All published articles of this journal are available on ScienceDirect.
CLABSI in Hemodialysis– New Face to an Old Foe; A Look at Current Trends and a Review of Literature
Abstract
Background:
Bloodstream infections are a potentially devastating complication of hemodialysis. This study aimed to elucidate the rates of CLABSI (Central Line Associated Blood Stream Infections)in a south Indian dialysis cohort and determine the catheter care and hygiene practices in the dialysis unit which might contribute to CLABSI
Methods:
A prospective observational study involving in-center dialysis patients with temporary access was conducted from June 2019 to December 2019. The catheter insertion and line handling techniques were monitored using a proforma adapted from the CDC (Center for Disease Control) guidelines. Adherence to infection control practices, rates of CLABSI identified using CDC surveillance definitions, and organisms identified were reported.
Results and Discussion:
The median rate of CLABSI in this dialysis unit was 1.68 per 1000 catheter days. Gram-positive organisms were the majority – 69.2% and 72.27% were drug-resistant pathogens. The adherence to infection control practices in hand sterility, use of barriers, and sterile insertion technique were 100% with respect to existing CDC guidelines. The use of sterile semi-transparent dressings, however, was 98.2%. 65% of all patients had soiled dressings on arrival to the hospital. Line handling protocols were followed in accordance with CDC guidelines in 97.1% of cases
Conclusion:
CLABSI rates in this institute are in keeping with those around the world. There is a predominance of gram-positive organisms causing CLABSI. Drug-resistant organisms represent an alarming 72.27%of all infections. Soiled dressing and home care of the dialysis catheters represent a neglected aspect of catheter care and should be addressed.
1. INTRODUCTION
Infection is the second most common cause of death among patients with ESRD (End Stage Renal Disease) [1]. CLABSI (Central Line Associated Blood Stream Infections) contributes to significant hospitalisation, morbidity, and mortality among dialysis patients. The cost imposed on the health care system attributed to CLABSI is enormous. The importance of identifying CLABSI rates and preventing CLABSI cannot be overemphasized. The first step to solving the problem of CLABSI is defining the magnitude of the problem. While in the United States, CDC (Center for Disease Control) monitors CLABSI rates through the NHSN (National Healthcare Safety Network), there is no central body to identify CLABSI rates in India. Hence, the paucity of data on CLABSI rates from different centers. The need for the current study arose to address this lack of information and provide an understanding of infection control practices within a tertiary-level dialysis care facility in India.
1.1. Aims and Objectives
The study aimed to identify the CLABSI rate, assess infection control practices, and identify organisms causing CLABSI within a tertiary-level dialysis facility in South India. The primary objective was to identify the CLABSI rate among dialysis patients attending the dialysis care facility over a period of 7 months. The other primary objectives of the study were to audit adherence to existing guidelines for insertion of central lines and line handling as set by the CDC. The study's secondary objectives were to identify the common pathogens associated with CLABSI in this dialysis unit and describe the clinical features and outcomes of patients with infection.
2. MATERIALS AND METHODS
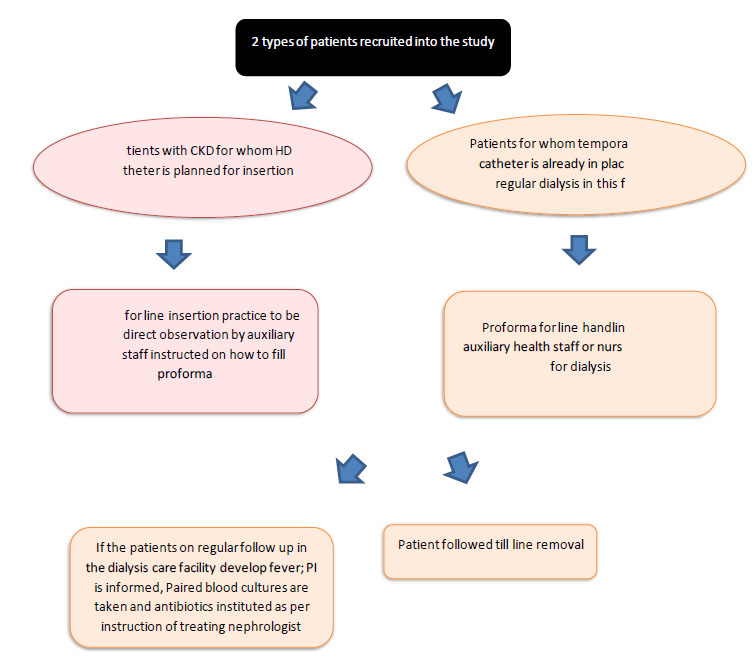
A prospective observational study was done in the period of June 2019 – December 2019. We included all consenting patients who attended the dialysis facility with a non-tunneled HD Catheter or were being planned to insert a non-tunnelled HD (Hemodialysis) catheter. All patients with tunneled catheters, AVF (Arteriovenous Fistula) and AVG were excluded from the study. Once patients were identified, line insertion practices and line handling practices were evaluated using a proforma adapted from the CDC to suit this study. Study personnel was trained in filling the proforma before study initiation. At each line insertion, the study personnel audited the line insertion using the proforma, and at each visit to the dialysis care facility, a second proforma was filled regarding line handling. In the case of suspicion of infection, the dialysis physician was informed, paired blood cultures were taken; antibiotics were instituted as per the treating physician's decision. Patients with infection were followed up until discharge. The study protocol is shown in Fig. (1).
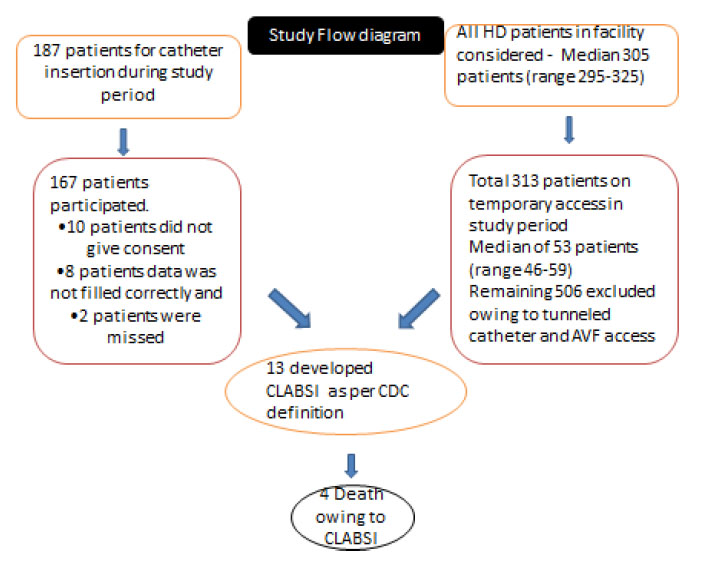
A total of 187 patients underwent temporary catheter insertion during the study period. Of these, 167 were recruited into the study for line insertion practice audit. A total of 313 patients were on temporary access using the dialysis care facility (Fig. 2). All fistulas, grafts, and tunnelled catheters were excluded. These 313 patients had 343 catheters between them since some had more than one catheter inserted – replaced for poor flow, infection or femoral change to IJV.
Definition- CDC surveillance definition of CLABSI
- CDC Laboratory diagnosis of CLABSI
- Recognised pathogens from 1 or more blood cultures OR
- The patient has at least one of the following – fever, chills, or hypotension associated with positive blood culture with common skin contaminants (like CONS, diphtheroid and Propionibacterium species) from 2 separate blood cultures
- AND no other source of infection
- Clinical definition CLABSI
- The patient has at least one of the following – fever or hypotension, and no organism can be cultured and the physician treated for sepsis
- AND no other source of infection
Abbreviations used in Fig. (1): CLIP (Central Line Insertion Proforma), AHS (Ancillary Health Service Providers), PI (principle Investigator), HD (Hemodialysis).
Blood cultures were collected from central and peripheral access and inserted in BACTEC containers. They were then transported to the central laboratory within 2 hours of collection. The proformas used are attached in Annexes 1-4. Results were analysed using SPSS 16 software.
3. RESULTS
In this dialysis care facility, a mean of 309.71 patients was dialyzed every month (Table 1). 16.74% of patients were on temporary access, representing an average of 51.85 patients per month. 96.5% of the lines were sited in the internal jugular; the remaining were in the femoral vein.
| Characteristic | Value (2 Standard Deviations from the Mean) |
|---|---|
| Mean number of patients per month Hemodialysis | 309.71 (2SD 16.02) |
| Mean number of patients per month on temporary access | 51.85 (2SD 6.20) |
| Mean number of catheter days for all patients | 1135 (2SD 230.84) |
| Mean number of catheter days per patient | 22.41 days (2SD 6.60) |
| Mean time between insertion of a catheter to culture positivity | 32.54 days (2SD 27.48) |
|
Site of access for patients on maintenance HD Internal Jugular vein Femoral vein |
331 (96.50%) 12 (3.49%) |
| Mean number of temporary catheters inserted each month | 26.71 (2SD 7.52) |
| Co-morbidities of the dialysis cohor | N=313 |
| Diabetes | 113 (36.10%) |
| Hypertension | 298 (95.20%) |
| Cardiac dysfunction | 250 (79.87%) |
| Hypothyroidism | 15 (4.79%) |
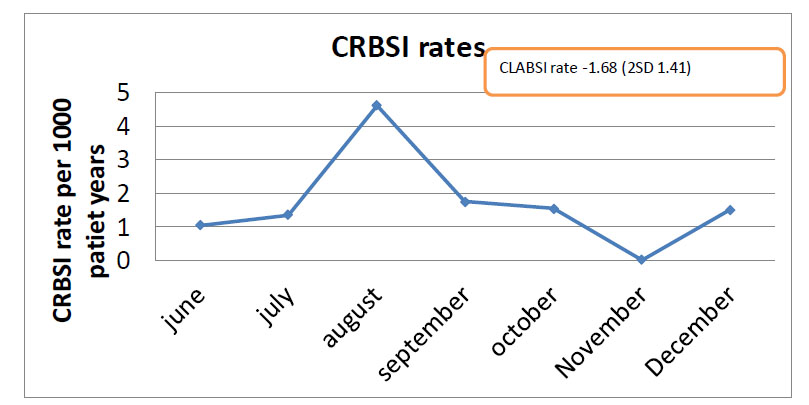
The median CLABSI rate was 1.68 per 1000 catheter days (Fig. 3).
A total of 313 patients were recruited into the study to evaluate compliance with sterile techniques. These 313 patients had 343 temporary accesses between them since the femoral lines were re-sited as internal jugular catheters, and any blocked catheters were replaced. During each session of hemodialysis, proformas regarding line handing were filled, generating 728 proformas overall. Compliance regarding the use of alcohol rub, the connection of sterile devices and the use of sterile barriers and compliance with infection control practice were assessed in 97.1% of hospital visits. With respect to dressing changes, however, sterile precautions were followed in only 69.91% of patients.
Among those whose dressings were changed, 98.61% had dressings changed in a sterile manner. In 66% of patients’ dressings were soiled on arrival. A total of 187 catheters were inserted during the study period, of which 167 patients were inducted into the study. A median of 24 catheters was inserted each month. Infection control protocols were followed 100% of the time regarding hand hygiene, use of barriers during insertion, skin preparation, and sterile handling of the line. Regarding sterile dressings, compliance with infection control practices was seen in 98.2% of patients.
During the study period, 69.23% of infections were found to be due to gram-positive organisms, and 15.38% were attributed to gram-negative organisms. 72.27% of infections were found to be resistant to first-line antibiotics (Fig. 4). The remaining patients fit the clinical diagnosis criteria, but cultures had remained sterile.
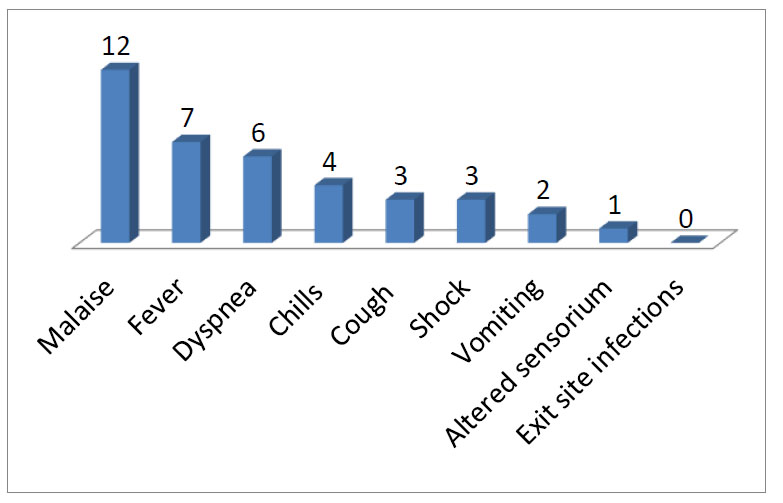
The most common presenting symptom was malaise (Fig. 5).
All patients were hospitalised, with a majority of them, 54% of them, requiring intensive care (Table 2). Death attributed to infection was 31%. 54.1% of patients recovered, and the outcome was unknown for 15% of patients since they had left treatment against medical advice. Two patients developed complications such as septic arthritis and infective endocarditis. The mean duration of hospitalisation was 8.61 days.
| Characteristic | Value |
|---|---|
|
Patients hospitalised Ward admission Intensive care |
13 (100%) 6 (46.15%) 7 (53.84%) |
| Mean duration of Hospitalisation | 8.61 days (2SD 8.12) |
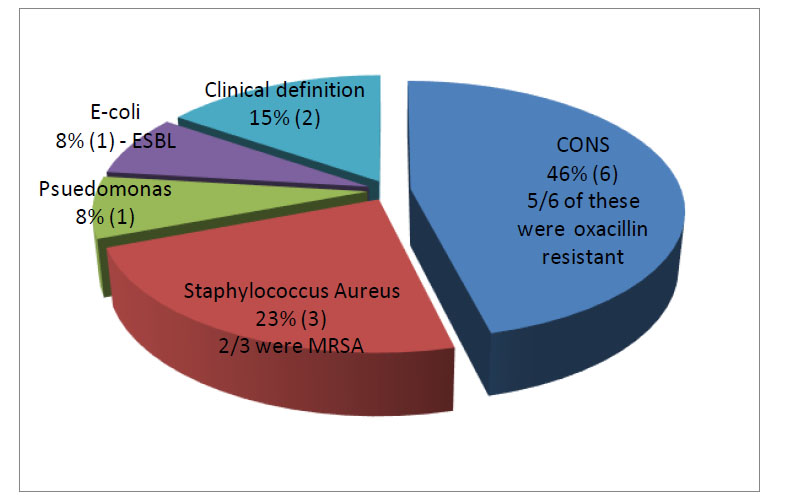
Abbreviations used in Fig. (4) : MRSA (Methicillin Resistant Staphylococcus Aureus), CONS (Coagulase Negative Staphylococcus), ESBL (Extended Spectrum Beta-lactamase). MRSA was seen in 2 of 3 staphylococcus aureus infections. The numbers in brackets refer to the actual number of patients. All infections were seen in patients with IJV catheters.
4. DISCUSSION
The issue of CLABSI among the dialysis population is important, leading to significant cost, morbidity and mortality. The incidence of CLABSI differs based on the definition used. The definition of CLABSI used in this study was the CDC CLABSI surveillance definition. Both the IDSA(Infectious Disease Society of America)and KDOQI (Kidney Dialysis Outcomes Quality Initiative) have differing definitions of CLABSI [2]. The KDOQI describes infections as definite, probable, and possible. It requires the catheter tip to be sent for culture for definitive diagnosis. Dialysis catheters are not often removed immediately for suspected CLABSI; hence this definition was not used as it might lead to underreporting of missing out on individuals with CLABSI who retain their catheter. The IDSA definition of CLABSI is more cumbersome with the requirement of timed culture specimens and quantitative and semiquantitative differences between the samples. It was found to be rather labour-intensive and impractical. The CDC definition for CLABSI surveillance has been used widely worldwide and involves paired blood cultures with clinical features of infection in the absence of other sources of infection. This definition was chosen for use in the current study for its ease of use and high likelihood of detecting CLABSI
The CDC, through the NHSN, has been monitoring the CLABSI rates in dialysis units in the US (United States). Rates published according to NHSN data show the CLABSI rate in the US to be 1 per 1000 catheter days for central venous catheter use [3]. Literature quotes CLABSI rates to be about 2.5-6 per 1000 catheter days, with wide-ranging inter-center variability [4]. Hemodialysis catheter infection rates in Saudi Arabia were quoted to occur in 23.6% of all inserted catheters [5]. In France, rates were 1.09 per 1000 catheter days [6]. In India, the CLABSI rate from another large center in South India had previously quoted CLABSI rates to be 7.34 per 1000 catheter days – a much higher incidence of CLABSI than that quoted from literature from other studies [7]. Current CLABSI rates in this study show a median of 1.68 per 1000 catheter days over a period of 7 months. This is in keeping with data from developed countries and contradicts previous higher rates of CLABSI thought to occur in developing countries.
The use of temporary access was associated with an increased rate of CLABSI in this study. The rate of bloodstream infections per 100 patients was 0.64 for AVF, 0.39 for AV graft, and 2.16 for the central venous catheter. The use of temporary access was associated with 9.87 times increased risk of CLABSI compared to other access [8]. In patients from the US, catheters were used as dialysis access in 29% of all maintenance dialysis patients. In this study, 16.4% of patients on maintenance dialysis were on a temporary catheter. In Brazil, 18.2% of all patients were on temporary catheters [9]. In Europe, CVC (Central Venous Catheter) was prevalent in 28-32% of all dialysis patients [10]. The overall rate of catheter use was lower in this center than in data reported abroad.
The duration of a temporary catheter has been considered an independent risk factor for the development of CLABSI in previous studies. Other studies have demonstrated that biofilm formation can occur within 3 days of catheter placement [11]. The cumulative hazard for the development of CLABSI increases from 8.3% at week 1 to 49.2% at week 12. The mean duration of temporary catheter use in this center was 22.41 days. In those with infections, the duration of catheter use was 32.54 days. Hence, the duration of line use was longer in those with infection.
Previous studies found no significant differences between CLABSI in the femoral and IJV (Internal Jugular Vein) sites with short-term dialysis. However, with a longer duration, femoral catheters had a higher risk of bacteraemia [12]. The NHSN data showed that CLABSI incidence was independent of the site of the catheter. In this study, all CLABSIs were noted in individuals with IJV. This probably represents the existing policy of early catheter change from femoral central access to IJV access in this center rather than an absence of infection among those with femoral catheters.
The NHSN data show that gram-positive organisms caused CLABSI in 56% of cases and gram-negatives in 12.5%. Indigenous data from other tertiary institutions reflect a predominance of gram-negative organisms causing CRBSI (Catheter Related Blood Stream Infection). In data from SGPGI (Sanjay Gandhi Post Graduate Institute of Medical Sciences), a large government-run tertiary level institute in India, CRBSI was caused by Pseudomonas aeruginosa in 47.4% of patients [13]. Data from CMC (Christian Medical College) Vellore, another large tertiary level hospital in India, also quote a predominance of gram-negative organisms (54.7% of isolates). In this study, a predominance of gram-positive infections was found, with rates being 69% gram-positive and 16% gram-negative infections; this is in keeping with data from abroad and probably more closely reflects a local bacterial epidemiological profile. This data is important since it determines empiric antibiotic therapy.
What is of even more concern is the alarming rate of multidrug-resistant organisms that were isolated. Of the 11 pathogens identified, 8 were drug-resistant. Almost all Staphylococcus aureus identified were oxacillin resistant. In series from NHSN, 39% of all Staphylococcus aureus were oxacillin resistant. In other data from India, MRSA (Methicillin Resistant Staphylococcus Aureus) was noted in 14.3% of isolates in a large series of non-tunneled HD catheters. The increased rate of MRSA may reflect the local antibiotic usage protocols. Although the study was done in India and may not be generalisable to the rest of the world community, it highlights the importance of knowing the local microbiological pattern in the dialysis community and data can be used to compare the rates of drug resistance across countries.
Few studies have evaluated various steps to maintain asepsis in a dialysis unit, from line insertion to line handling. Data on hand hygiene is available from a cohort of dialysis patients in Spain, where hand hygiene was observed in only 15.2% of cases [14]. Data from Indian dialysis centers also reflect abysmal rates of hand hygiene, showing adherence to hand hygiene in 52 and 66% of patients before and after patient contact, respectively. In this study, hand hygiene protocols were followed in 97.1% of cases regarding line handling and followed 100% during line insertion. These rates are better than those described in other studies and possibly contribute to the low rate of CLABSI. As shown in previous studies, adherence to hand hygiene depends on the institution and patient-to-staff ratio. Other practices, such as catheter lock solutions and antimicrobial ointment, are not currently part of the infection control protocol in this institution. Staff and patient education are mandatory at the initiation of training and dialysis. A recent study on citrate locks compared with heparin locks was carried out in this institution. The results showed citrate locks to be comparable to heparin locks. The rates of CLABSI in that study were in the range of 2.71-2.89 [15]. Of note in this study, 66% of patients arrived at the hospital with soiled dressings representing a neglected aspect of home care of the dialysis catheter
The mean duration of hospitalisation in this study was found to be 8.61 days. In data from abroad, the mean duration of hospitalisation varied from 9 -13 days from CDC collaborative data. This adds to the financial burden of CLABSI incurred by the patient. Of note, all patients in this cohort were hospitalised, and 54% required intensive care. The mortality associated with CLABSI was also higher than expected- 31%. Data from the CDC suggest mortality due to CLABSI to be between 19-25% [16]. The high rates of drug-resistant organisms and the severity of the infections identified are alarming compared to western data. The reasons for the above were not known. One possibility was that patients were presenting later during the illness, with 50% of patients presenting with dyspnoea and 25% presenting with septic shock. 2 patients also had metastatic complications of septic arthritis and infective endocarditis. Both succumbed to their illness.
CONCLUSION
Mortality and hospitalisation owing to CLABSI were higher than in previous data. The CLABSI rates in this center were in keeping with those in developed countries. Gram-positive organisms were more common than gram-negative infections in this center. The rate of drug-resistant organisms was higher than expected compared to previous data. Adherence to infection control protocols was seen in 97.1% of cases, and further corrections must be made to reach 100% adherence to CDC guidelines. Of note, 66% of dressings were soiled on arrival and present an area for improvement where home care of the dialysis catheter should be improved.
LIST OF ABBREVIATIONS
| CLABSI | = Central Line Associated Blood Stream Infections |
| CDC | = Center for Disease Control |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the “Institutional Research Ethics committee” of SRIHER- Sri Ramachandra Institute of Higher Education and Research.
HUMAN AND ANIMAL RIGHTS
No animals were used for studies that are the basis of this research. All the humans used were in accordance with the ethical standards of Helsinki Declaration of 1975.
CONSENT FOR PUBLICATION
Informed consent has been obtained from all of the participants involved.
STANDARDS OF REPORTING
STROBE guidelines were followed.
AVAILABILITY OF DATA AND MATERIALS
The data supporting this study's findings are available within the article.
FUNDING
None.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


