All published articles of this journal are available on ScienceDirect.
Identifying Predictors for Rapid Kidney Function Deterioration among Chronic Kidney Disease Patients in Thailand Settings of Real-life Practice
Abstract
Background:
Chronic kidney disease (CKD) and progression to end-stage renal disease (ESRD) are major public health problems around the world. In Asia, the incidence of ESRD is increasing and consuming more health expenditures. Prevention of ESRD is essential.
Objective:
This study aims to assess for factors associated with a rapid decline of estimated glomerular filtration rate(eGFR) among the Thai CKD population.
Methods:
This was a retrospective cohort study of 1,263 CKD patients in a tertiary university hospital from July 2013 to December 2021. Patients were categorized into 2 groups based on their annual rate of eGFR decline: the rapid group (>5 ml/min/1.73 m2 /year) and the non-rapid group (≤5 ml/min/1.73 m2 /year). All baseline and follow-up variables were assessed to identify predictors of rapid eGFR decline.
Results:
The median rate of eGFR decline was 3.0 ml/min/1.73m2/year in the non-rapid group (362 patients) and 6.6 ml/min/1.73m2/year in the rapid group (901 patients). Patients in the rapid group had a longer median follow-up time than in the non-rapid group (7.7 years versus 7.1 years respectively). In multivariate logistic analysis, diabetic mellitus, high baseline serum total carbon dioxide (TCO2), NSAID usage, and ACEI usage predicted rapid eGFR decline, but serum albumin during follow-up protected rapid eGFR decline.
Conclusion:
The main predictors for rapid kidney function deterioration were diabetic mellitus, high serum TCO2 during follow-up, NSAID usage, and ACEI usage. Serum albumin during follow-up protected kidney function.
1. INTRODUCTION
Chronic kidney disease (CKD) is a serious public health concern across the world and significantly contributes to the global burden of disease complications when progressing to end-stage renal disease (ESRD) [1]. Furthermore, CKD is associated with a low quality of life, a financial burden on healthcare systems as well as a high risk of hospitalization and cardiovascular disease (CVD) event [2, 3]. In Asia, the CKD burden is increasing. According to previous literatures, studies on CKD prevalence among 16 countries in Asia found that up to 434.3 million adults have CKD, with up to 65.6 million patients having advanced CKD leading to consuming more healthcare expenses [4]. Hence, preventing CKD progression is the goal of treatment.
The Kidney Disease Improving Global Outcomes (KDIGO) 2012 Clinical Practice Guideline for The Evaluation and Management of Chronic Kidney Disease emphasizes identifying CKD patients with rapid estimated glomerular filtration rate(eGFR) decline which is defined as a sustained decline in eGFR of more than 5 ml/min/1.73 m2 /year because of the more certain rapid progression, the more increased risk of all causes mortality [5, 6]. Several studies report the factors associated with rapid loss of glomerular filtration rate (GFR) in CKD, including baseline proteinuria, low serum albumin, elevated systolic blood pressure, previous cardiovascular disease, and diabetes mellitus [7, 8]. The landmark study in identifying the risk of CKD progression is The Chronic Renal Insufficiency Cohort (CRIC) Study. This study was conducted in the US and found the relationship between low estimated glomerular filtration rate (eGFR) by Modification of Diet in Renal Disease (MDRD) estimated equation and CVD. Although the study included patients with various ethnicities, the rate of eGFR decline was affected by race and Asians tend to have a faster rate of CKD progression than Caucasians [9, 10]. Furthermore, The Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation more accurately classified the risk for mortality and ESRD than the MDRD Study equation using the same variables across a broad range of populations [11].
For the Asian population, a recent large longitudinal analysis conducted in Japan collected data from 3,673,829 patients who underwent health check-ups. The study identified several factors associated with a decline in kidney function, including high systolic blood pressure, inadequate or strict control of diabetes mellitus, increased urinary protein excretion, and decreased blood hemoglobin levels [12]. Furthermore, a study from South Korea revealed that patients in the rapid glomerular filtration rate (GFR) decline group are associated with poor mental and physical health outcomes [13]. Therefore, identifying factors contributing to rapid GFR decline assumes significant importance. However, Asia comprises numerous countries, each possessing unique characteristics and region-specific genotypes. To our knowledge, there are few studies examining the risk of CKD progression in the Thai population. Therefore, this study aims to identify the factors associated with rapid eGFR decline among CKD patients in Thailand settings of real-life practice.
2. MATERIALS AND METHODS
2.1. Study Designs
This was a retrospective cohort study conducted in Songklanagarind Hospital which is a university hospital in Southern Thailand from July 2013 to December 2021. The study enrolled adult patients aged more than 18 years old diagnosed with CKD stage 3-4 according to KDIGO 2012 guideline defined as eGFR 15-59 ml/min/1.73 m2. eGFR was calculated by using serum creatinine from enzymatic methods with the CKD-EPI formula. Patients were followed up until death or December 2021. Exclusion criteria were patient loss to follow-up for 24 months, body mass index (BMI) < 18.5 kg/m2, history of knee amputation, cirrhosis, Child-Pugh C, pregnancy, and received medications that affect serum creatinine such as cimetidine, fenofibrate, trimethoprim, cobicistat, dolutegravir, rilpivirine.
2.2. Outcome Measurement
Eligible patients were categorized into two groups; the non-rapid group (eGFR decline ≤5 ml/min/1.73 m2/year) and the rapid group (eGFR decline >5 ml/min/1.73 m2/year). The electronic medical records of participants were reviewed and collected data such as age, gender, blood pressure, body weight, height, body mass index (BMI), comorbidities, and disease by searching from the International Classification of Disease, 10th revision (ICD 10). We collected laboratory results at baseline and follow-up periods. Medication and other potential events that affected CKD progression were assessed as follows; angiotensin-converting enzyme inhibitors (ACEI), angiotensin receptor blockers (ARB), sodium bicarbonate, lipid-lowering agents, non-steroidal anti-inflammatory drugs (NSAIDs), history of congestive heart failure, history of angiography, history of exposure to contrast media, coronary artery bypass surgery. The CKD-EPI formula was used to calculate eGFR = for women with plasma creatinine < 0.7, (plasma creatinine / 0.7)-0.329 x (0.993)age (x166 if black; x 144 if white or other); for women with a plasma creatinine > 0.7, (plasma creatinine / 0.7)-1.029 x (0.993)age (x166 if black; x 144 if white or other); for men with a plasma creatinine < 0.9; (plasma creatinine / 0.9)-0.411 x (0.993)age (x163 if black; x 141 if white or other); for men with a plasma creatinine > 0.9, (plasma creatinine / 0.9)-1.209 x (0.993)age (x166 if black; x 144 if white or other) [14]. The values of each variable observed during the follow-up period represent the change in the variable between the study's beginning and end. The primary outcome was to evaluate clinical and laboratory parameters that predict rapid eGFR decline among CKD patients. The secondary outcomes were the rate of eGFR decline between the two groups and time to ESRD or renal survival between diabetes and non-diabetes.
2.3. Statistical Analysis
R software 4.1.1 (R Foundation for Statistical Computing, Vienna, Austria) and RStudio 1.2.5001 (RStudio, Inc., Boston, MA) were used for statistical calculations. The continuous variables were expressed as means ± standard deviation or median(interquartile), and categorical variables were expressed as frequency and percentage. For comparison between groups, the student’s t-test was used for continuous variables and the chi-square test was performed for categorical variables. The multivariate logistic regression was performed to evaluate the association between rapid eGFR decline with clinical and laboratory variables selected by important parameters with statistical significance in the univariate analysis. The Kaplan–Meier method was used to estimate renal survival time. A P-value < 0.05 was considered statistically significant.
3. RESULTS
1,263 patients were enrolled. 362 patients were in the non-rapid group and 901 patients were in the rapid group. The median rate of eGFR decline was 3.0 ml/min/1.73m2/year in the non-rapid group and 6.6 ml/min/1.73m2/year in the rapid group. Baseline characteristics are shown in Table 1. The median age was 69.5 and 70 years in non-rapid and rapid groups, respectively. No differences in gender, blood pressure, and BMI between the two groups were observed. Diastolic blood pressure was lower in the rapid group (77 mmHg versus 80 mmHg; p = 0.044). There were significantly higher proportions of diabetic patients in the rapid group than the non-rapid group (32.4% versus 22.7%; p <0.001). The other parameters revealed no significant difference between the two groups.
The clinical and laboratory parameters during follow-up are shown in Table 2. Patients in the rapid group had a longer median follow-up time (7.7 years versus 7.1 years). Diastolic blood pressure and serum albumin were significantly lower in the patients with the rapid group, whereas urine protein to creatinine ratio (UPCR) and serum total carbon dioxide (TCO2) were higher among these groups. The history of contrast media exposure and cardiovascular events were not different between the two groups. There was a higher percentage of participants who used ACEIs, ARBs, metformin, statins, and NSAIDs in the rapid group than the non-rapid group.
The multivariate logistic regression analysis showed that there were 5 parameters significantly associated with rapid eGFR decline as shown in Table 3. Diabetes mellitus, serum TCO2 during follow-up, NSAIDs usage, and ACEIs usage predicted kidney function deterioration, while serum albumin during follow-up protected kidney function deterioration.
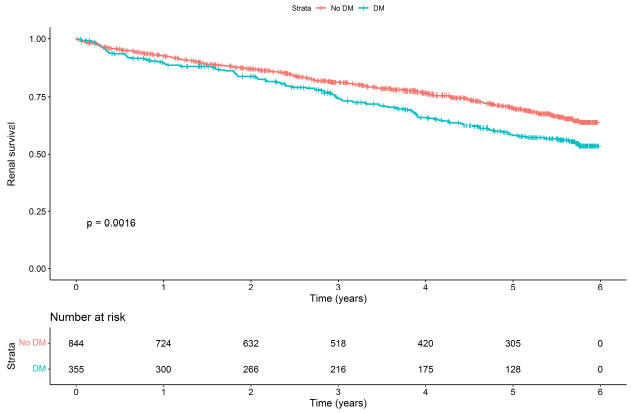
The 6-year renal survival rate in CKD patients with diabetes was significantly lower than patients without diabetes (58.2% versus 70.2%; p = 0.0016). The Kaplan-Meier curves between these groups were shown in Fig. (1).
| Variables | Non-rapid eGFR Decline (n=356) |
Rapid eGFR Decline (n=901) |
P-value |
|---|---|---|---|
| Age | 69.5 (60,77) | 70 (61,78) | 0.451 |
| Gender (male %) | 235 (64.9) | 613 (68) | 0.317 |
| Body mass index (kg/m2) | 25.2 (22.7,27.4) | 25.3 (23.4,28.2) | 0.482 |
| Comorbidities (%) | - | - | - |
| Hypertension | 184 (50.8) | 441 (48.9) | 0.587 |
| Diabetes mellitus | 82 (22.7) | 292 (32.4) | < 0.001 |
| Dyslipidemia | 70 (19.3) | 200 (22.2) | 0.296 |
| Ischemic heart disease | 31 (8.6) | 67 (7.4) | 0.575 |
| Systolic blood pressure (mmHg) | 142 (126,158.8) | 140 (126,156.8) | 0.395 |
| Diastolic blood pressure (mmHg) | 80 (70,90) | 77 (68,87) | 0.044 |
| Hemoglobin (g) | 11.8 ± 2.2 | 11.6 ± 2.3 | 0.192 |
| eGFR CKD-EPI (ml/min/1.732) | 35 (25,44.5) | 37 (25.5,50.5) | 0.238 |
| Serum potassium (mEq/L) | 4.3 (3.9,4.6) | 4.2 (3.8,4.5) | 0.242 |
| Serum TCO2 (mEq/L) | 21.3 (2.4) | 21.6 (2.2) | 0.259 |
| Serum cholesterol (mg/dL) | 203.5 (168.2,233.5) | 203 (171,240) | 0.897 |
| Serum albumin (mg/dL) | 4.3 (3.9,4.5) | 4.2 (3.8,4.5) | 0.150 |
| Serum uric acid (mg/dL) | 8 (7.1,9.1) | 7.7 (6.5,9.4) | 0.620 |
| Serum calcium (mg/dL) | 9 (8.7,9.4) | 9.3 (8.9,9.6) | 0.363 |
| UPCR (mg/mg) | 0.68 (0.13,3.81) | 0.70 (0.34,4.14) | 0.301 |
| Variables | Non-rapid eGFR Decline (n=356) |
Rapid eGFR Decline (n=901) |
P-value |
|---|---|---|---|
| Duration of follow up | 7.1 (3.6,9.9) | 7.7 (3.9,10.4) | 0.005 |
| Systolic blood pressure (mmHg) | 139.2 (129.8,149) | 139 (130.1,148.5) | 0.611 |
| Diastolic blood pressure (mmHg) | 76.7 (70.3,84.1) | 75.8 (69.3,82.3) | 0.043 |
| Hemoglobin (g) | 11.4 (10.1,12.8) | 11.3 (9.8,12.9) | 0.321 |
| Serum albumin (g/dl) | 4.2 (4.0,4.4) | 3.8 (3.7,4.3) | < 0.001 |
| Serum TCO2 (mEq/L) | 22.6 (19.9,25.1) | 23.5 (21.4,25.6) | 0.011 |
| Serum uric acid (mg/dL) | 7.8 (6.8,8.7) | 7.5 (6.5,8.7) | 0.089 |
| UPCR (mg/mg) | 0.70 (0.28,1.54) | 0.83(0.30,1.57) | 0.008 |
| Contrast media exposure (%) | 11 (3) | 40 (4) | 0.324 |
| Cardiovascular events (%) | 29 (8.5) | 75 (8.7) | 1 |
| ACEIs usage (%) | 107 (31.5) | 391 (45.4) | < 0.001 |
| ARBs usage (%) | 38 (11.2) | 149 (17.3) | 0.017 |
| Metformin usage (%) | 44 (12.9) | 269 (31.2) | < 0.001 |
| Statin usage (%) | 216 (63.5) | 603 (70) | 0.037 |
| Sodium bicarbonate (%) | 263 (77.4) | 649 (75.3) | 0.498 |
| Diuretic drug (%) | 311 (91.5) | 787 (91.3) | 1 |
| NSAIDs usage (%) | 69 (19.1) | 240 (26.6) | 0.006 |
| Variable | Crude OR (95%CI) | Adjusted OR (95%CI) | P-value |
|---|---|---|---|
| Diabetic mellitus | 2.8 (1.92,4.06) | 2.48 (1.66,3.7) | < 0.001 |
| Serum TCO2 (mEq/L) * | 1.04 (0.98,1.11) | 1.13 (1.05,1.21) | < 0.001 |
| Serum albumin (mg/dL) ** | 0.36 (0.26,0.52) | 0.29 (0.2,0.43) | < 0.001 |
| NSAIDs usage | 1.41 (1,1.98) | 1.44 (1.01,2.07) | 0.043 |
| ACEIs usage | 1.64 (1.22,2.21) | 1.28 (1.03,1.76) | 0.013 |
*Serum TCO2 during follow up
**Serum albumin during follow up
4. DISCUSSION
This large retrospective cohort study of CKD patients found that the rate of eGFR decline was 6.6 ml/min/1.73m2/year in the rapid group. The rapid kidney function decline among CKD patients is associated with DM, the high baseline serum TCO2, the low level of serum albumin during follow-up, and two medication used as NSAIDs and ACEIs. Again, we found that renal survival in a patient with DM is significantly lower than a patient without this condition.
DM remained the best predictor of yearly rapid eGFR decrease after accounting for known risk variables and confounders. This result was similar to previous literatures reported by Zoppini et al [15] that demonstrated a significantly faster rate of eGFR reduction in diabetes. DM patients had a faster decline in renal function during the observation period (Fig. 1). This is consistent with the study conducted by Fujii et al [12] that reported 20.83% of patients with DM had a rapid decline in renal function for approximately 1 year. Various mechanisms, such as albuminuria, inflammation, impaired angiogenesis, excessive activation of regenerative pathways, as well as genetic and epigenetic factors, may be associated with the progression of chronic kidney disease in diabetic patients [16-22]. These mechanisms need to be further investigated through analyses that incorporate additional molecular outcomes.
We found that elevated serum bicarbonate during follow-up was associated with rapid kidney function decline. These contradict the MESA study conducted by Driver et al. [23]. The serum bicarbonate below 21 mEq/L is linked to a 35% greater risk of rapid renal function decline in adjusted models compared to 23-24 mEq/L. Raphael et al. [24] also reported serum bicarbonate at 28–30 mmol/L was associated with the lowest risk of GFR event or dialysis in the AASK study. Additionally, the large cohort study from Tangri et al. [25] found that U.S patients with non-dialytic CKD stage 3-5 with higher baseline serum bicarbonate was significantly associated with lower composite outcomes including CKD progression and all-cause mortality. Although several studies have identified a relationship between metabolic acidosis and the progression of chronic kidney disease, a definitive consensus on the specific serum bicarbonate level associated with adverse renal outcomes has yet to be reached. Fukasawa et al. [26] reported that within the lower TCO2 group, which was found to be correlated with deteriorating renal composite outcomes, some patients exhibited normal range serum bicarbonate levels ranging from 22 to 25.3 mEq/L. These findings highlight the importance of early intervention before metabolic acidosis becomes clinically evident. Another study reported both lower and higher serum bicarbonate are related to increased all-cause mortality in CKD patients as a significant U-shaped association [27]. Our results have revealed a statistically significant correlation between higher acid levels and kidney failure. However, this finding lacks clinical significance due to the minimal difference in acid levels between the two groups, which is only 0.9 mEq/L. The optimal range for correcting serum TCO2 levels still needs to be determined.
There are many studies reporting the effect of hypoalbuminemia associated with mortality in the CKD population, but few have examined the association of kidney function decline [28-30]. Our study showed a strong predictor of hypoalbuminemia related to rapid eGFR decline. Similar to our results, Lang et al. [30] evaluated the association between serum albumin and longitudinal kidney function decline in 2,598 patients, and they found that serum albumin levels below 3.80 g/dL were significantly higher risk of rapid kidney function decline. Cheng et al [31] demonstrated a significant correlation between higher serum albumin levels and a notable reduction in the risk of renal composite endpoint as well as a decrease in the rate of annual estimated glomerular filtration rate (eGFR) decline. Interestingly, the relationship between serum albumin and renal function decline is characterized by a non-linear pattern with the lowest log relative hazards and the annual rate of eGFR decline was observed within the range of 4.1-4.3 g/dL. To our knowledge, the mechanisms of this association could be explained by several reasons: the chronic inflammatory process in CKD, malnutritional status, or heavy albuminuria causing hypoalbuminemia. In our population, proteinuria is not the cause of these observed relationships because of low baseline and follow-up albuminuria. Unfortunately, our study did not provide data on inflammatory markers and other nutritional assessments to determine the impact of these variables. We believe that there is a multifactorial cause of the relationship between hypoalbuminemia and rapid eGFR decline including the process of inflammation, co-morbidity, poor nutrition, and atherosclerosis.
It is well known that NSAIDs have been proven as nephrotoxic agents, having chronic effects on renal function in long-term exposure [32-35]. Unsurprisingly, our study found the same relationship between NSIAD exposure and developing rapid kidney function decline. Nevertheless, it is arguable that the risk of rapid chronic kidney progression was largely dependent on the dosage used as metanalysis reported by Nderitu et al. [32]. They found that high-dose NSAID usage was related to a greater risk of CKD progression with pooled OR of 1.26 but no effect of regular-dose usage. However, the definition of a high dose is unclear, and we have no information on cumulative dosage use in our cohort, we recommended that NSAIDs should be avoided in CKD patients with eGFR less than 30 ml/min/per 1.73 m2 and to limit prolonged usage in those with an eGFR between 30 and 59 ml/min per 1.73 m2 according to KDIGO consensus guidelines [5].
Of interest, chronic kidney disease patients who received ACEI therapy are associated with rapid worsening of kidney function. We hypothesize that the benefit of the renal outcome of ACEI use is limited to the anti-proteinuria effect but not to advanced ischemic CKD. This hypothesis is supported by previous studies that reported a significant decline in eGFR in advanced CKD patients treated with RAS (renin, angiotensin aldosterone) inhibitors [35, 36]. Post-hoc analysis of REIN trial results that the renoprotection effect was shown in patients with heavy proteinuria [37]. This is dissimilar to our cohort that most cases had proteinuria less than 1 gm/24 hr. However, discontinuation of RAS inhibitors should be a concern since this may worsen CV events. The physician should balance the potential risk of complications against the potential benefits in each case.
There are several limitations that need to be discussed. Firstly, we used indirect measurement of GFR by CKD-EPI using only serum creatinine. This may not be accurate and can lead to under or overdiagnosis of CKD. However, we excluded the population at risk (e.g., low muscle mass, medication, pregnancy). Secondly, some information is not available in our database such as indications of medication use (e.g., ACEIs, ARBs), herbal use, inflammatory markers, or nutrition assessment. Thus, we could not determine some important parameters that may affect CKD progression. Thirdly, the application of this result to other hospital levels or ethnicities should be interpreted with caution due to the difference in patient severity and health behavior.
CONCLUSION
Our patient had a high proportion of rapid kidney function deterioration. Patients with diabetes mellitus, high serum TCO2 during follow-up, low serum albumin during follow-up, NSAIDs, and ACEI usage significantly predict a faster decline of eGFR among the Asian CKD population. This evidence should be important for developing a good healthcare system for CKD management.
AUTHORS' CONTRIBUTIONS
AP, PD, and JC were included in preparing the concept and design. JC, SW, and UB collected data. AP performed data analysis. AP, PD, and JC draft the first article. All authors participated in preparing the final draft of the manuscript, revising the manuscript, and critically for important intellectual contents. All authors have read and approved the content of the manuscript to be published
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Ethics Committee of the Faculty of Medicine, Prince of Songkla University (REC. 64-403-14-4)
HUMAN AND ANIMAL RIGHTS
No animals were used for studies that are the basis of this research. This research was conducted on humans and is in accordance with the Helsinki Declaration of 1975, as revised in 2013 (http://ethics.iit.edu/ecodes/node/3931).
CONSENT FOR PUBLICATION
Written informed consent was obtained from the patients for publication.
STANDARDS FOR REPORTING
STROBE guidelines and methodology were followed.
AVAILABILITY OF DATA AND MATERIALS
The datasets used and/or analysed during the current study are available from the corresponding author [A.P] on reasonable request.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
ACKNOWLEDGEMENTS
The authors would like to express their sincere gratitude to the Ethics Committee and the Hospital for granting approval to conduct this study. Furthermore, special appreciation is extended to all the patients who participated in this research.


