All published articles of this journal are available on ScienceDirect.
Manual Acute PD with Rigid Catheters: A Relook
Abstract
Peritoneal Dialysis (PD) has been an underutilized modality in the treatment of Acute Kidney Injury (AKI). Concerns regarding clearance, fluid removal, infection, complications of therapy, and the hypercatabolic state of AKI has led to PD falling into disrepute. Recent studies have challenged this notion of ineffectiveness. The lower cost, and simplicity of the procedure makes it a particularly attractive option for the developing world which may lack even basic HD facilities, and patients continue to die for want of Renal Replacement Therapy (RRT). We present a review of the available literature about PD in the AKI setting with special reference to the developing world, including the procedure, costs, and effectiveness of the treatment. We also describe the procedure in detail to help ‘hand hold’ physicians interested in performing this lifesaving procedure.
INTRODUCTION
PD as a modality of therapy in AKI continues to evoke mixed reactions from physicians.
Significant controversy exists regarding its indications, adequacy, and effectiveness vis a vis hemodialysis (HD) and the more advanced forms of Continuous Renal Replacement Therapy (CRRT). It is a widely held belief that PD compares poorly versus hemodialysis in the more severe and hypercatabolic states of AKI, and is associated with a significantly higher mortality rate in these situations [1, 2]. This notion has been challenged by the findings of the Brazilian group, which concluded that PD had at least equivalent efficacy [3, 4]. Moreover, recent studies from India and Brazil [5, 6] suggest that PD was comparable to intermittent HD/CRRT with regards to mortality, and also had a salutary effect on early recovery of renal function. Also, when compared to the mortality rates reported from other studies which assessed critically ill patients requiring RRT, the PD and HD groups in these studies performed comparably [7, 8]. Clearly, in resource poor settings, PD offers potential benefits that are hard to overlook. For example, in India there still exist remote rural areas where thousands of people lack even basic medical facilities. Hemodialysis equipped hospitals are concentrated largely in the cities and are mostly operated by the private sector making accessibility and cost a major hindrance. The cost of a single session of in hospital/ICU dialysis in a private hospital in India is around $ 50, and a huge proportion of Indians cannot afford it [9]. The situation is similarly bleak in several parts of the developing world. PD with rigid catheters and manual exchanges, although considered an archaic practice in full-resource centers in the developed world, remains a logical choice in meeting the needs of patients with AKI in resource-poor settings. For example, centers in Nepal [10] and Tanzania [11] have achieved good results with low mortality, by training medical residents to perform Acute PD.
ACUTE PD WITH RIGID CATHETERS -EXPERIENCE FROM THE DEVELOPING WORLD
A recent paper reported the experience of a center in Nigeria [12]. This center in Sub Saharan Africa, representing one of the worlds most impoverished areas, used commendable innovation to deliver cost effective PD to its pediatric AKI population, usually the case mix most in need of intervention. Stylet percutaneous catheters, available in the developing world for as little as $ 8 were used as peritoneal access. In an innovative move, nasogastric catheters were also used as access in a few cases. Nasogastric tubes were modified by making fenestrations at the distal end and inserting a rigid metal probe through one of the fenestrations, thereby acting as a stylet. Initially 10-20 mL/Kg of fluid was used to fill the abdomen, which was later increased to 30-40 mL/Kg. Exchanges were performed manually, and a PD record sheet was used to record the dwell time, inflow and outflow. With this regimen, they were able to achieve survival rates of up to 80%. Peritonitis rates of up to 37% were seen in this study, as expected with the use of percutaneous rigid catheters. Closed PD delivery systems, and tunneled catheters can be used to decrease peritonitis rates [13] but are more expensive. Tenckhoff catheters can be used to prolong the period of dialysis with lower risk of infections [14]. The Nigerian study used commercially available PD fluids to perform the exchanges. However, Intravenous Fluids like Normal Saline, Lactated Ringer’s solution and 5% Dextrose in Water can be suitably modified to freshly prepare fluids for the PD exchanges [15].
ACUTE PD: THE PROCEDURE
Indications: Any patient who has acute kidney injury needing RRT.
Contraindications: Deranged coagulation profile, presence of hernia, distended bowel loops, features of peritoneal adhesions due to repeated intrabdominal surgery or intrabdominal infections like tuberculous peritonitis.
The procedure: Acute PD procedure can be divided into the following four sequential steps:
Step 1- Preparation of the abdomen
Step 2- Priming of the abdomen with fluid,
Step 3- PD catheter insertion and
Step 4- Conducting PD fluid exchanges.
Step 1. Preparation of the Abdomen
The rectum and urinary bladder are to be emptied before the procedure for which rectal enema and evacuation of urinary bladder may be needed. The abdominal wall hair is to be clipped shaved in case of adult males. The skin of all patients should be cleaned initially with chlorehxidine/isoprolyl alcohol or if chlorhexidine unavailable, povidone iodine scrub and later povidone iodine lotion to ensure sterility. The operator should use sterile gloves, gown and mask. The patient should wear a mask over moth and nose.
Step 2. Priming of the Peritoneal Cavity with Fluid
Placement of the priming needle in the correct place is a crucial step in the procedure. Instruments required for priming include long wide bore needle or wide bore lumbar puncture needle, two intravenous (IV) fluid of blood administration sets, surgical blade and PD fluid bottles (or bags), 2 liters in case of adults and 50 ml/kg body weight fluid in case of children (Fig. 1). Most preferred site of PD catheter insertion is in the midline one inch below the umbilicus. The other sites are McBurney’s point on right side or similar position in left iliac fossa. After thorough cleaning of the surface of the abdomen with chlorhexidine/isopropyl alcohol or povidone iodine a sterile central hole towel is placed on the abdomen. The IV fluid transfusion set is connected between the PD fluid and the priming needle. Approximately 10 ml of local anaesthesia is instilled at the proposed site of PD catheter placement and a small nick is made at the site with a surgical blade. Prior to introduction of the priming needle the anterior abdominal wall is to be made taut so as to create an intraperitoneal space. The anterior abdominal wall can be made taut by making the patient raise the head against resistance applied on the forehead by the assisting person at the time of introduction of the priming needle. The needle is to be introduced vertically into the abdominal wall, and the layers of the abdominal wall which are to be pierced are skin, linea alba and finally the parietal peritoneum. The most important phenomenon to be experienced at the time of piercing each layer of abdominal wall is initial resistance followed by a feeling of ‘giving in’. The final ‘giving in’ feeling is felt when the parietal peritoneum is pierced after which one should be careful not to advance the needle further as the intestine might get punctured. Once the priming needle positioning is over, the priming needle is anchored on the surface of abdomen with an artery forceps and the infusion of fluid is commenced. Correct position of the priming needle is ascertained by visualisation of good flow of fluid in the air chamber of the IV set. Wrong placement of the needle tip in the intestinal lumen will result in immediate diarrhea and, urinary bladder puncture will cause sudden increase in ‘urine’ flow which is actually the PD fluid draining out. Hence it is necessary to ascertain absence of intestinal distention and distention of urinary bladder before start of PD.

Prepackaged sterile acute PD kit.
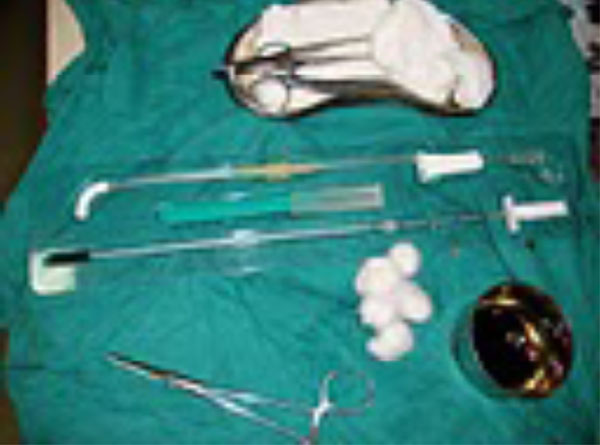
Acute PD kit with disposables during procedure.

Acute PD with stiff catheter, after insertion, ready for exchanges.
Step 3. PD Catheter Insertion
Instruments needed are, PD catheter with stylet, two IV sets, a 3 way connector, PD fluid bottles or bags and drain bag (Figs. 1, 2).
Step 4. PD Exchanges
The three way connector is used to regulate the direction of PD fluid. Each fluid cycle has three components a) PD fluid inflow (10 minutes) b) 35 minutes dwell c) drain time (10 to 15 minutes) (Fig. 3). Maintaining sterile connections, exchanges are done at hourly intervals. If hypercatabolic state or hyperkalemia the dwell time is shortened to 10 minutes for faster correction. If extra ultrafiltration is necessary 25 to 100 ml of high concentration dextrose (25% or50%) is added into each PD fluid bottle/bag before instilling the fluid into the abdomen. Blood glucose needs to be monitored every 4 hours in diabetics and insulin is to be administered as per sliding scale. Hourly inflow, hourly outflow, total inflow and total outflow are tabulated. A minimum of 24 hours and a maximum of 72 hours of PD is done without significant chance of infection. Most non-catabolic AKI recover with this length of Acute PD.
IS PD EFFECTIVE IN AKI? A LOOK AT THE EVIDENCE
The role of PD in AKI continues to be the subject of debate. Only 3 prospective RCTs compared PD with extracorporeal therapies, all from developing regions of the world, and all on ICU patients [1, 3, 5]. Interestingly, manual exchanges with rigid catheters were used in 2 of these studies [1, 5]. The Vietnamese experience [1] concluded that CRRT was significantly superior, while the studies from Brazil and India [3, 5] had similar mortality rates. CRRT performed better in most of the expected areas: solute
SAVING LIVES IN THE DEVELOPING WORLD
Many parts of Sub Saharan Africa, South East Asia and Latin America grapple with a resource crunch, low number and uneven distribution of trained nephrologists, and poor hygiene and sanitation leading to a high incidence of infectious disease [1, 11-13]. This is a situation where the incidence of AKI is bound to be high, and training Physicians in acute PD can go a long way in improving healthcare delivery. Fellowship and training programs of the International Society of Nephrology (ISN), and the International Society of Peritoneal Dialysis (ISPD) are catering to the needs of developing countries. We have had the opportunity to host teams from South East Asian Countries under the aegis of the ISPD, and train them in Acute PD. They are able to perform this therapy with confidence in their respective countries, with gratifying results. The technique may be further refined/adapted to increase safety and acceptability, by using flexible catheters (1 cuffed Tenckhoff) with bedside placement using trocars or Seldinger technique. More institutions should involve themselves in the training programs and help save lives in regions most needing them.
CONCLUSION
Preventable deaths due to AKI continue to occur in the lesser privileged areas of the world. Acute PD is simple, inexpensive, and effective in most forms of AKI. Governments and private institutions should also contribute to enable efficient transfer of skills of this simple technique to practitioners in the direly deficient developing countries and empower them to save patients dying from what is potentially completely curable AKI. Cheap disposables to perform this treatment should also be made easily available.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
Declared none.


