All published articles of this journal are available on ScienceDirect.
6-month Formulations of Androgen Deprivation Therapy for Advanced Prostate Cancer: Effectiveness and Rationale for Extended Dosing
Abstract
Background:
Luteinizing hormone-releasing hormone (LHRH) agonists and GnRH antagonists are the most widely used androgen deprivation therapy to achieve castration levels of serum testosterone (T). Adherence to dosing schedules is important to avoid treatment failure. A recent analysis found a high non-adherence rate of 84% for LHRH agonist injections based on dosing schedules used in pivotal trials.
Method:
Narrative review of LHRH therapy and evaluation of which offers optimal efficacy, safety, and practicality.
Results:
6-month LHRH agonist formulations require fewer appointments for injections than shorter-acting formulations. Therefore, the frequency of late/missed doses and overall non-adherence may be reduced compared to options requiring frequent dosing (e.g., oral therapies and shorter-acting injections). This flexibility may be preferable for patients who live in multiple locations throughout the year, live long distances from clinics, and/or lack access to reliable transportation. 6-month formulations may also have cost benefits compared to shorter-acting doses. Despite similar levels of T suppression during the labeled dosing period, individual 6-month LHRH agonist formulations appear to have unique profiles, e.g., 6-month subcutaneous leuprolide acetate (LA) results in lower T escape rates compared to 6-month intramuscular LA, if dosing is late.
Conclusion:
The efficacy and practicality offered by 6-month LHRH formulations suggest these could reduce opportunities for late injections by requiring fewer office visits and provide greater confidence that efficacy will be maintained should there be extenuating circumstances leading to delays in therapy administration, as experienced during the recent pandemic.
1. INTRODUCTION
Androgen deprivation therapy (ADT) is the standard of care for the management of advanced prostate cancer (PCa) [1]. Luteinizing hormone-releasing hormone (LHRH) agonists and gonadotropin-releasing hormone (GnRH) antagonists, which decrease androgen production from the testes, are the most widely used methods of ADT to achieve castration levels of serum testosterone (T) [1]. Historically, improvements in dosing have focused on progression from shorter- to longer-acting formulations to increase adherence, reduce chemical instability, and avoid bioavailability challenges [2]. Innovations in leuprolide acetate (LA), a well-established LHRH agonist, have followed this trend. Leuprolide acetate was first launched in 1985 in the United States as a daily injection. Monthly intramuscular (IM) depot injections were approved in 1989, followed by 3- and 4-month IM formulations in 2002 and 2003, respectively [3]. The first (1-month) subcutaneous (SC) LA formulation was approved in 2002, with 3-, 4-, and 6-month durations approved by 2004 [4] (Table 1).
| Therapeutic Option | Active Ingredient | Therapy | Route of Administration | Duration of Action (Months) | Needle Gauge |
Dose (mg) | Volume per Dose (mL) |
|---|---|---|---|---|---|---|---|
| LHRH Agonist | Leuprolide Acetate | ELIGARD® | Subcutaneous Injection | 1 | 20 | 7.5 | 0.3 |
| 3 | 20 | 22.5 | 0.4 | ||||
| 4 | 20 | 30 | 0.5 | ||||
| 6 | 18 | 45 | 0.4 | ||||
| LUPRON DEPOT® | Intramuscular Injection | 1 | 23 | 7.5 | 1.0 | ||
| 3 | 23 | 22.5 | 1.5 | ||||
| 4 | 23 | 30 | 1.5 | ||||
| 6 | 23 | 45 | 1.5 | ||||
| Triptorelin | TRELSTAR® | Intramuscular Injection | 1 | 21 | 3.75 | 2 | |
| 3 | 21 | 11.25 | 2 | ||||
| 6 | 21 | 22.5 | 2 | ||||
| Leuprolide Mesylate | CAMCEVI™ | Subcutaneous Injection | 6 | 18 | 42 | 0.4 | |
| Goserelin | ZOLADEX® | Subcutaneous Injection | 1 | 14 | 3.6 | N/A | |
| 3 | 14 | 10.8 | N/A | ||||
| LHRH Antagonist | Degarelix | FIRMAGON® | Subcutaneous Injection | 1 | 27 | 240/80 | 2x3.0, 4.0b |
| Relugolix | ORGOVYX™ | Oral | daily | N/A | 120 | N/A |
b: Initial dose of 2 x 3 mL, followed by monthly maintenance doses of 4 mL N/D=No data; N/A=Not applicable.
While the T level currently accepted as indicative of castration is <50 ng/dL [1], recent evidence suggests that a level of <20 ng/dL is more representative of what is observed following surgical castration [5]. Achieving and maintaining T at this lower level of <20 ng/dL may delay disease progression and further improve survival [6]. Some professional societies and regulatory agencies have updated the recommended T threshold accordingly. For example, the European Association of Urology updated its guidelines in 2014 to recommend T <20 ng/dL during ADT [7], and the Canadian Urological Association encouraged the adoption of ≤20 ng/dL as a new castration threshold [8]. Additionally, draft guidance in 2019 from the FDA stipulated the addition of T <20 ng/dL as a secondary efficacy endpoint in trials of new ADT drugs and the inclusion of these data in labels [9]. Incomplete T suppression with LHRH therapies has been associated with adverse effects on prostate-specific antigen (PSA) kinetics [10], and higher T and PSA concentrations have been correlated with a poor prognosis and increased risk of mortality [11]. Therefore, the ability of LHRH therapies to achieve and maintain T <20 ng/dL should be assessed and considered standard of care.
Adherence to dosing schedules and regular monitoring of PSA/T levels help avoid treatment failure [12], thus potentially improving survival and avoiding increased costs due to greater healthcare utilization as a result of disease progression [13, 14]. Analyses of real-world data have shown that nonadherence to LHRH therapies is common and can negatively impact T suppression, leading to T breakthrough >50/20 ng/dL [12, 15]. However, there is some evidence that T suppression is sustained beyond the labeled dosing interval [16], and subcutaneous leuprolide acetate is associated with a lower risk of T breakthrough than intramuscular leuprolide acetate when dosing is late [15, 17]. The approval of Relugolix, a daily oral LHRH antagonist for advanced PCa, also carries adherence concerns because the patient and caregiver are responsible for ensuring regular daily dosing as per the label. In light of new evidence on optimal T suppression and the unique conditions imposed by the COVID-19 pandemic, clinicians should reassess which LHRH therapies offer the optimal combination of efficacy and practicality. Access to medical care for men with PCa was sharply reduced in 2020 during the COVID-19 pandemic, according to research by Verana Health and the American Urological Association [18]. This manuscript presents support for 6-month formulations as a good choice both during a pandemic and for clinical practice in general.
2. 6-MONTH LHRH FORMULATIONS ARE PROVEN EFFECTIVE FOR T SUPPRESSION
Importantly, 6-month IM and SC LA have similar T suppression efficacy as shorter-acting formulations (1-, 3-, and 4-month) [19]. Efficacy data across 6-month formulations are similar (Table 2), although cross-study comparisons should be viewed with caution due to differences between trials that may impact results (e.g., different patient populations). Over 90% of patients with PCa treated with IM LA achieved T ≤50 ng/dL [20]. Mean T levels decreased from 435 ng/dL at baseline to 16 ng/dL by week 4 and remained ≤11 ng/dL at the end of each treatment cycle (baseline to week 24, week 24 to 48) until the end of the study [20]. A study of SC LA showed that T ≤50 ng/dL was achieved in 99% of participants at month 12 [21], and an additional analysis showed that 93% of participants reached a nadir T ≤5 ng/dL [22]. In a study of patients with advanced PCa treated with triptorelin pamoate, 93% of patients maintained T ≤50 ng/dL from month 2 to month 12 (week 48) [23]. Testosterone suppression data for a recently approved, pre-mixed depot formulation of leuprolide mesylate were similar to the three other 6-month LHRH formulations [24].
| 6-month Therapy | Other Dosing Durations? | Approval Year | Manufacturer | T ≤ 50 ng/dL Data Available/Refs |
T ≤ 20 ng/dL Data Available/Refs |
|---|---|---|---|---|---|
| Intramuscular Leuprolide Acetate | 1-, 3-, 4-month | 2004 | Abbvie Inc. | Yes [20] | Yes [49] |
| Subcutaneous Leuprolide Acetate | 1-, 3-, 4-month | 2006 | Tolmar, Inc | Yes [21] | Yes [21] |
| Intramuscular Triptorelin Pamoate | 1-, 3-month | 2010 | Watson Laboratories, Inc. | Yes [23] | Yes [50] |
| Subcutaneous Leuprolide Mesylate | None | 2021 | Foresee Pharmaceuticals | Yes [24] | Yes [24] |
2.1. Late Dosing Negatively Impacts Clinical Outcomes
Fewer office visits, and thus fewer opportunities for late dosing, are significant because late dosing is common in clinical practice and has been shown to negatively impact clinical outcomes [12]. An analysis of 22,860 United States patients with PCa found that 84% of LHRH injections were late according to the “28-day” definition of the month used in therapy labels [12]. 60% and 29% of injections were late by over one week and over two weeks, respectively [12]. The 28-day definition of the month was used in analyses to align with prescribing information but clinicians are more likely to schedule dosing based on calendar months due to insurance coverage. Therefore, an “extended month” definition, which means late dosing after days 32, 97, 128, and 194, was also used as it is likely more reflective of real-world clinical practice.
Late dosing was shown to negatively impact the clinical efficacy of the therapy. Mean T levels across formulations were higher when injections were late (79 ng/dL) in comparison to when they were early or on-time (21 ng/dL) [12] (Fig. 1). Late dosing also increased the risk of T breakthrough across formulations: 27% of T values exceeded 50 ng/dL when dosing was late according to the extended month definition and only 4% of T values exceeded this level when doses were administered early/on-time [12]. 43% of T values exceeded 20 ng/dL for late injections, while only 21% exceeded this level for early/on-time injections [12]. As T levels during ADT appear to correlate with clinical outcomes and mortality risk, it is very important that patients achieve and maintain the lowest possible T levels [11]. While there is some evidence that T-based dosing is associated with a lower relative risk of progression to early castrate resistance than calendar-based dosing, this hypothesis was generated in a retrospective study and should be tested in a randomized trial [25].
2.2. 6-month LHRH Formulations Minimize Office Visits
2.2.1. Fewer Opportunities for Failure of Continuous Effective Testosterone Suppression from Late Injections
Longer-acting formulations require fewer office visits for injections (generally two visits/year for 6-month formulations vs. up to 12 visits/year for 1-month formulations) [15], and may also relieve the labor burden for healthcare providers. Fewer in-person visits translate to less opportunity to miss or be late for a scheduled dosing appointment, thus decreasing the potential for failure of continuous T suppression [15]. On average, the expected number of late injections per year for 6-month formulations (0.6) is lower than for shorter-acting formulations (0.8 for 3- and 4-month; 5.4 for 1-month) [26], confirming that 6-month formulations reduce the potential for late dosing (Fig. 2). Use of a 6-month dose maintains control of ADT in the hands of healthcare providers and reduces the likelihood of nonadherence that may be seen with more frequent dosing, especially with daily oral therapies and injections with shorter durations of action.
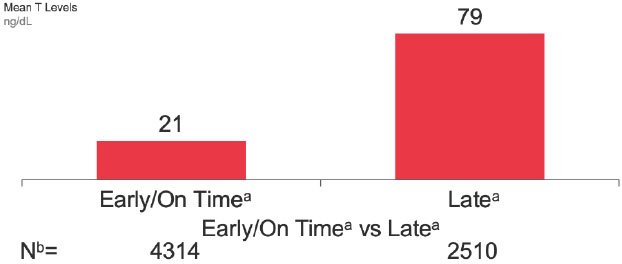
When ADT injections were administered late, mean T concentrations were almost 4-fold greater than those associated with early or on-time injections. These data indicate that late dosing negatively impacts T suppression.
a "Early/On-time" if prior to, or "Late" if on/after day 33 (1-M formulation), 98 (3-M formulation), 129 (4-M formulation), or 195 (6-M formulation).
b Number of patients. Total of 5,933 patients included in this analysis. 891 patients have both early/on-time and late T value.
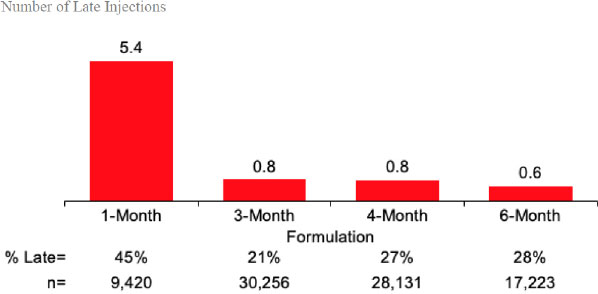
The number of late injections per year was lower for longer-acting formulations than for 1-month formulations. 6-month formulations were associated with the lowest number of late injections per year.
a Expected number is calculated by the proportion of late injections multiplied by the number of injections per year for each formulation.
b“Early/On-Time” if prior to, or “Late” if on/after day 33 (1-M formulation), 98 (3-M formulation), 129 (4-M formulation), or 195 (6-M formulation).
2.2.2. Convenience for Patients During a Pandemic and Beyond
Formulations that reduce the need for in-person appointments may be particularly valuable during a pandemic, as patients will likely be reluctant to visit clinics due to fear of infection, increasing the risk that they will be late for, or miss, their scheduled injections. Although oral medications may allow for fewer in-person appointments, these therapies require consistent daily dosing, and if inadequate, may increase the risk of non-adherence and consequent T escape. A retrospective review of Medicare Part D patients (July 2013-June 2015) who were prescribed oral PCa therapies (abiraterone or enzalutamide) found a mean nonadherence rate of 25% (range: 23%-31%) [27].
Even outside a pandemic environment, fewer in-person appointments may be valuable for patients with limited or seasonal accessibility to clinical practices, such as ‘snowbirds,’ patients with multiple residences, those who lack transportation, patients who live in nursing homes or in remote areas susceptible to inclement weather and/or require long-distance travel to receive medical care. A 2019 Cleveland Clinic survey found that men in the US are reluctant to visit the doctor, with 7% responding that they never visit the doctor and 40% not attending routine check-ups [28]. Therefore, a long-acting formulation addresses the issue of reluctance to keep clinic visits.
Telemedicine tools have been available for some time, and their usage has escalated during the current pandemic. As a baseline, 76% of US hospitals were using telemedicine to connect with patients and over 60% had implemented remote patient monitoring capabilities in 2017 [29]. A recent survey of urology practices reported that >70% of patients with cancer were judged by the treating physician to be eligible for telemedicine [30]. Telehealth visits increased by 154% in March 2020 compared to the same period in 2019, likely due to pandemic-related policy changes and public health guidance [31]. Over 80% of patients expressed a preference for telemedicine consultation during the pandemic and only 3% preferred personal contact with their physician [30]. A separate survey showed that 99% of patients with cancer were satisfied with their telehealth visits and 87% felt that they provided the same level of care as an in-person visit [32]. Although patients with PCa may require frequent “check-ins” in addition to physical visits for injections, telemedicine could substitute for many, but not all, in-person office visits.
Other resources that can provide continuity of care between office visits include oncology nurse navigators and nurses, care partners, and mobile technologies. The inclusion of oncology nurse navigators in multidisciplinary cancer care teams has been shown to improve the quality of care and clinical outcomes [33]. These healthcare professionals have maintained the quality of care during the pandemic by helping to educate providers and patients on telemedicine technologies, scheduling virtual appointments, and facilitating office visits as needed [34]. Home care visits from nurses have also been shown to improve the management of adverse events and decrease anxiety in oncology patients [35]. Empowering nurses and caregivers to engage with patients between clinic visits is an important and complementary way to maintain continuity of care. Mobile health technologies such as smartphone applications can provide patients with ongoing support [36].
Patients receiving 6-month LHRH formulations can also leverage nurses, caregivers, and mobile health technologies to help make important lifestyle changes and manage the side effects of ADT during the periods between clinic visits. These changes could include exercise to combat loss of muscle mass and bone density [37], weight management and a healthier diet [38], counseling to manage sexual issues [39], and quitting smoking [40]. Maintaining normal blood pressure to avoid hypertension [41] and effective treatment of higher cholesterol and diabetes (particularly in conjunction with higher body mass indexes) [42] are also very important for patients with PCa [41, 42]. Using these supplementary resources between twice-yearly injection appointments, rather than between the three to 12 appointments a year required for shorter-acting formulations, will likely be more manageable for patients’ schedules.
Although these resources are also available to patients prescribed shorter-acting formulations and oral therapies, these dosing options do not offer the flexibility or the assurance of dosing adherence that 6-month formulations supplemented with telemedicine tools can provide.
2.3. 6-month LHRH Formulations Options Have Unique Profiles
6-month formulations are not necessarily interchangeable as they have distinct characteristics (Table 1). Subcutaneous LA has a small injection volume of 0.375mL and a short 18-gauge needle [4]. Subcutaneous injection can be performed on multiple sites (e.g., lateral aspect of the lower part of the upper arm, posterior aspect of the upper arms, abdomen, thighs, back lower loins). A pharmacokinetic study of 1-month SC LA vs. 1-month IM LA demonstrated that the duration of quantifiable LA was longer for SC LA compared to IM LA (56 days vs. 42 days), resulting in extended T suppression [17]. This may be due to the difference between IM LA’s lyophilized microsphere delivery system and SC LA’s novel polymeric gel drug delivery system (ATRIGEL® Delivery System). Intramuscular injections are commonly performed at the vastus lateralis, ventrogluteal, and deltoid, which have the potential to cause bone and nerve injury due to needle length [43]. It is recommended that IM injections be avoided in patients who are on anticoagulants due to hematoma risk [44]. Triptorelin pamoate uses a larger needle gauge and employs a unique MIXJECT® system that requires immediate administration after reconstitution of the lyophilized microgranules to avoid separation of the suspension (2mL), which may be inconvenient for clinicians [45]. Comparing triptorelin pamoate and IM LA, triptorelin reduced T levels less rapidly but maintained castration T levels as effectively as IM LA [46]. The recently approved 6-month formulation of leuprolide mesylate is supplied as a pre-filled, pre-mixed emulsion that uses an 18-gauge needle, requires refrigeration, and only has a 6-month formulation [47]. Comparative efficacy and long-term data are not yet available for this new option. Thus, each 6-month LHRH formulation offers a unique combination of features, advantages, and disadvantages, allowing clinicians and patients to select the best option for each individual.
Further distinctions include differences in needle type and injection volume. IM LA uses a finer needle than SC LA, triptorelin pamoate, and leuprolide mesylate, and has an injected volume of 1-1.5mL [3, 4, 45, 47]. However, IM LA suppresses T less effectively than SC LA when dosing is late [15, 17].
It should be noted that all gonadotropin-releasing hormone analogs cause a transient increase in serum T concentrations during the first one to two weeks of treatment [4]. Therefore, potential exacerbations of signs and symptoms of the disease during the first weeks of treatment are of concern in patients with vertebral metastases and/or urinary obstruction or hematuria [4]. In clinical trials for 6-month SC and IM LA, common treatment-related adverse events (TEAE) were hot flush, fatigue, and injection site discomfort [3, 4]. For triptorelin pamoate, the most common TEAE in the clinical trial were hot flush and erectile dysfunction [45]. Common TEAEs observed in the clinical trial for 6-month leuprolide mesylate were hot flush, hypertension, injection site reactions, upper respiratory infection, musculoskeletal pain, pain in the extremities, and fatigue [47].
2.4. 6-month Formulations May Generate Health Care Cost Savings
Total costs associated with 6-month LHRH formulations have been shown to be lower than those for shorter-acting formulations [19]. Analyses performed by Wex et al. showed that in European countries, the average annual cost of 3-month LA formulations was 3%-43% higher than 6-month formulations, and the cost of 1-month LA formulations was 16%-152% higher [19]. This is consistent with a separate study that concluded that 6-month formulations provided the lowest-cost treatment options, compared with shorter-acting formulations, despite the higher per-unit cost (Table 2) [48]. The cost driver in this study was determined to be the reduction in the frequency of required treatments [48]. The lower risk of T breakthrough after late dosing for 6-month SC LA in comparison to IM LA [15] may reduce the risk of treatment failure and avoid additional associated costs. Longer-acting formulations may also provide some level of protection to patients against the impact of unexpected product shortages.
CONCLUSION
The combination of efficacy, safety, and practicality offered by 6-month LHRH formulations may make them a preferred choice for patients and clinicians. 6-month formulations require fewer office visits for injections, which translates to fewer opportunities for missed or late dosing. As even short delays in scheduled dosing may lead to significant increases in T levels, longer formulations with demonstrated T suppression should be considered during the current pandemic and beyond. 6-month doses may also be preferred to oral therapies and injections with shorter durations of action because they allow physicians to be more in control of ADT and lower the risk of nonadherence that could be associated with more frequent dosing. Patients prescribed 6-month formulations can leverage resources including telemedicine tools and oncology nurse navigators to effectively monitor and manage their health between twice-yearly injection appointments. Twice yearly dosing may be particularly beneficial for patients who are ‘snowbirds,’ live far from clinics, lack reliable transportation, live in remote areas susceptible to inclement weather, and/or lack caregivers who can help with therapy adherence. Total healthcare costs associated with 6-month formulations are lower than those associated with shorter-acting formulations. Although approved 6-month LHRH formulations appear to have similar efficacy profiles, each formulation has a distinct combination of key characteristics such as route of administration, needle length and gauge, and injection volume. Taken together, the benefits of 6-month formulations appear to address many of the issues related to adherence and clinical outcomes, especially during a pandemic. Although clinicians may be limited by the type of ADT they have available, clinicians and patients can review all options and select the most appropriate therapy.
LIST OF ABBREVIATIONS
| ADT | = Androgen deprivation therapy |
| GnRH | = Gonadotropin-releasing hormone |
| IM | = Intramuscular |
| LA | = Leuprolide acetate |
| LHRH | = Luteinizing hormone-releasing hormone |
| T | = Serum testosterone |
| PCa | = Prostate cancer |
| PSA | = Prostate-specific antigen |
| SC | = Subcutaneous |
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
Funding support is provided by Tolmar, Inc.
CONFLICT OF INTEREST
The authors declare no conflict of interest financial or otherwise.
ACKNOWLEDGEMENTS
Writing support is provided by Xelay Acumen Group.


