the hospital for in-centre hemodialysis (HD), require frequent visits to health care facilities, and sharing standard rooms with other patients [12]. Therefore, once infected, dialysis patients become sources of spreading the infection within these high-risk groups, and reports suggest a critical course in patients with ESKD on hemodialysis [13]. Even though earlier small case series have suggested a milder course [14], more and more studies have reported the significantly critical nature of the disease. A study conducted in Qatar by Hamid et al. found the incidence of COVID-19 to be 7.1% in a dialysis cohort versus a 4% national cohort [15]. They also suffered from more complications and mortality, given their age and comorbidities. The study population had approximately a hundred times higher risk of mortality than Qatar's general population (15% in the dialysis cohort versus 0.15% countrywide) [15].
1.3. Initial Challenges for RRT
The globally widespread nature of the pandemic has also placed enormous demands on the need for RRT across the world. The RRT requirements among patients with COVID-19 appear to be five-fold higher than those observed in historical populations (4.9% versus 0.9%) [16]. In datasets of community cohorts reported by Chan et al., 5%–15% of patients hospitalized with COVID-19 required dialytic support [17]. In specific United States centres, the requirement of CRRT increased 370% over typical levels [18]. Thus, the demand outweighed the supply worldwide during the initial six months of the COVID-19 pandemic [19]. Moreover, this extraordinary demand for RRT has strained the delivery of all dialysis therapies across the world. As a result, manufacturers of RRT have often faced unprecedented challenges in supply.
In view of the COVID-19 pandemic by April 2020, our hospital allotted two blocks with a bed strength of 1,500, with 1400 beds having oxygen facilities. We also created 400 ICU beds in three divided areas. In addition, we had allotted ten hemodialysis machines and one CRRT machine. We also dedicated a team of four nephrology consultants, four intensivists, eight junior residents in nephrology, ten staff nurses, and eight well-trained certified technicians to our dialysis unit.
However, even with the above facilities, we could not provide extracorporeal dialysis methods to all needed patients. The factors that made hemodialysis difficult were, first, many patients were too critically ill to tolerate intermittent hemodialysis (IHD); second, there was an immediate need for RRT at the bedside; third, sometimes patients could not be shifted to hemodialysis unit; and last, there was a limited number of portable HD units. To avoid viral transmission and increased propensity to clot membranes, bloodline, and circuits in COVID-19 infection, we adapted the policy of single-use dialyzer, which needed more supply of consumables. In addition, the need for more CRRT machines and CVVH facilities was severely limited due to the lockdown. Nevertheless, so far, we have done RRT for 750 COVID-19 infected patients. But providing dialysis support to critically ill hemodynamically unstable patients was a big challenge for us.
1.4. Peritoneal Dialysis: An Option in Crisis and Critically ill Disease
Acute intermittent peritoneal dialysis (PD) can be performed at the bedside with minimal technical support and infrastructure. Even though the argument leans towards PD as an inferior procedure, more studies have shown PD to be effective in critically ill patients where continuous venovenous hemodiafiltration (CVVH) facilities are limited. The efficacy of PD is at least as greater as HD and it is possibly as efficient as hemodiafiltration [20, 21]. A meta-analysis by Liu et al. (analysis of six studies with 484 patients) compared PD therapy and extracorporeal RRT for management of AKI; PD therapy showed little or no difference from other extracorporeal therapies concerning all-cause mortality and recovery of kidney function [22]. Other clinical trials that have compared extracorporeal therapies versus PD indicate that the correction of uremia, acidosis, fluid overload, hyperkalemia, and incidence of infectious complications are not statistically different between the groups. Moreover, the cost-benefit advantage is more favourable for the PD group, especially in resource-limited settings [20]. The efficacy of correcting acidosis or Kt/V dosage may be higher in extracorporeal therapies; however, the evidence for this argument is meagre [22]. However, one limitation is that PD may be slightly less effective in removing ultrafiltration volume.
1.5. Peritoneal Dialysis in COVID-19: Myths and Facts
It is believed that the elevation of intraperitoneal pressure (IPP) generated by PD may alter diaphragmatic movement, thereby decreasing pulmonary compliance, which could hinder mechanical ventilation and worsen respiratory failure [22-24]. However, a recent prospective cohort study by Almeida et al. evaluated the respiratory mechanics of 154 patients with AKI and on PD versus HD; they showed that resistance in the respiratory system remained stable among the patients with PD and decreased among the HD patients, and oxygenation index increased in both the groups. Thus, the authors concluded an improvement in ventilatory mechanics in both the groups, without significant differences [25].
The clearance of inflammatory cytokines in PD has been well demonstrated in experimental models. The study by Altmann et al. [26] also proved interleukin 6 (IL-6) to be eliminated through PD effluent. Zhao et al. showed that inflammatory factors, including TNF- alpha, are removed by PD. The clearance of inflammatory cytokine developed during COVID-19 disease is also a possible advantage of PD. TNF-α, IL-6, and PCT are removed effectively in peritoneal dialysis [27].
To overcome the crisis during the pandemic, our hospital started an acute intermittent PD program in critically ill COVID-19 patients with renal failure, including both AKI and ESKD. Initial planning took place in the weeks after the surge and took about two months to implement the peritoneal dialysis program. From June 2020 to the end of September 2021, a total of 627 COVID-19 patients received intermittent hemodialysis, and 91 COVID-19 patients received acute intermittent peritoneal dialysis in our hospital (Fig. 1: x-axis represents months and the y-axis represents the total number of cases undergoing procedure).
Though the vaccination and COVID-prevention strategies are being implemented throughout, there is still a global concern about the third wave of COVID-19. So, the use of acute peritoneal dialysis as an option for RRT in critically ill patients with COVID-19 in resource-limited settings and with mass needs is still an area to explore.
2. MATERIALS AND METHODS
2.2. Study Population
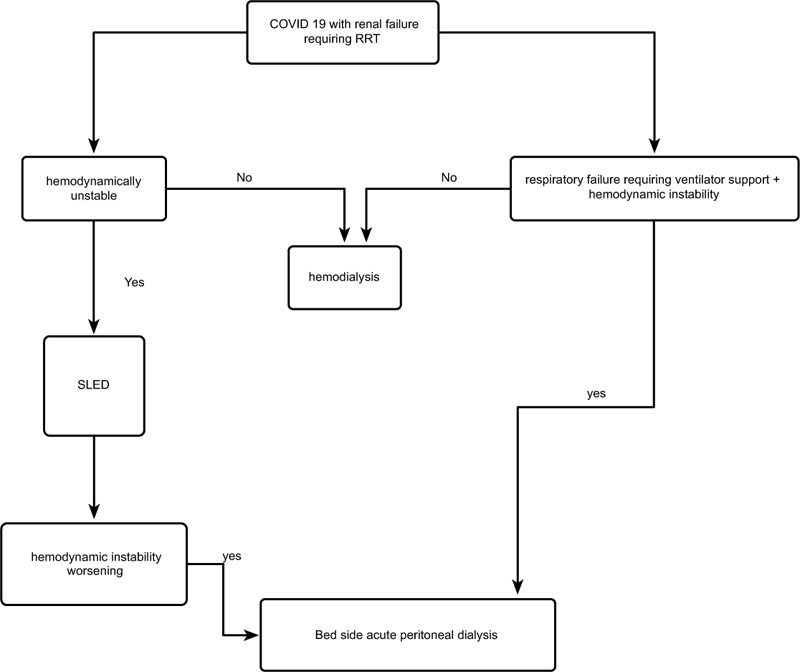
The study population included 91 patients diagnosed with hemodynamically unstable critically ill SARS-CoV-2 infection (Table 1) with renal failure, both AKI and CKD, and admitted to the intensive care unit of our hospital from July 2020 to September 2021.

| Milder disease | Individuals having fever, cough, sore throat, malaise, headache, muscle pain, nausea, vomiting, diarrhoea, loss of taste and smell |
| Moderate disease | Individuals with a lower respiratory disease during clinical assessment or imaging and having an oxygen saturation (SpO2) ≥94% at room air |
| Severe disease | Individuals with SpO2 <94% at room air, a ratio of arterial partial pressure of oxygen to fraction of inspired oxygen (PaO2/FiO2) <300 mm Hg, respiratory rate >30 /min, or lung infiltrates >50% |
| Critically ill | Individuals with respiratory failure, septic shock, or multiple organ dysfunction |
2.3. Operational Definitions
- Primary outcome: This outcome involves recovery from critical illness or death of patients.
- Secondary outcome: This involves efficacy (urea reduction ratio calculated at 24 and 48 cycles) of peritoneal dialysis and complications of peritoneal dialysis.
Sampling procedure: Individuals who had positive nasopharyngeal swab for SARS-Cov-2 by reverse transcriptase method or a CT chest suggestive of COVID-19 (CORADS 4-6) or both were considered to have COVID-19 disease. Patients who were admitted with COVID-19 disease were classified as having mild, moderate, severe, and critically ill disease (Table 1). Patients who were diagnosed to be critically ill were admitted to the intensive care unit (Fig. 2).
- Inclusion criteria: Those patients who were diagnosed with severe critically ill COVID-19 infection along with with acute kidney injury or chronic kidney disease and undergoing acute peritoneal dialysis were included in the study.
- Exclusion criteria: Those patients who had developed poor PD flow, major leaks, bleeding complications, prone ventilated patients, sudden worsening of hypoxia on a ventilator soon after initiation of PD were taken for IHD/SLED/CVVH and excluded from the study.
2.4. PD Procedure Methodology
After obtaining an informed written consent from the patient or first-degree relative (if the patient is in an altered sensorium), peritoneal dialysis was initiated. Bedside percutaneous catheterization was done as per our department protocol. After aseptic abdomen preparation and filling the peritoneal cavity with 20ml/kg of peritoneal dialysis fluid as priming (Table 2), a non-cuffed rigid plastic catheter was inserted just below the umbilicus in the midline by blind Seldinger procedure under local anaesthesia. Each acute peritoneal dialysis cycle (30 ml per kg) was prescribed at a frequency of one hour thirty minutes with an intraperitoneal dwell time of one hour and inflow time, and outflow (drain) time being 15 minutes each. The peritoneal dialysis cycles were repeated and continued until the patients were hemodynamically stable or recovered from renal failure or death. Patients with hypervolemia, CT chest suggestive of a mixed pattern of ground glass opacities, and pulmonary edema were prescribed hypertonic PD fluids (addition of 100 ml of 25% dextrose along with existing fluid and shorter dwell time). For those patients with pre-existing diabetes mellitus, eight units of short-acting insulin were added to each litre of PD fluid and subcutaneous or intravenous insulin for control of blood sugar as per patient glycaemic status and as per the protocol.
| Content | Gram per litre |
|---|---|
| Dextrose IP (as anhydrous) | 17 |
| Sodium chloride | 5.56 |
| Sodium acetate | 4.76 |
| Calcium chloride | 0.22 |
| Magnesium chloride | 0.152 |
| Sodium metabisulphite | 0.15 |
| Sodium | 130 mmol/L |
| Calcium | 1.5 mmol/L |
| Magnesium | 0.75 mmol/L |
| Bicarbonate (as acetate) | 35 mmol/L |
| Osmolality | 363mosm/l |
2.5. Treatment and Laboratory Methodology
All patients were treated with medications as per covid pandemic guidelines (Annexure 1). All patients were either on non-invasive ventilation (high flow nasal oxygen /HFNO) or invasive ventilation. The patients were managed in the ICU until hemodynamic stability was attained or weaned off the ventilator. Patients who recovered from hemodynamic instability were converted to regular hemodialysis or followed conservatively according to need. Furthermore, those patients who recovered were discharged after the isolation period.
All patients admitted in our covid block underwent routine biochemistry investigations, complete blood count, renal function test, serum electrolytes, serum ferritin, CRP, and radiological investigations immediately at the time of admission. Moreover, for patients who underwent peritoneal dialysis, RFT and electrolytes assessment was repeated at the end of 24 and 48 cycles to calculate the urea reduction ratio in order to assess the recovery and decide whether to continue or stop peritoneal dialysis.
2.6. Data Collection Procedure
From our hospital, COVID-19 database history, clinical findings, routine laboratory parameters, including a complete blood count and serum biochemistry (blood urea, serum creatinine, electrolytes, C-reactive protein, and ferritin) and radiological findings, were taken. The lung involvement in CT chest was divided into 4 grades (grade 1 = < 25% involvement, grade 2 = 25% to 49% involvement, grade 3 = 50% to 74% involvement, and grade 4 = > 74% involvement). The data related to the total number of PD cycles, complications like poor flow, haemorrhage, and peritonitis, were extracted from our department's COVID-19 databases. After 24 cycles and 48 cycles of peritoneal dialysis, the urea reduction ratio (URR 1 and URR 2) was assessed.
2.7. Ethical Issue
The study was approved by the Institutional Ethical Committee, Madurai medical college, Madurai. The confidentiality of research subject's personal information was ensured in accordance with the ethical principle of the Declaration of Helsinki.
2.8. Data Entry and Analysis
SPSS 22.0 version and R-programming were used to carry out the statistical analysis. Demographic and comorbidity statuses of the patients were compared among the patients who recovered and the patients who expired using the chi-square test/Fisher's exact test. Shapiro-Wilk normality tests were used to find out the normality of the continuous data.
Box transformation was used to transform the non-normally distributed data into normally distributed data. Independent sample t-test was used to compare the biochemical parameters (continuous data) between the two group patients. The multivariate statistical model was used to find out the key factors associated with mortality. A five percent level of significance was considered statistically significant.
3. RESULTS
3.1. Demographic and Clinical Characteristics
The demographic and clinical characteristics of the cohort are shown in Table 3a. 91 patients were included in the study. Of the 91 patients in our study population of renal failure with critically ill COVID-19, 60 (65.9%) patients were ESKD on regular hemodialysis before admission, 7 (7.7%) patients were known CKD on conservative management prior to COVID-19 infection, and 24 (26.3%) patients were newly diagnosed AKI by appropriate investigations. Males constituted 63.7% of our cohort, but neither sex was significantly associated with mortality. The average age in the survivor group was around 49.88 years, while in non-survivors, it was 59.07 years. Patients belonging to the age group more than 75 years and 50 to 74 years were found to have significantly higher mortality (p-value = 0.04). The common comorbidities were diabetes mellitus (53.8%), coronary artery disease (29.6%), and pre-existing pulmonary disease (16.4%). When considering the comorbidity status of the patients' coronary artery disease, Type 2 diabetes mellitus and pre-existing lung disease were significantly associated with mortality (P-value <0.05). The most common symptoms at initial presentation were dyspnoea (98.9%), fever (91.2%), cough (86.8%), and diarrhoea (29.6%).
| Variable | Survivors | Non-survivors | P-value |
|---|---|---|---|
| Hypertonic PD | 21 (63.6) | 22 (37.9) | 0.018 |
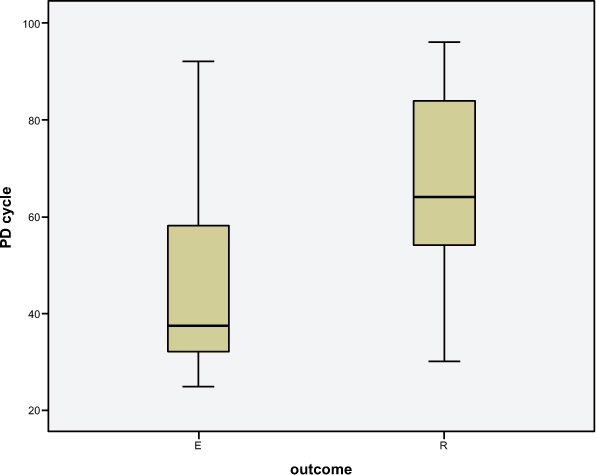
| Number of PD cycle | 66.52±17.7 | 44.26±15.53 | <0.001 |
| Hemorrhage | 1(3%) | 5(8.6%) | 0.716 |
| Peritonitis | 2 (6.1%) | 4 (6.8%) | 0.716 |
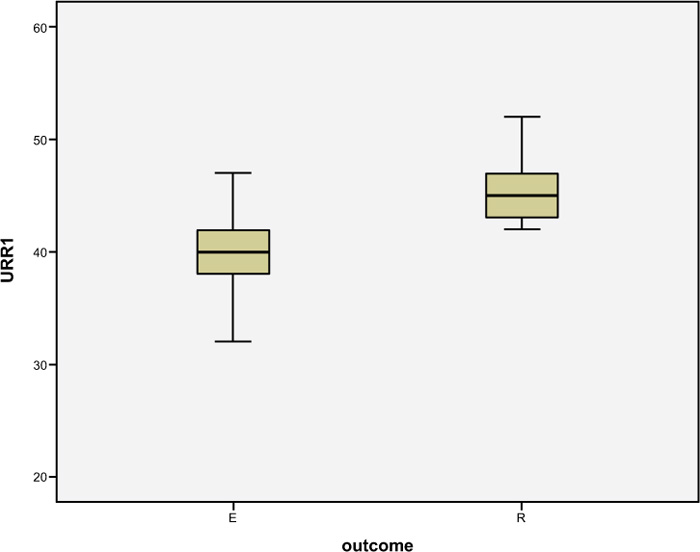
| Urea reduction ratio URR1 | 44.33±5.52 | 39.84±4.633 | <0.001 |
| Urea reduction ratio URR 2 | 59.55±6.15 | 53.62±5.17 | 0.004 |
| Variable | Survivor | Non-survivor | P-value |
|---|---|---|---|
| Hemoglobin (g/dl) | 8.82±1.18 | 7.86±1.36 | 0.023 |
| Total count | 9927.27±5145.28 | 13115.52 | 0.017 |
| Platelet (lakhs /mm3) | 2.07±0.59 | 1.89±0.55 | 0.150 |
| Neutrophil lymphocyte ratio | 7.63±0.88 | 8.93±0.76 | <0.001 |
| Urea (mg/dl) | 173.85±41.19 | 217.76±38.49 | <0.001 |
| Creatinine (mg/dl) | 10.40±2.63 | 14.08±3.35 | <0.001 |
| RBS (mg/dl) | 181.97±1.35 | 213.79±1.44 | 0.036 |
| Urine Acetone (%) | 3(9%) | 4(6.8%) | 0.701 |
| Sodium (meq/L) | 133.82±4.76 | 129.57±6.25 | 0.001 |
| Potassium (meq/L) | 4.32±0.69 | 4.63±0.64 | 0.032 |
| Bilirubin (mg/dl) | 0.93±1.77 | 1.51±1.62 | 0.001 |
| SGOT (U/L) | 28.84±2.04 | 42.66±1.82 | 0.007 |
| SGPT | 33.88±2.09 | 51.29±1.86 | 0.004 |
| C reactive protein (mg/L) | 75.66±15.78 | 92.24±14.01 | 0.001 |
| Ferritin (ng/ml) | 650.67±117.48 | 833.33±104.18 | 0.001 |
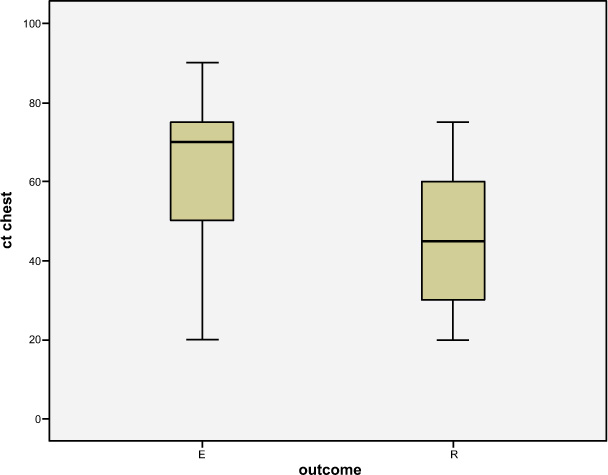
| Mean percentage of lung involvement in CT chest | 57.31 | 75.01 | 0.001 |
| CT chest grade 1 | 3 | 3 | 0.003 |
| CT chest grade 2 | 18 | 14 | |
| CT chest grade 3 | 12 | 32 | |
| Ct chest grade 4 | 0 | 9 |
| Predictors | P-value | Odds ratio | 95% confidence interval for odds ratio | |
|---|---|---|---|---|
| Lower | Upper | |||
| CT chest | 0.002 | 0.898 | 0.838 | 0.936 |
| PD cycle | 0.002 | 1.146 | 1.051 | 1.250 |
| Urea reduction ratio 1 (URR1) | 0.003 | 1.385 | 1.121 | 1.712 |
| Neutrophil lymphocyte ratio | 0.002 | 0.120 | 0.031 | 0.458 |
3.2. Disease Specific Parameters
Among the patients, 26.3% (n=24) were diagnosed with AKI and 73.7% as CKD. Patient admission urea (173.85 mg/dl vs. 217.76 mg/dl) and creatinine (10.4 vs. 14.08) values exhibited a significant correlation with mortality (value < 0.05) . Hypertonic PD was done in 47.2% (n=43) of patients, and it was significantly associated with decreased mortality (Table 3b). The urea reduction ratio was calculated after 24 cycles (URR1) and 48 cycles (URR 2) of PD. Both URR1 and URR 2 were significantly higher in the survivor group (Fig. 3). The mean number of PD cycles was significantly higher in the survivor group than in the non-survivor group (66.52 vs. 44.26, p-value < 0.05). Complications, like haemorrhage and peritonitis, were found in 13.1% (n=12) of patients. However, none of the complications were significantly associated with mortality. The mean dialysis vintage in the non-survivor group was 7.38 months and was significantly higher than in the survivor group (2.57 months). Mean blood pressure at the time of presentation in survivor vs. non-survivors was systolic (84.3 mm of Hg vs. 77.5 mm of Hg) and diastolic BP (59.09 mm of Hg vs. 42.93 mm of Hg), and it was found to be statistically significant with the outcome (p-value < 0.05). In the non-survivor group, around 83% of patients were in double ionotropic support and 17% in single ionotropic support. Patients with double ionotropic supports were found to have significantly increased mortality (p-value < 0.05). Mechanical ventilation was used in 50.5% (n= 46) of patients, and its use was significantly associated with mortality. The mean duration of ICU stay (duration from entry to ICU to an outcome) among survivors was 127.28±88.69 hours (6-9days on average), whereas, in non-survivors, it was 65.32±30.79 hours (3-4 days on average) and found to be statistically significant.
3.3. Laboratory Parameters
Patients in the non-survivor group had significantly lower hemoglobin when compared to the survivor group (p-value=0.02). Biochemical parameters (Table 3c), including total leukocyte count, neutrophil-lymphocyte ratio, blood urea and creatinine at presentation, were significantly increased in the non-survivors group. Patients who expired had significant hyperglycemia (p-value <0.05). Despite higher blood glucose levels in the non-survivor group, only four patients in the expired group had diabetic ketoacidosis (DKA) with urine acetone positive (p-value =0.701). The lung involvement in the CT chest was divided into four grades. Patients having grade 3 and grade 4 were found to have significantly higher mortality. 47.2% (n=43) patients were detected to have features of both COVID-19 pneumonia and pulmonary edema in the CT chest, but the combination was not found to be a significant risk factor for mortality. Significant elevation of serum bilirubin, SGOT, and SGPT was observed in the non-survivors group. Serum sodium was found to be significantly lower in the non-survivor group (p-value < 0.05). Inflammatory markers, CRP, and ferritin were significantly increased in the non-survivors group (p-value< 0.01). The mortality in our cohort was 63.7% (n=58) (Table 3d).
3.4. Predictors of Mortality
Multivariate analysis was conducted to find out the crucial predictors of mortality. The higher percentage of lung involvement in CT chest (Fig. 3a) and increased neutrophil-lymphocyte ratio were found to be independently associated with increased mortality, whereas a higher number of PD cycles (Fig. 3b) and higher urea reduction ratio at 24 cycles (URR1) (Fig. 3c) were found to be independently associated with decreased mortality (Fig. 3d).

4. DISCUSSION
Males were predominant in our study with 63.7%, and mortality was around 67.2% compared to females. Though in the previous studies, sex has been considered one of the risk factors for developing critically ill COVID-19 disease [28], gender variation was not statistically significant in our study. The mean age in the survivors was around 49.88, whereas the average mean age in non-survivors was around 59.07. In previous studies, individuals aged more than 75 have been found to be more severely affected [29]. In our study, common clinical presentations were fever in 91%, cough 86.8%, breathlessness in 98%, and diarrhea in 29%, similar to the previous studies [30]. The common comorbidities were diabetes mellitus (53.8%), coronary artery disease (29.6%), and pre-existing pulmonary disease (16.4%), which correlated with previous studies showing a higher incidence of critically ill COVID-19 disease in patients with comorbid conditions [31, 32]. Furthermore, when considering the comorbidity status of the patients, there was a significant difference observed in the proportion of patients who had diabetes mellitus, coronary artery disease (p<0.05 and pre-existing lung disease (p<0.05) among the survivor group and non-survivor groups.
In our study, 68% had CKD, with a mean dialysis vintage of 2.57 months in survivors and 7.38 months in non-survivors, respectively. Our study showed those with longer dialysis duration to be associated with significant mortality. The probable reason may be that chronic dialysis patients are often malnourished. Moreover, defective innate and adaptive immunity can also affect the immune system's function [33, 34]. In a study published by Goicoechea et al., longer dialysis vintage was associated with increased mortality [35]. In our study, 32% of patients had AKI, and among AKI patients, sepsis was the causal factor in 24%, diarrhea in 22%, and in 54%, AKI was probably due to COVID-19-induced MODS. The markers of inflammation, like total leukocyte count, neutrophil-lymphocyte ratio, and serum ferritin, were higher in the non-survivor group, indicating the severity of the underlying disease. On analysis, all three exhibited a significant correlation with mortality. Even though serum ferritin is usually high in any CKD, the mean level and statistically significant impact on mortality are also equally observed in AKI subsets. However, serum ferritin as a marker of inflammation in COVID-19 in the backdrop of CKD is of limited value. Nevertheless, the neutrophil-lymphocyte ratio with p-value <0.001 concords with the severity of disease-causing mortality, which is in accordance with the previous study done by Madani et al. [36]. Our study involved a significant proportion of diabetic patients, and the presence of DKA was 9.1% in survivors and 7.9% in non-survivors. The study by Steenblock et al. suggested direct pancreas involvement in COVID-19 and associated insulin resistance, and that steroid intake can precipitate DKA [37]. Though patients received steroids and peritoneal fluid with dextrose, the presence of DKA had no impact on the outcome of our study, probably due to strict glycemic control.
Upon CT scan of the chest, the patients presented either an isolated COVID-19 pattern or one mixed with pulmonary edema. However, the statistical difference was not significant with the outcome. A higher degree of lung involvement exhibited a very high significance with mortality in our study, which correlates with the study by Lei et al., where diffuse lesions and higher CT scores were associated with higher mortality [38]. In our study, patients on mechanical ventilation demonstrated a poor outcome. The percentage of non-survivors on the ventilator was 70.7%, similar to previously published reports [39, 40]. The study by Arentz et al. indicated 50–67% of COVID-19 patients admitted to the ICU and 71–75% of those having invasive mechanical ventilation to have died [41]. Auld et al. reported a high rate of mortality in 88.1% among those who were mechanically ventilated [42].
The mean number of PD cycles was significantly higher in the survivor group than the non-survivor group (66.52 vs. 44.26, p-value < 0.05), probably reflecting adequate dialysis received in the survivor group. Since ultrafiltration and solute removal are slow and unpredictable, PD is not recommended for emergency indications [43-45]. However, in our study, the use of hypertonic PD in patients with pulmonary edema had a significant impact on mortality. Hypertonic PD was done in 47.2% (n=43) of patients with pulmonary edema, and it was significantly associated with decreased mortality. One of the key findings in our study was the significant URR both at 24 and 48 cycles. Though there was observed a significant reduction, the reduction was not up to the target levels in the initial hours, probably due to shock, resulting in decreased splanchnic blood flow, similar to the observation of Nitish Garg et al. [46]. Even though the calculation of KT/V was not possible in our study, the urea reduction ratio was statistically significant. Peritonitis occurring in PD catheter infection was reported up to a frequency as high as 40%. However, with better catheter-implantation techniques and universal precautions, the incidence of peritonitis was reduced, and the risk of infection in PD was similar to the risk with other forms of extracorporeal blood purification for AKI [47-49]. Our study reported peritonitis and complications as minimal, probably due to the short period of treatment, additional use of Y set for infusion of PD fluid to avoid too much handling, and strict aseptic catheterization.
The mortality in our cohort was 63.7% (n=58), which is similar to the previous studies on critically ill COVID-19 patients in ICU settings. A study conducted by Nitish Garg et al. showed the number of survivors and non-survivors in the study sample as 21 (28%) and 54 (72%), respectively. The lower number of survivors could be due to the comorbidities and involvement of multiple organs. The study conducted by George et al. and Ponce et al. demonstrated a similar mortality rate [20, 50]. The study conducted at Bellevue Hospital during the COVID-19 surge observed a mortality rate of 63% for patients, suggesting PD's ability to deliver adequate therapy [51]. The mortality rate observed in our study was also similar to this study.
In the study conducted by Pisharody et al. comparing CVVH with PD in AKI in critical in-patients, urea and creatinine clearances and acidosis correction were better in the CVVH group, but no significant differences were observed in the correction of hyperkalaemia, altered sensorium, or hemodynamic disturbance. The mortality rate was observed to be 84% in the CVVH group and 72% in the PD group. Factors that determined the outcome were the APACHE II score (p=0.02) and ventilatory support (p < 0.01). The favourable factor in the PD group was cost-effectiveness [20]. Our study found increased URR, use of hypertonic PD, and higher PD cycles to decrease the mortality rate compared to the previous data.
The limitations of our study are that adequacy of PD cannot be wholly assessed without KT/V and the volume of fluid removed cannot be calculated without measuring the effluent. The measurement of inflammatory markers to identify the significance of peritoneal dialysis in the removal of inflammatory mediators and complete assessment of ventilator kinetics may add more validity to the study. But due to the massive burden of the pandemic and retrospective nature of the study, we were not able to do the same.
CONCLUSION
In our study on peritoneal dialysis in critically ill COVID-19 infection patients with renal failure, older age group, presence of comorbid conditions (diabetes mellitus, CAD, lung diseases), low systolic and diastolic BP at presentation, elevated inflammatory markers like NLR and CRP, admission serum creatinine and blood urea, higher hemodialysis vintage before COVID-19 infection, higher CT chest scores, and use of mechanical ventilation were significantly associated with higher mortality. On the other hand, younger age group, higher duration of ICU stay, longer duration and increased number of PD cycles, usage of hypertonic PD, and a higher urea reduction ratio were significantly associated with better survival. The good urea clearance achieved, hemodynamic stability, cost-effectiveness, bedside initiation, minimal staffing, and resource requirements are features that favour peritoneal dialysis. Hence, we suggest to consider PD as an option for critically ill patients with renal failure in resource-limited settings, where resources for CRRT are in short supply.
LIST OF ABBREVIATIONS
| CVVH | = Continuous venovenous hemofiltration |
| CRRT | = Continuous renal replacement therapy |
| ESKD | = End-stage kidney disease |
| HD | = Hemodialysis |
| ICU | = Intensive care unit |
| PD | = Peritoneal dialysis |
| RRT | = Renal replacement therapy |
| SARS-CoV 2 | = Severe acute respiratory syndrome coronavirus 2 |
| SLED | = Sustained low-efficiency dialysis |
| URR | = Urea reduction ratio |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Institutional Ethical Committee, Madurai medical college, Madurai (CDSCO: Reg.No.ECR/1365/Inst/TN/2020; DHR Reg.No.EC/NEW/ INST/2020/484).
HUMAN AND ANIMAL RIGHTS
No animals were used for studies that are the basis of this research. All the human procedures used were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013 (http://ethics.iit.edu/ecodes/node/3931).
CONSENT FOR PUBLICATION
All participants signed an informed consent form.
STANDARDS OF REPORTING
STROBE guidelines and methodologies were followed.
AVAILABILITY OF DATA AND MATERIALS
The datasets generated and/or analysed during the current study will be made available by the corresponding author [J.A.] on reasonable request.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publisher’s website along with the published article.


