All published articles of this journal are available on ScienceDirect.
PLA2R Staining is Useful for the Diagnosis and Treatment of Membranous Nephropathy in Pediatric Patients
Abstract
Background:
Idiopathic membranous nephropathy (IMN) is a rare disease in children. The incidence is 1.5% in children with nephrotic syndrome. A few studies have also investigated the M-type phospholipase A2 receptor’s (PLA2R) potential role in pediatric IMN, reporting a low positive rate in pediatric kidneys.
Objective:
Therefore, we conducted immunofluorescence staining using an anti-PLA2R antibody in the renal biopsy specimens of eight pediatric patients with IMN.
Methods:
We studied the glomerular expression of PLA2R using tissues from children with IMN, and searched for papers on PLA2R staining in pediatric IMN on PubMed.
Results and Discussion:
Clinical characteristics of patients diagnosed with IMN in this study and the other three studies: A total of 20 pediatric (aged 2–12 years; mean age 7.4 ± 2.8 years) patients and 25 adolescent (aged 13–19 years; mean age 15.9 ± 2.0 years) patients, comprising 25 male (55.6%) and 20 female (44.4%) patients, with 23 (51.1%) patients with IMN being PLA2R-positive, were found to be eligible for this study. Furthermore, we found three papers through our online search.
Conclusion:
PLA2R expression can be approximately half positive in children with IMN, and it is useful to investigate the causative antigen of PLA2R in children.
The intensity of anti-PLA2R antibody expression reflected the disease activity (urinary protein level) of the patients in this study.
It is possible to adjust the drug dose in immunosuppressive therapy with reference to the expression intensity of PLA2R.
1. INTRODUCTION
Membranous nephropathy (MN) is a type of glomerulonephritis. Immune complexes that form subcutaneously under the glomerular basement membrane cause glomerular epithelial disorders. In Japan, MN is the most common cause of adult nephrotic syndrome (NS).
In 2009, Beck reported the presence of a 200 kDa molecule that reacts with about 33% of the serum in idiopathic MN (IMN), which is called the M-type phospholipase A2 receptor (PLA2R) in the hoof epithelial cells [1]. An IgG4 subclass that reacts with PRA2R was specifically identified in approximately 70% of patients with IMN. PLA2R was found in normal human glomerular epithelial cells and was co-localized with IgG4 in immune deposits in the glomeruli of patients with IMN. IgG extracted from immune deposits of patients with IMN recognized PLA2R and decreased antibody titer. Improvement of urinary protein was shown with the course of treatment. These results indicate that the majority of patients with IMN have antibodies against the conformation-dependent epitope in PLA2R, indicating that PLA2R is the major self-antigen of IMN [1].
Subsequent studies reported that the sensitivity of the presence of PLA2R autoantibodies in the serum was 70%–82%, and the sensitivity of PLA2R staining in the glomerular tissue was 74%, which is a useful marker for IMN diagnosis [1, 2].
The antibody positive rate in bloodshed in Japanese IMN is about 50%, which is lower than the reports so far, but when the tissue expression is combined, the positive rate of either one is approximately 70% [3, 4].
However, most of these reports investigated adult patients with IMN.
IMN is a rare disease in children. The incidence is 1.5% in children with NS [5].
A few studies have also investigated PLA2R’s potential role in pediatric IMN, reporting a low positive rate in the pediatric kidney [6-8].
Therefore, we conducted immunofluorescence staining using an anti-PLA2R antibody in the renal biopsy specimens of eight pediatric patients with IMN.
2. MATERIALS AND METHODS
2.1. Patients
We enrolled eight patients and two patients with a second biopsy whose diagnosis of IMN was pathologically established using kidney biopsies at Kumamoto University Hospital and Kumamoto Chou Hospital from January 2015 to March 2021. We excluded patients with possible causes of secondary membranous nephropathy (SMN), such as autoimmune diseases, hepatitis viral infection, and malignancy. The following data were collected from the patients’ medical records: age, gender, serum creatinine (Cr), serum albumin, urine protein-to-creatinine ratio, hematuria at the time of renal biopsy, the period from onset to renal biopsy, treatment, and the period from treatment initiation to remission. We estimated the glomerular filtration rate using serum Cr according to the new Schwartz formula [9]. We also reviewed the results of light and electron microscopy and immunofluorescence staining of all patients. All direct immunofluorescence specimens using frozen sections were stained with fluorescein-tagged rabbit polyclonal antibodies to immunoglobulin G (IgG), IgA, IgM, complement component 1q (C1q), C3, and Fibrinogen.
The Institutional Ethics Committee approved this study (2018, approval number 1593). The patients or their family members provided informed consent for the analysis of PLA2R expression.
2.2. PLA2R Immunofluorescence
Frozen renal biopsy sections (4-µm-thick) were fixed in acetone for 10 min at room temperature for immunostaining. The sections were then incubated overnight at 4°C with rabbit polyclonal anti-PLA2R1 antibodies (ATLAS ANTIBODY, Sweden) at 1:1000 dilution. After that, primary antibodies were detected using Alexa Fluor 488-conjugated goat anti-rabbit IgG (Invitrogen, USA) at a dilution of 1:500 as secondary antibodies. We examined the sections under a conventional immunofluorescence microscope (Olympus, Japan). Two independent observers recorded PLA2R expression using an arbitrary scale as follows: (−), negative; (+), faintly positive; (2+), positive; and (3+) strongly positive.
2.3. Statistical Analysis
We searched for papers on PLA2R staining in pediatric IMN on PubMed and found three papers [10-12]. Data were expressed as numbers, percentages, and mean and standard deviation or median as appropriate. The chi-square test or Fisher’s exact test was used for association analysis to compare distributions between groups. All p values were two-tailed, with values of <0.05 considered statistically significant.
3. RESULTS
Table 1 shows the clinical data of the eight patients and two patients with a second biopsy who were diagnosed with IMN. The mean age at the time of biopsy was 7.6 years (range, 2–14 years), and there were four boys and four girls. Seven patients were diagnosed based on school or other urine screenings. One patient (case 7) was diagnosed based on the appearance of hematuria after streptococcal infection. NS was not found at the time of renal biopsy. At the time of renal biopsy, the average proteinuria level was 1.01 g/g·Cr, the average serum albumin level was 3.7 mg/dL, and serum Cr levels were normal in all patients.
Immunofluorescence examination revealed that all patients had granular deposits of IgG along the glomerular capillary loops, and three patients were positive for IgA staining along the glomerular capillary loops. Electron microscopy results demonstrated that all patients had subepithelial immune complex deposits, a characteristic finding of MN. Based on the Ehrenreich and Churg classification, one patient was in stage 1, one patient was in stage 2, four patients were in an intermediate stage between 1 and 2, and two patients were in an intermediate stage between 2 and 3. The stage was not examined in one patient.
As shown in Fig. (1), the immunofluorescence staining for PLA2R expression revealed weak (+) expression in three patients, moderate (2+) expression in one patient, and strong (3+) expression in one patient. After 3 years, the two patients who underwent a second renal biopsy showed a decreased number of glomeruli and positive PLA2R staining.
Case 7: This patient was a 3-year-old girl who was diagnosed based on the appearance of hematuria after streptococcal infection. She had moderate proteinuria (0.69 g/gCr) and hematuria at the time of renal biopsy.
Table 2 and Fig. (1) show this patient’s pathological characteristics.
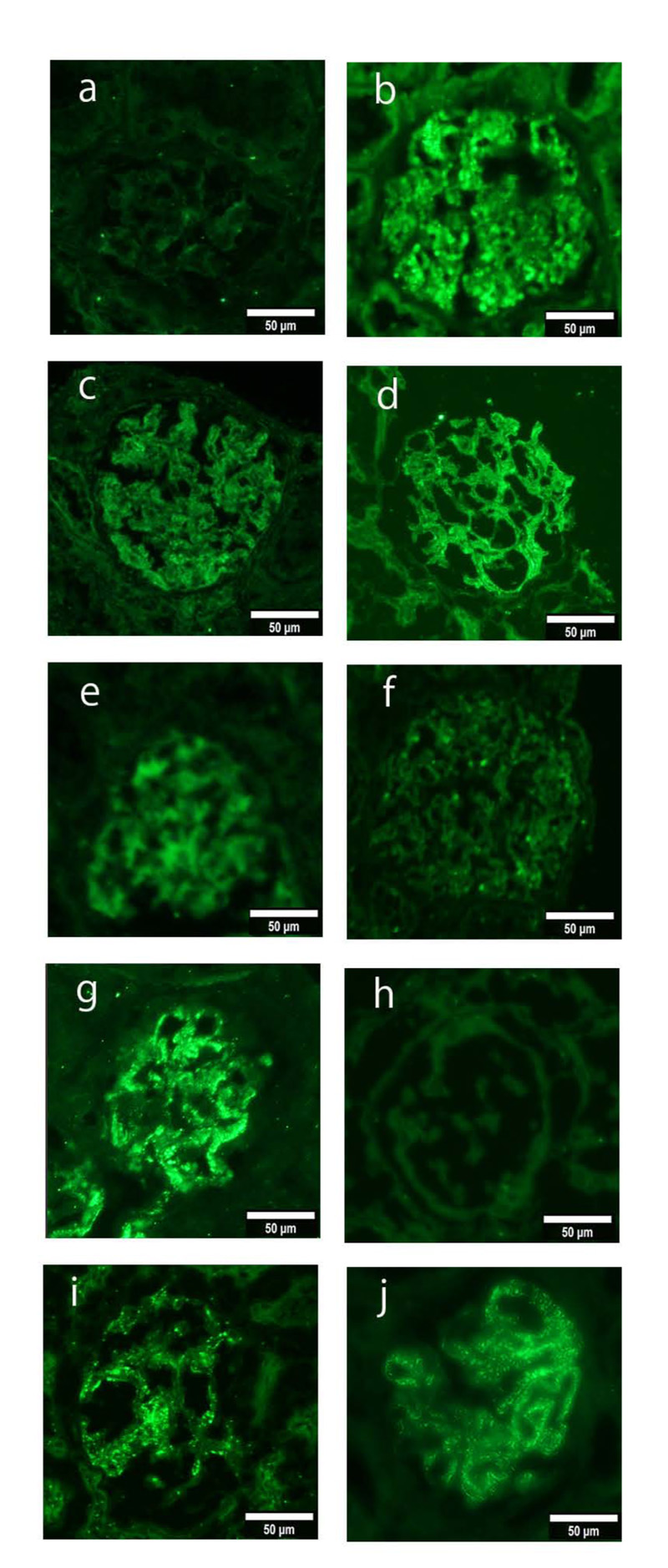
An immunofluorescence study of PLA2R (a-j; green), in biopsy samples from patients with IMN. Figure a is case1(PLA2R staining -), b is case2 (PLA2R staining 2+), c is case3 (PLA2R staining +), d is case4 (PLA2R staining +), e is case5 (PLA2R staining +), f is case6 (PLA2R staining -), g is case7 (PLA2R staining 3+), h is case8 (PLA2R staining -), i is second biopsy of case2 (PLA2R staining +), j is second biopsy of case7 (PLA2R staining +), Magnification: ×200 (a–j).
After a renal biopsy, her urinary protein level worsened, and 2 mg/kg prednisolone and ACE-I treatment were started. Immediately after the treatment, she entered into complete remission. Prednisolone dose was reduced and finally stopped within 3 months.
Thereafter, her urinary protein level worsened with each infection, but her urinary protein level improved spontaneously when the infection was cured. However, 2 years after the initiation of treatment, there was relapse due to the infection, and she resumed steroid treatment.
The second renal biopsy revealed improvement, showing PLA2R +m, but it was still PLA2R staining positive in the glomerular tissue, and hence mizoribine (MZR) treatment was added. At 6 months after the start of MZR treatment, her urinary protein level became negative. Fig. (2) shows the patient’s course of treatment.
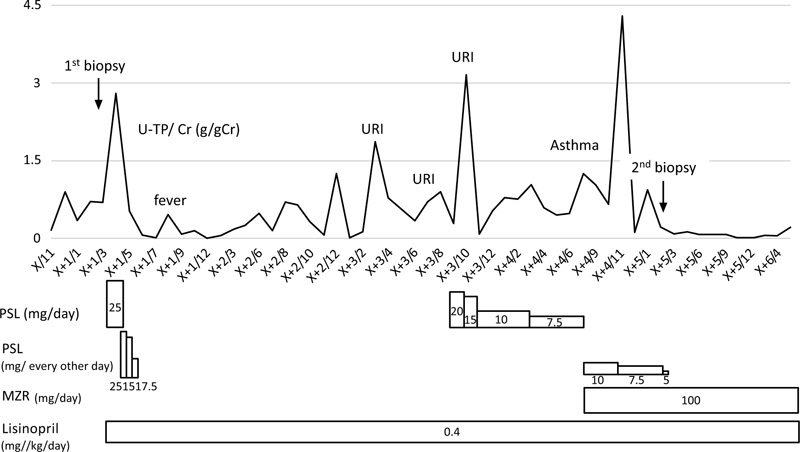
Urinary protein-creatinine ratio (U-P/Cr ratio) and the treatment.
Case 2: This patient was a 12-year-old boy diagnosed based on the appearance of proteinuria and hematuria in school urinalysis. There were no findings of SLE, such as complement reduction, rash, and joint pains. He had moderate proteinuria (0.76 g/g·Cr) and hematuria at the time of renal biopsy.
Table 2 and Fig. (1) show this patient’s pathological characteristics.
After the renal biopsy, he was started on 15 mg prednisolone, MZR, and ACE-I treatment. At 1 year after treatment, his urinary protein level became negative. At 2 years after starting treatment, the second renal biopsy revealed improvement with a decrease in PLA2R expression. However, hematuria was still positive, and hence treatment continued. One month after the second biopsy, the hematuria became negative. Fig. (3) shows the course of treatment of this patient.

Urinary protein-creatinine ratio (U-P/Cr ratio) and the treatment.
|
Age/Sex (Years) |
u-pro/cre | u-RBC(/HPF) | Albumin(g/dL) | Creatinine(mg/dL) |
e-GFR (ml/min/1.73m2) |
Medication | Outcome | |
| 1 | 14/F | 1.5 | 1-4 | 3.6 | 0.51 | 112.05 | PSL,MZR, ACE-I |
PR |
| 2 | 12/M | 0.76 | 20-30 | 4.0 | 0.35 | 227.9 | PSL,MZR, ACE-I |
CR |
| 3 | 9/M | 0.61 | 1-4 | 3.8 | 0.44 | 107.68 | ACE-I | CR |
| 4 | 7/F | 0.54 | 30-40 | 3.4 | 0.30 | 135.5 | PSL,MZR, ACE-I |
CR |
| 5 | 7/F | 0.54 | 30-49 | 3.6 | 0.37 | 114.41 | ACE-I | CR |
| 6 | 7/M | 1.37 | 50-100 | 3.4 | 0.51 | 133.6 | PSL,ACE-I | CR |
| 7 | 3/F | 0.69 | 20-29 | 3.8 | 0.36 | 88.8 | PSL,ACE-I | PR |
| 8 | 2/M | 2.04 | 30-40 | 4.0 | 0.20 | 169.35 | PSL,ACE-I | CR |
| 2-2 | 15/M | 0.22 | 20-29 | 4.0 | 0.35 | 240.4 | PSL,MZR, ACE-I |
CR |
| 7-2 | 7/F | 0.22 | 20-29 | 4.0 | 0.35 | 116.33 | PSL,MZR, ACE-I |
CR |
Table 1 shows the clinical data of the eight patients and two patients with a second biopsy who were diagnosed with IMN
| PLA2R | IgG | IgA | IgM | C3 | Fib | C1q | Electron Microscopy | |
| 1 | - | + | - | - | - | - | - | stage1-2 |
| 2 | 2+ | 2+ | + | + | + | 2+ | 2+ | stage2-3 |
| 3 | + | 2+ | - | - | - | + | - | stage1 |
| 4 | + | 2+ | + | - | ± | + | + | stage1-2 |
| 5 | + | 2+ | + | - | - | - | - | stage2 |
| 6 | - | 2+ | - | - | - | + | ± | stage1-2 |
| 7 | 3+ | + | - | - | - | + | - | stage1-2 |
| 8 | - | + | - | ± | ± | + | - | NE |
| 2-2 | + | ± | + | ± | - | - | + | NE |
| 7-2 | + | + | NE | NE | NE | NE | NE | stage2-3 |
Table 2 show this patient’s pathological characteristics. staining intensity on five- grade scale of 0 - 3+; 0, negative; <1+, spare; 1+, weak; 2+, moderate; 3+, strong
3.1. Clinical Characteristics of Patients Diagnosed with IMN in this Study and the other Three Studies
A total of 20 pediatric (aged 2–12 years; mean age 7.4 ± 2.8 years) patients and 25 adolescent (aged 13–19 years; mean age 15.9 ± 2.0 years) patients, comprising 25 male (55.6%) and 20 female (44.4%) patients, with 23 (51.1%) patients with IMN being PLA2R-positive, were found to be eligible for this study. Table 3 shows the clinical characteristics of the patients.
3.2. Comparison of Clinical Characteristics between PLA2R-positive and PLA2R-negative Patients with IMN
As shown in Table 4, PLA2R-positive patients had less complete remission (CR) compared with PLA2R-negative patients, showing a significant difference (8/23 (34.8%) vs. 16/22 (72.7%), p = 0.011).
3.3. Comparison of Clinical Characteristics between Adolescent and Pediatric Patients with IMN
As shown in Table 5, PLA2R staining was positive in 15/25 (60.0%) adolescent patients with IMN and in 8/20 (40.0%) pediatric patients. The adolescent population consisted of more PLA2R-positive patients, but there was no significant difference between pediatric and adolescent patients. The adolescent population had more NS+ (5/18 (27.8%) vs. 20/25 (80.0%), p = 0.001) and less CR (16/20 (80.0%) vs. 8/25 (32.0%), p = 0.001) compared with pediatric patients.
4. DISCUSSION
MN is classified into IMN (approximately 80% of cases) and SMN (approximately 10%–20% of cases) with various etiologies. The cause of IMN remains unknown, and it is conceptually considered a kidney-specific autoimmune disease. Autoantibodies in the blood bind to target antigens on the foot process of glomerular epithelial cells form immune complexes on the epithelium, accompanied by complement activation, and finally, cause proteinuria due to epithelial cell damage.
In recent years, studies have reported that approximately 75% of IMN cases have PLA2R as the causative antigen, and autoantibodies against PLA2R may be a marker of therapeutic responsiveness and the cause of the onset of IMN [1, 13, 14].
| - |
Age/Sex (Years) |
Nephrotic Range Proteinuria |
Albmine (g/dL) |
e-GFR (ml/min/1.73m2) |
Creatinine (mg/dL) |
PLA2R | Medication | Outcome | Ref |
| 1 | 14/f | No | 3.6 | 112.05 | 0.51 | - | PSL,MZR,ACE-I | PR | This case |
| 2 | 9/m | No | 3.8 | 107.68 | 0.44 | + | ACE-I | CR | This case |
| 3 | 7/f | No | 3.6 | 114.41 | 0.37 | + | ACE-I | CR | This case |
| 4 | 3/f | No | 3.8 | 88.8 | 0.36 | + | PSL,ACE-I | PR | This case |
| 5 | 2/m | Yes | 4 | 169.35 | 0.2 | - | PSL,ACE-I | CR | This case |
| 6 | 7/f | No | 3.4 | 135.5 | 0.3 | - | PSL,MZR,ACE-I | CR | This case |
| 7 | 7/m | No | 3.6 | 136 | 0.4 | + | PSL,ACE-I,ARB | CR | This case |
| 8 | 12/m | No | 4 | 227.9 | 0.35 | + | PSL,MZR,ACE-I | CR | This case |
| 9 | 4/f | No | 2.9 | 176.8 | 0.25 | - | ACE-I | CR | 10 |
| 10 | 14/f | Yes | 1.2 | 134.9 | 0.49 | - | PSL | CR | 10 |
| 11 | 6/m | No | 1.6 | 138.5 | 0.34 | - | PSL,ACE-I,ARB | CR | 10 |
| 12 | 4/m | NA | 4.1 | 143.2 | 0.3 | - | PSL | CR | 10 |
| 13 | 6/m | NA | 3.5 | 118.7 | 0.4 | - | PSL | CR | 10 |
| 14 | 5/m | Yes | 2.1 | 131.4 | 0.33 | - | PSL | CR | 10 |
| 15 | 11/m | No | 3.9 | 144.6 | 0.38 | - | ACE-I | CR | 10 |
| 16 | 6/m | No | 3 | 168.2 | 0.28 | + | ACE-I | CR | 10 |
| 17 | 7/f | No | 4.4 | 124.9 | 0.43 | - | ACE-I | CR | 10 |
| 18 | 8/m | No | 2.9 | 170.4 | 0.32 | - | ACE-I | PR | 10 |
| 19 | 11/f | No | 2.8 | 170.1 | 0.34 | - | ACE-I | PR | 10 |
| 20 | 13/m | No | NA | 135 | 0.46 | + | PSL,CsA | NR | 11 |
| 21 | 14/f | Yes | NA | 99 | 0.7 | + | PSL,Tac,RTX | CR | 11 |
| 22 | 14/f | Yes | NA | 113 | 0.6 | + | PSL,CsA,MMF,Tac | PR | 11 |
| 23 | 15/f | No | NA | 152 | 0.42 | + | PSL | NR | 11 |
| 24 | 17/m | No | NA | 78 | 0.9 | - | No | CR | 11 |
| 25 | 10/m | Yes | NA | 154 | 0.4 | - | PSL,CsA | CR | 11 |
| 26 | 13/m | Yes | NA | 146 | 0.46 | - | MMF | NR | 11 |
| 27 | 14/f | No | NA | 135 | 0.49 | - | PSL,MMF | PR | 11 |
| 28 | 12/f | Yes | 3.4 | NA | NA | + | PSL,CTX | CR | 12 |
| 29 | 17/f | Yes | 1.82 | NA | NA | - | PSL,Tac | CR | 12 |
| 30 | 17/f | Yes | 1.9 | NA | NA | + | PSL,Tac | NR | 12 |
| 31 | 16/m | Yes | 2.3 | NA | NA | - | PSL | CR | 12 |
| 32 | 16/f | Yes | 2.1 | NA | NA | + | PSL,CTX | CR | 12 |
| 33 | 14/m | Yes | 1.1 | NA | NA | + | PSL,CTX | NR | 12 |
| 34 | 13/m | Yes | 1.91 | NA | NA | + | PSL,CTX | PR | 12 |
| 35 | 17/m | Yes | 1.5 | NA | NA | - | PSL,CTX | NR | 12 |
| 36 | 11/f | Yes | 3.2 | NA | NA | + | PSL,Tac | PR | 12 |
| 37 | 17/m | Yes | 2.2 | NA | NA | + | PSL | NR | 12 |
| 38 | 18/f | Yes | 1.5 | NA | NA | + | PSL,Tac | NR | 12 |
| 39 | 18/m | Yes | 1.9 | NA | NA | + | PSL,Tac | NR | 12 |
| 40 | 18/m | Yes | 1.8 | NA | NA | - | PSL,CTX | CR | 12 |
| 41 | 19/m | Yes | 1.35 | NA | NA | + | No | RF | 12 |
| 42 | 15/m | Yes | 3.2 | NA | NA | - | PSL,Tac | PR | 12 |
| 43 | 17/f | Yes | 1.04 | NA | NA | + | No | RF | 12 |
| 44 | 19/m | Yes | 2.2 | NA | NA | + | PSL,CTX | PR | 12 |
| 45 | 19/m | Yes | 1.53 | NA | NA | + | PSL,CTX | NR | 12 |
Table 3 shows the clinical characteristics of the patients. A total of 20 childhood (aged 2–12 years; mean age 7.4 ± 2.8 years) patients and 25 adolescent (aged 13–19 years; mean age 15.9 ± 2.0 years) patients, comprising 25 male (55.6%) and 20 female (44.4%) patients, with 23 (51.1%) patients with PLA2R-positive
| - | PLA2R(+) (n=23) | PLA2R(-) (n=22) | P |
| Male | 12/23(52.2%) | 14/22(63.6%) | 0.436 |
| Childhood | 8/23(34.8%) | 12/22(54.5%) | 0.144 |
| NS | 15/23(65.2%) | 10/20(50.0%) | 0.313 |
| CR | 8/23(34.8%) | 16/22(72.7%) | 0.011 |
All p values were two-tailed, with values of <0.05 considered statistically significant.
| - | Childhood (n=20) | Adolescent (n=25) | P |
| PLA2R(+) | 8/20(40.0%) | 15/25(60.0%) | 0.182 |
| Male | 12/20(60.0%) | 14/25(56.0) | 0.787 |
| NS(+) | 5/18(27.8%) | 20/25(80.0%) | 0.001 |
| CR(+) | 16/20(80.0%) | 8/25(32.0%) | 0.001 |
All p values were two-tailed, with values of <0.05 considered statistically significant.
Only a few studies have reported PLA2R-positive pediatric IMN cases globally, and Cossey et al. reported that of 22 children with IMN, 10 (45%) were positive for PLA2R staining in the renal tissue [6]. Furthermore, Kumar et al. reported that serum anti-PLA2R antibody positivity in pediatric IMN was lower in three of five cases (60%) than that in adults [8].
Nevertheless, no studies have investigated the treatment responsiveness and prognosis of pediatric patients with PLA2R-positive IMN, and further research is needed based on the accumulation of future cases.
In our cases, the PLA2R antibody positivity rate in pediatric IMN was as high as 62.5%. In this survey as well, the result was 50%. Although the positive rate was reportedly higher in adolescents than in children, the positive rate in the present study tended to be higher in adolescents, but there was no significant difference.
Due to the differences in clinical features and the varying positive PLA2R staining rates among adults, adolescents, and children, PLA2R staining may be identified as the etiology of disease for half of the children with IMN. Debiec et al. identified neutral endopeptidase in podocytes as an antigen for antenatal membranous nephropathy in infant patients with MN [15]. They also detected antibodies to dietary cationic bovine serum albumin in young infants with MN [16]. Therefore, more diverse and underlying causes for IMN in the pediatric population remain to be identified. In particular, a previous study suggested that PLA2R expression is absent in early-stage IMN [17]. This suggests that PLA2R positivity in pediatric IMN is lower than that in adult IMN.
As the incidence of pediatric IMN remains unclear, and there are few cases Japan, there is no established treatment policy and conducting randomized controlled trials is difficult.
Because the prognosis of IMN is good, aggressive treatment may be unnecessary in most cases.
However, there are cases of developing NS and presenting end-stage renal disease in pediatric IMN.
In adult MN, risk stratification is performed, and the treatment policy is determined based on the risk; however, in children, the treatment policy varies according to the institution.
The anti-PLA2R antibody titer in serum reflects disease activity (urinary protein level) in adults, such as spontaneous remission of MN, relapse, or decreased renal function. It is considered possible to predict the prognosis of IMN.
Moreover, the drug dose in immunosuppressive therapy can be adjusted with reference to the antibody titer.
In fact, in adults, the time to remission is prolonged with high antibody titers [18-20].Currently, it is useful to investigate the causative antigen of PLA2R even in children, and the expression intensity of anti-PLA2R antibody reflects the disease activity (urinary protein level) and the spontaneous remission or relapse of IMN. Alternatively, it is possible to predict a decrease in renal function.
In addition, it is possible to adjust the drug dose in immunosuppressive therapy regarding the expression intensity of PLA2R.
We recommend the algorithm shown in Fig. (4) for the diagnosis and treatment of pediatric IMN.
This study had some limitations. First, only a small sample of patients was included. Second, the serum PLA2R with patients was not investigated and IgG subclass with patients renal tissue did not stain. Although PLA2R staining of glomerular tissue is equivalently or more sensitive than serological measurements for the diagnosis of IMN in adults, if we had evaluated serum anti-PLA2R antibodies, it might have been revealed that the serum findings correlate with the renal biopsy findings [10].
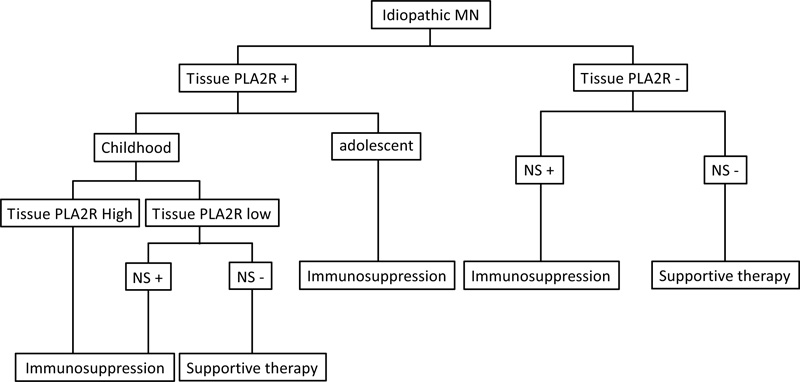
Algorithm for diagnosis and treatment decisions in pediatric membranous nephropathy. If a patient is known to have PLA2R-associated MN by virtue of biopsy staining for PLA2R within the deposits, only those nephrotic patients with high or rising staining for PLA2R require immunosuppression. Treatment decisions in patients with non- PLA2R should be made on clinical grounds.
Third, the relationship between renal function and PLA2R was not examined. Future studies with a higher number of patients are required to confirm these findings.
CONCLUSION
PLA2R expression can be approximately half positive in pediatric patients with IMN, and it is useful to investigate the causative antigen of PLA2R in-pediatric patients. The intensity of anti-PLA2R antibody expression reflected the disease activity (urinary protein level) of the patients in this study. It is possible to adjust the drug dose in immunosuppressive therapy with reference to the expression intensity of PLA2R.
LIST OF ABBREVIATIONS
| MN | = Membranous Nephropathy |
| NS | = Nephrotic Syndrome |
| IMN | = Idiopathic MN |
| PLA2R | = Phospholipase A2 Receptor |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The Institutional Ethics Committee approved this study (2018, approval number 1593).
HUMAN AND ANIMAL RIGHTS
No animals were used for studies that are the basis of this research. All the humans were used in accordance with the Helsinki Declaration of 1975.
CONSENT FOR PUBLICATION
All participants signed an informed consent form. This study does not disclose personally identifiable information of any of the participants in any form.
STANDARDS OF REPORTING
STROBE guidelines were followed.
AVAILABILITY OF DATA AND MATERIALS
The datasets generated during and/or analyzed during the current study are available from the corresponding author [H.T] on reasonable request.
FUNDING
The authors received no financial support for the research, authorship, and/or publication of this article.
CONFLICT OF INTEREST
All authors declare that this manuscript has no conflict of interest.
ACKNOWLEDGEMENTS
We would like to thank the patient for participating in this study.


