All published articles of this journal are available on ScienceDirect.
Pelvic Floor Muscle Training for Stress Urinary Incontinence, with and without Biofeedback: A Systematic Review and Meta-analysis of Randomized Controlled Trials
Abstract
Background:
Pelvic floor muscle training (PFMT) with biofeedback is used widely in treating patients with stress urinary incontinence (SUI), despite unclear evidence. We conducted a meta-analysis of the literature to evaluate the efficacy of treatment after PFMT with and without biofeedback in SUI patients.
Methods:
We searched PubMed, CENTRAL, CINAHL, and Science Direct for randomized controlled trials (RCTs) of PFMT with and without biofeedback for SUI. RCTs were screened with our eligibility criteria, and the risk of bias was assessed according to the Cochrane risk of bias tool for randomized trials. The outcomes analyzed were pelvic floor muscle (PFM) strength, incontinence episode, daytime micturition, and nighttime micturition, all measured as mean difference (MD) with 95% confidence intervals (CIs). Heterogeneity and publication bias were analyzed using the I2 test and a funnel plot, respectively.
Results and Discussion:
Pooled analysis of five RCTs involving 207 patients showed that the difference in PFM strength and nighttime micturition between both groups was significant. Although PFM strength improvement favors biofeedback-assisted pelvic floor muscle training (BPFMT) (MD 12.29, 95% CI 2.33, 22.25, p=0.02), in contrast, nighttime micturition was significantly reduced in the PFMT group (MD 0.44, 95% CI 0.12 to 0.77, p=0.007). Differences in incontinence episode and daytime micturition were not significant (MD -0.08, 95% CI -0.57 to 0.41, p=0.75 and MD 0.55, 95% CI -0.36 to 1.46, p=0.24, respectively).
Conclusion:
This meta-analysis showed that BPFMT had a better outcome in improving PFM strength, while nighttime micturition was, on the contrary, better in PFMT only. Meanwhile, no significant differences in incontinence episodes and daytime micturition outcomes were noted between both groups. With the present evidence, routine use of BPFMT is not necessary for current clinical practice.
1. INTRODUCTION
Urinary incontinence has a global prevalence of 8.7% worldwide [1]. It is more common in women, experienced by 10% - 20% of all women and 77% of older women inhabiting nursing homes [2-7]. Despite the high prevalence, only about a quarter of women with urinary incontinence seek medical care, with only half of them eventually receiving treatment [8].
Stress urinary incontinence (SUI) is the most common etiology of urinary incontinence, defined as the involuntary loss of urine during physical activity that increases abdominal pressure. Stress urinary incontinence occurs when there is a failure of the urinary sphincter to withstand the pressure inside the bladder or failure of support of the lower urinary tract (bladder neck and urethra), resulting in urethral hypermobility. Both could happen concurrently, usually resulting from neurological disease, trauma, previous surgery, or in ageing women [9]. Physicians are required to explore risk factors including age, obstetric history, gynecological status, previous illnesses, daily activities, and pharmacological status in patients with urinary incontinence.
The treatment of SUI may include both non-surgical and surgical methods. Surgical therapy is typically utilized in patients who have failed non-surgical treatment, have moderate or severe SUI, poor quality of life, or have pelvic organ prolapse [10]. Traditional open surgery or minimally invasive surgery (laparoscopic) approach of Burch colposuspension, mid-urethral synthetic slings such as tension-free vaginal tape (TVT), trans obturator tape (TOT), and urethral bulking agents are among the procedures available [10-12]. Sling surgery is considered to be the current gold standard for treating SUI [10]. Lately, SUI treatment using stem cell therapy has a promising potential to improve the mechanism of urethral sphincter insufficiency, but further studies are needed in order to clarify the role of cell-based therapies for the treatment of SUI patients [13, 14]. The type of intervention should be tailored to the patient's symptoms, level of discomfort, goals and expectations, as well as the risks and benefits [12].
Patients more often choose conservative treatments rather than more invasive ones [15]. Pelvic floor muscle training (PFMT) is indicated for women with stress urinary incontinence, with less efficacy for urgency incontinence [16]. Aiming to improve pelvic floor muscle function, PFMT includes various exercises directed toward maximizing functional capabilities, including Kegels. Arnold Kegel first utilized Kegels to enhance the strength of floor muscles in 1940, and it is still widely used [17, 18]. Exercise may be done supervised or at home, with the latter currently emerging as it is more cost-effective [19]. Physicians use several ways to assist patients while exercising with electrostimulation, biofeedback (BF), and vaginal cones. Biofeedback is the most commonly used monitoring method for PFMT, with some studies showing no benefit from using BF augments [19-23]. Thus, the effectiveness of these adjunctive modalities remains unclear.
This study aims to systematically review and quantitatively analyze selected randomized controlled trials that evaluate PFMT and biofeedback-assisted pelvic floor muscle training (BPFMT) for patients with stress urinary incontinence.
2. MATERIALS AND METHODS
2.1. Study Methods
An extensive literature search was conducted through online databases, such as CINAHL, CENTRAL (Cochrane Library), MEDLINE (PubMed), and EMBASE (Science Direct) up to November 2021. Literature search process was carried out using medical subject headings (MeSH) terms of “stress urinary incontinence”, “pelvic floor muscle strength”, and “biofeedback”, and according to the preferred reporting items for systematic reviews and meta-analysis (PRISMA) guidelines. The literature search and selection process were performed independently by all authors involved.
2.2. Eligibility Criteria
Study literature was included only if it met the following criteria: RCT studies available in full text studies evaluating women with stress urinary incontinence who received BPFMT compared with PFMT only. Meanwhile, the exclusion criteria were inaccessible, with full text and irrelevant outcomes. The evaluated outcomes should include one of the following parameters: perineometry, incontinence episodes, daytime micturition, and nighttime micturition.
2.3. Data Collection and Statistical Analysis
Study inclusions were based on our eligibility criteria. Included studies were then examined by using the EndNote application for possible study duplication. Included studies were also assessed using the Cochrane Risk Index of Bias tool to determine the study quality. I2 test was performed to evaluate the heterogeneity of the study. If the I2 test result showed more than 50%, the heterogeneity was considered significant. On that occasion, a random-effects model would be selected for the analysis. Otherwise, if the I2 test result is valued at less than 50%, a fixed-effect model would be selected. We considered the analysis as statistically significant if the p-value was less than 0.05. All quantitative analysis was performed using Review Manager 5.4 software by Cochrane, Oxford, United Kingdom.
3. RESULTS
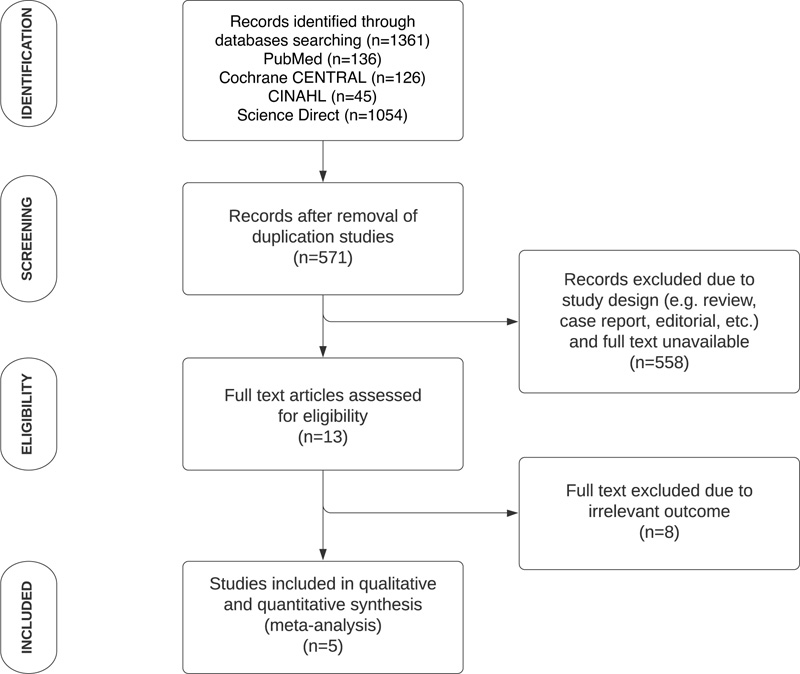
Summarization of the article selection process (Fig. 1) was conducted based on PRISMA guidelines. Initial database searching resulted in 1361 studies, and all were assessed using the EndNote application for study duplication. According to our inclusion criteria, the remaining 571 studies were then assessed manually by all authors. As many as thirteen articles were further analyzed for eligibility, resulting in five articles finally being analyzed qualitatively and quantitatively [24-28]. The outcomes analyzed in this study were: change of pelvic muscle floor (PFM) strength, incontinence episode, daytime micturition, and nighttime micturition. All data were pooled as mean difference (MD) since all of them were continuous data.
3.1. Study Characteristics
The analyzed studies were conducted in 4 different countries, two from Brazil, and the remaining three were from Germany, Japan, and Turkey (Table 1). A total of 96 patients received biofeedback as an adjuvant to PFMT, and 111 patients received PFMT alone. The duration of the intervention varied from 4 weeks to 12 weeks. When reported, the studies used different biofeedback methods and instruments.
| Study Authors | Year | Country | Design | Sample Size | Age | BMI (kg/m2) |
Training Period |
Outcome | BF Method | |
|---|---|---|---|---|---|---|---|---|---|---|
| BPFMT | PFMT Only | |||||||||
| Pages, et al. | 2001 | Germany | RCT | 13 | 27 | 51.1 | N/A | 4 weeks | Perineometry, daytime micturition, nighttime micturition, incontinence episode | Supine, intravaginal + sensor using Gemini 2000TM apparatus. Five times per week for 4 weeks, each for 15 minutes. |
| Aksac, et al. | 2003 | Japan | RCT | 20 | 20 | 52.5 | N/A | 8 weeks | Perineometry, incontinence episode, 1 hour pad test, VAS, PFM strength from digital palpation |
Lithotomy, intravaginal + sensor using Myomed-932. Three times per week for 8 weeks, each for 20 minutes. |
| Schmidt, et al. | 2009 | Brazil | RCT | 10 | 11 | 53.3 | 30.3 | 12 weeks | Perineometry, daytime micturition, nighttime micturition, KHQ questionnaire | Supine, intravaginal + sensor using device developed by the researcher team. First 12 weeks appointment were conducted at the hospital with evaluation after further 12 weeks at home without biofeedback |
| Hirakawa, et al. | 2013 | Turkey | RCT | 23 | 23 | 56.8 | 23.2 | 12 weeks | Perineometry, incontinence episode, 1 hour pad test, KHQ and ICIQ-SF questionnaire |
Position was not stated, intravaginal + sensor. Initial training at the hospital using FemiScan Clinic System, followed with further 12 weeks of training twice a day each for 8 minutes using FemiScan Home Trainer |
| Fitz, et al. | 2017 | Brazil | RCT | 30 | 30 | 56.3 | 29 | 12 weeks | Perineometry, incontinence episode, oxford scale | Supine for the 1st month, sitting for the 2nd month, standing for the 3rd month. intravaginal + sensor using Neurodyn Evolution pressure biofeedback. Twice a week for the first 3 months, each 40 minutes. Followed with further training at home for 6 months. |
SD, standard deviation; IV, inverse variance; CI, confidence interval.
SD, standard deviation; IV, inverse variance; CI, confidence interval.
3.2. Pelvic Muscle Strength Comparison
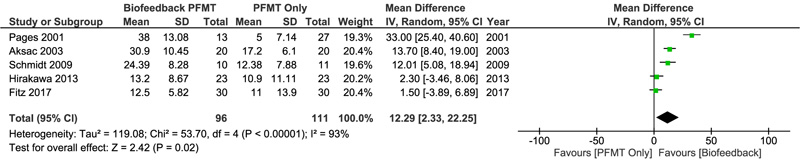
Five studies evaluating 207 participants were included in the pooled analysis for pelvic muscle strength between both groups. Significant heterogeneity was found in this analysis (I2=93%), so a random-effect model of the analysis was used. Pooled analysis in Fig. (2) showed that there was significant PFM strength improvement in the group receiving BPFMT compared to PFMT only (MD 12.29, 95% CI 2.33 to 22.25, p=0.02)
3.3. Incontinence Episode Comparison
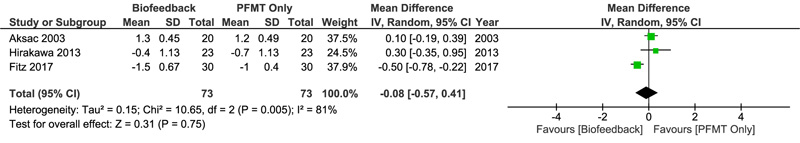
Three studies, including 146 subjects allocated into the BPFMT (n=73) and PFMT only (n=73) groups, described the number of incontinences experienced. Pooled analysis presented in Fig. (3) showed no significant difference between both treatment groups in terms of incontinence episodes that occurred (MD -0.08, 95% CI -0.57 to 0.41, p=0.75). A random-effects model was again selected for the calculation due to significant heterogeneity.
3.4. Daytime Micturition Comparison
Two studies with 61 women from both BPFMT (n=23) and PFMT only (n=38) groups reported daytime micturition frequency differences before and after treatment. Pooled analysis showed no significant difference between both groups (MD 0.55, 95% CI -0.36 to 1.46, p=0.24). Fig. (4) showed that the fixed-effect model was selected because no significant heterogeneity was found among the studies (I2=41%).
3.5. Nighttime Micturition Comparison
As well as the daytime micturition comparison, two studies involving 61 participants were also included for the nighttime micturition comparison before and after the treatment (23 from the BPFMT group and 38 from the PFMT group). Pooled MD values (Fig. 5) were estimated using a fixed-effects model of calculation since there was no significant heterogeneity among those two trials (I2=0%), resulting in a significant difference in nighttime micturition reduction, which favored the PFMT group (MD 0.44, 95% CI 0.12 to 0.77, p= 0.007).
4. DISCUSSION
In this meta-analysis, higher pelvic muscle strength was shown in patients undergoing BPFMT when compared to PFMT without BF group. In contrast, PFMT only group was associated with improved nighttime micturition. No significant difference was found in incontinence episodes and daytime micturition. These findings contradict the classical belief that stronger pelvic muscle is associated with better incontinence manifestations.
Despite various studies that have been undertaken to address the issue, there is little consensus on the effectiveness of PFMT over BPFMT. Pages et al. [24], Aksac et al. [25] and Schmidt et al. [26] found that the BPFMT group had much higher changes in PFM strength than the PFMT alone group. Some studies, such as Hirakawa et al. [27] and Fitz et al. [28], found no significant difference in PFM strength change in patients between the two groups.
Schmidt et al. [26] hypothesized that BPFMT would develop pelvic muscle floor strength even more because the patient can adjust or improve the training by seeing the intensity and contraction waveform as visual feedback. Another study by Mørkved et al. [29] found that using equipment during training could motivate many women and that it should be considered as a treatment option. Adding biofeedback to pelvic floor muscle training supports pelvic floor exercise, according to Berghmans et al. [30], it is most significant at the beginning of the therapy period. This may justify a considerable improvement in PFM function by boosting exercise performance and treatment motivation and as an easier guide for the patient to learn the exercises correctly [28].
PFM strength improvement following BPFMT or PFMT will consequently increase urethral sphincter strength [31]. The decrease in incontinence episodes and micturition frequency, either daytime or nighttime, are expected from the consequent increase of urethral sphincter strength [32]. Although BPFMT is associated with better PFM strength, in this meta-analysis BPFMT group does not manifest better outcomes in incontinence episodes, daytime micturition, and nighttime micturition.
In comparison to PFMT, Fitz et al. [28] found that BPFMT is related to improved incontinence episodes. However, investigations by Aksac et al. [25] and Hirakawa et al. [27] found that the BPFMT group had similar episodes of incontinence and daytime micturition to the PFMT group. These results are consistent with the findings of this review, which found no significant difference in incontinence episodes or daytime micturition between the BPFMT and PFMT alone groups.

SD, standard deviation; IV, inverse variance; CI, confidence interval

SD, standard deviation; IV, inverse variance; CI, confidence interval.
We discovered that BF has some advantages of its own, particularly in terms of pelvic muscle strength and function. Through audio-visual feedback, the BF supports patients in isolating and training the right muscle groups, which can be difficult to do without assistance [24]. Biofeedback may be a viable option, particularly for women with low or absent PFM strength or who are unaware of voluntary pelvic floor muscle contraction [27]. Although the use of devices may increase treatment costs; biofeedback provides the patient with an instrument to correctly conduct pelvic floor muscle workouts. Patients with more complicated disease presentations will likely benefit more from the additional information that biofeedback provides.
In summary, the results of all studies revealed heterogeneity in terms of BPFMT's efficacy as a treatment of choice for improving PFM strength. Unfortunately, full-text versions of some literature were not available during the research. Second, there was a scarcity of data on daytime and nighttime micturition. Evaluation variables, such as satisfaction rate, changes in quality of life, and events of recurrence incontinence should be considered in future research on the topic, as these are important clinical features to evaluate once the therapy is completed.
CONCLUSION
PFMT alone offers a reliable noninvasive exercise in improving incontinence symptoms with current evidence. Despite the significant PFM strength improvement associated with BPFMT, similar incontinence episodes were observed in BPFMT and PFMT. With periodical monitoring by healthcare professionals, PFMT may be done at home to increase cost-effectiveness and the number of hospital visits required throughout the exercise regimen.
LIST OF ABBREVIATIONS
| BF | = Biofeedback |
| BMI | = Body Mass Index |
| BPFMT | = Biofeedback-assisted Pelvic Floor Muscle Training |
| CI | = Confidence Interval |
| ICIQ-SF | = International Consultation On Incontinence Questionnaire – Short Form |
| IV | = Inverse Variance |
| Kg | = kilogram |
| KHQ | = King’s Health Questionnaire |
| m | = Meter |
| MD | = Mean Difference |
| MeSH | = Medical Subject Headings |
| PFM | = Pelvic Floor Muscle |
| PFMT | = Pelvic Floor Muscle Training |
| PRISMA | = Preferred Reporting Items For Systematic Reviews And Meta-analysis |
| RCT | = Randomized Controlled Trial |
| RR | = Risk Ratio |
| SUI | = Stress Urinary Incontinence |
| VAS | = Visual Analog Scale |
AUTHORS’ CONTRIBUTIONS
Study concept and design: Kevin Leonardo, Farhan Haidar Fazlur Rahman, Radhyaksa Ardaya; Data acquisition: Kevin Leonardo, Farhan Haidar Fazlur Rahman, Radhyaksa Ardaya; Data analysis: Kevin Leonardo, Farhan Haidar Fazlur Rahman, Radhyaksa Ardaya; Drafting of the manuscript: Kevin Leonardo, Farhan Haidar Fazlur Rahman; Critical revision of the manuscript: Kevin Leonardo, Radhyaksa Ardaya
CONSENT FOR PUBLICATION
Not applicable.
STANDARDS OF REPORTING
PRISMA guidelines were followed.
AVAILABILITY OF DATA AND MATERIALS
The data that support the findings of this study are openly available in the:
• American Journal of Physical Medicine and Rehabilitation at https://doi.org/10.1097/00002060-200107000-00006, reference number [19],
• Gynecologic and Obstetric Investigation at https://doi.org/10.1159/000072327, reference number [20],
• International Journal of Gynaecology and Obstetrics at https://doi.org/10.1016/j.ijgo.2009.01.013, reference number [21],
• International Urogynecology Journal at https://doi.org/10.1007/s00192-012-2012-8 and https://doi.org/10.1007/s00192-012-1707-1 reference number [22, 23].
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
FUNDING
None.
ACKNOWLEDGEMENTS
The authors thank Florentina Priscilla and Meutia Putri Aristya for their help in the preparation of this manuscript.
SUPPLEMENTARY MATERIAL
PRISMA checklist is available as supplementary material onthe publisher’s website along with the published article.


