All published articles of this journal are available on ScienceDirect.
Inflammatory Response Variance Based on Quality of Ultrapure Water in Hemodialysis Patients
Abstract
Purpose:
Both the quality of water and biocompatibility of dialyzers are responsible for systemic inflammation in hemodialysis patients. As the biocompatibility of dialyzers has improved, focus for procedural improvements has shifted to water quality. Although ultrapure water reduces the inflammatory response, it is not clear whether different qualities of ultrapure water can further decrease the inflammatory response. This study aims to evaluate the relationship between water quality and the inflammatory response in hemodialysis patients.
Materials and Methods:
We enrolled five patients (two men and three women, mean age 44.6 ± 7.36 years) in maintenance hemodialysis (HD) for three sessions a week. Three quality grades of water were applied in successive weeks: Standard water (N0), single-filter ultrapure water (N1), and double-filter ultrapure water (N2). N2 water was created by mixing N1 water with N1 dialysate and passing the mixture through a second filter. Each patient was exposed to different water quality (N0, N1, N2) for a period of 1 week. Monocyte chemoattractant peptide-1 (MCP-1) mRNA expression in peripheral blood mononuclear cells (PBMCs) was determined by real-time PCR. MCP-1 protein in plasma was measured by ELISA.
Results:
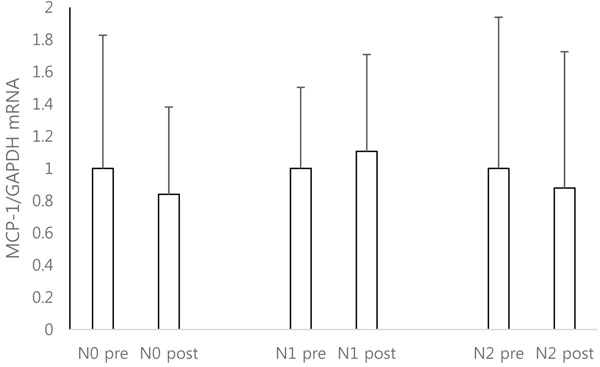
The expression of MCP-1 mRNA in isolated PBMCs after HD was invariant between the N1 and N2 water. The expression of MCP-1 mRNA decreased by 16.1% compared to pre-HD in the N0 system. The expression of MCP-1 mRNA increased by 10.5% in N1 system, and decreased by 12.2% in the N2 system. The water quality did not have a significant impact on MCP-1 protein expression. MCP-1 protein expressions pre- and post-HD were 160.9 ± 13.9 and 153.6 ± 51.6 pg/mL, respectively, when no filter was used (N0). In the case of single-filtered water (N1), the expression levels were 166.6 ± 41.9 and 190.7 ± 88.2 pg/mL pre- and post-HD, respectively. For double-filtered water (N2), the expression levels were 147.8 ± 40.0 and 169.1 ± 52.5 pg/mL pre- and post-HD, respectively.
Conclusion:
There was no difference in MCP-1 response with respect to the number of water filtration steps in HD patients. Further study with especially ultrapure and sterile water is needed to examine the long-term consequence of water purity on inflammatory reactions, and will require more participants in a longer examination window.
1. INTRODUCTION
Inflammatory reactions are a common issue for Hemodialysis (HD) patients [1]. During HD, 200-300 mL of blood per min contacts the dialysis membrane and dialysate for 4 h. The extended exposure inevitably results in some degree of inflammatory response in the patient. Biocompatible dialysis membrane and highly purified water are thought to be able to minimize unwanted inflammatory reactions [2]. Recently, technical developments have allowed the use of biocompatible membrane for most HD patients [3].
Now attention is shifting toward the quality of water used during HD. The quality of purified water is important because patients are exposed to 120 L of dialysate during one session of HD. Standard water used in HD is purified by successive courses of pre-processing, reverse osmosis, post-processing, and distribution. Standard water is more highly purified by a filter installed in the dialyzer before or after being mixed with dialysate, which upgrades the quality of water to ultrapure [4]. Compared to standard water, higher quality water such as ultrapure water has been reported to improve the response to hematopoietics [5, 6], nutritional status [7], hyperlipidemia, and cardiovascular disease [8-10].
There are several clinical studies addressing the quality differences between standard water and ultrapure water in HD. However, it has not been reported whether different qualities of ultrapure water affect the inflammatory response in HD patients. To compare the inflammatory reaction differences between water quality treatments, we compared the expression of MCP-1 mRNA and protein between samples taken from patients treated with HD using traditional ultrapure water and water more highly purified with an additional filter.
2. MATERIALS AND METHODS
2.1. Subjects
Inclusion and exclusion criteria were used to select subjects for this study. We included HD patients between 20 and 60 years old, currently being treated with maintenance HD three times a week, who had a hemoglobin content >9.0 g/dL, serum albumin >3.5 g/L, white blood cell count at 5,000-10,000/uL and low-density lipoprotein cholesterol level of <130 mg/dL. We excluded patients who were scheduled for renal transplantation within a month or had been diagnosed with diabetes mellitus, liver disease, or autoimmune disease. We also excluded patients who had an infectious disease or a history of renal transplantation, and those who were planning pregnancy, were taking immunosuppressant medication, or were enrolled in other clinical trials. Within these parameters, five patients were selected for this study. The Institutional Review Board of Inje University, Ilsan-Paik Hospital approved this study (IB-3-1209-040). The guidelines of the Declaration of Helsinki were followed and all patients provided written informed consent.
2.2. Clinical Procedures
Subjects were dialyzed using the same low flux polyamide dialyzer (Polyflux 14L, Gambro, Sweden). Three types of water quality were investigated: Standard water (no additional filter: N0) and ultrapure water processed with one or two filtration steps (N1 and N2, respectively). In the N1 system, standard water was mixed with dialysate, resulting in ultrapure water. In the N2 system, ultrapure water was mixed with dialysate, and the mixture was treated again through a second filter, resulting in more highly purified ultrapure water. Each patient was exposed to the quality numbers (N0, N1, N2) for 1-week intervals. The N0 and N1 treatments were applied to Phoenix (Gambro, Sweden), and N2 was applied to AK200 (Gambro, Sweden).
2.3. Isolation of Peripheral Blood Mononuclear Cells
A 5 mL blood sample was collected from the subjects before and after each dialysis treatment. Plasma was separated from whole blood and Peripheral Blood Mononuclear Cells (PBMCs) were extracted using Ficoll solution (Biochrom AG, Berlin, Germany). In brief, phosphate-buffered saline and blood concentrates were mixed in a 1:1 ratio within a 50 mL tube and centrifuged at 1400 rpm, 25°C for 25 min. Another 50 mL tube was prepared for gathering buffy coat. After gentle mixing to wash the cells, the tube was centrifuged at 2000 rpm for 3 min to separate cells, and the upper layer was discarded. Separated PBMC pellet was stored at -70°C before mRNA separation and successive experiments.
2.4. Measurement of MCP-1 mRNA
Total RNA was extracted with TRIzol and reverse transcribed using a cDNA synthesis kit (Fermentas, Burlington, Canada) as previously described [11]. Gene expression was measured by quantitative real-time polymerase chain reaction (qRT-PCR). RT-PCR was performed by following a standard three-step cycling condition using SYBR Green Master mix. Primers were designed from the respective gene sequences using Primer3 software. The expression value of MCP-1 mRNA was adjusted by normalizing the data to the house-keeping gene GAPDH before calculation and comparison. The primer sequences used for MCP-1 and GAPDH were; MCP-1 sense 5ʹ GTG AAA CAT TAT GCC TTA A 3ʹ; anti-sense 5ʹ CCA AGT CTC TGT ATC TAA A 3ʹ; GAPDH sense 5ʹ TCT AGG CAC CAA GGT GTG 3ʹ; anti-sense 5ʹ TCA TGA GGT AGT CCG TCA GG 3ʹ.
2.5. Measurement of MCP-1 Protein
The concentration of MCP-1 in plasma was determined with a commercial quantitative sandwich Enzyme-linked Immunosorbent Assay (ELISA) kit (R&D systems, Minneapolis, USA) according to the manufacturer's instructions. Each assay was performed in duplicate, and color intensity was measured with an ELISA reader at 450 nm. MCP-1 concentration is expressed as a proportion of total protein in each sample.
2.6. Statistical Analysis
We used non-parametric analysis because most of the variables were not normally distributed even after logarithmic transformation. The Mann-Whitney U test was used to compare differences between two groups. Statistical significance was defined as p value < 0.05. All statistical analyses were performed using SPSS version 10.0 (SPSS Inc., Chicago, IL, USA). Data are expressed as mean ± standard error.
3. RESULTS
3.1. Basic Characteristics of the Subjects
There were two male and three female subjects. Their mean age was 44.6 ± 7.36 years. The mean vintage on HD was 26.6 ± 8.2 (range 18-40) months (Table 1).
| Characteristics | Mean ± SD |
|---|---|
| Sex | M:F 2:3 |
| Age (years) | 44.6 ± 7.36 |
| Duration of HD (month) | 26.6 ± 8.2 |
| Kt/V | 1.38 ± 0.17 |
| Hemoglobin (g/dL) | 9.7 ± 0.89 |
| White blood cells(/ul) | 6142 ± 1436 |
| Albumin (g/L) | 3.83 ± 0.62 |
| Creatinine (mg/dL) | 10.1 ± 1.73 |
| Total CO2 (mmol/L) | 19.2 ± 9.45 |
| Calcium (mg/dL) | 8.28 ± 0.57 |
| Phosphorus (mg/dL) | 5.56 ± 1.09 |
3.2. MCP-1 mRNA Expression in PBMC
The expression of MCP-1 mRNA in isolated PBMCs after HD was not significantly changed by the number of filtration steps. The expression of MCP-1 mRNA decreased by 16.1% compared to pre-HD in the N0 system. The expression of MCP-1 mRNA increased by 10.5% in the N1 system, and decreased by 12.26% in the N2 system (Fig. 1).
3.3. MCP-1 Protein Expression
MCP-1 protein expression pre- and post-HD was 160.9 ± 13.9 and 153.6 ± 51.6 pg/mL, respectively for the N0 system. In the case of single filtration (N1), the expression was 166.6 ± 41.9 and 190.7 ± 88.2 pg/mL respectively. For double filtration (N2), the expression was 147.8 ± 40.0 and 169.1 ± 52.5 pg/mL respectively. There was no significant difference between the three treatment qualities (Table 2).
| Number of filter | Pre HD | Post HD | P value |
|---|---|---|---|
| N 0 | 160.9 ± 13.9 | 153.6 ± 51.6 | NS |
| N 1 | 166.6 ± 41.9 | 190.7 ± 88.2 | NS |
| N 2 | 147.8 ± 40.0 | 169.1 ± 52.5 | NS |
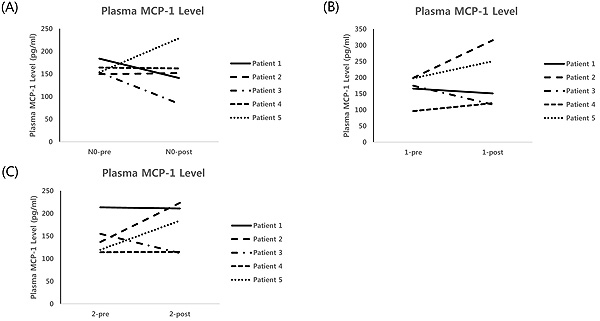
Individual changes in MCP-1 protein expressions pre- and post-HD were evaluated for each patient individually. When no filter was used (N0), the expression increased in one patient, decreased in two patients, and did not change in two patients. In the case of single filtration (N1), the expression increased in two patients and did not change in the remaining patients. For double filtration (N2), the expression increased in one patient, decreased in two patients, and did not change in two patients (Fig. 2).
4. DISCUSSION
Altering the number of water filtration steps did not change the expression of inflammatory marker MCP-1. These findings show that improving ultrapure water with repeated filtration does not guarantee improvement in the inflammation response of hemodialysis patients, at least when measured just before and after HD.
Many studies using different dialysis methods have revealed that they have not made much difference, contrary to expectations. In the HEMO (hemodialysis) study, there was no major benefit to higher dose dialysis and high-flux dialysis than the usual dialysis dose and low-flux dialysis, respectively. High-flux dialysis was only effective in patients having undergone HD for over 3.7 years [12]. In an MPO (membrane permeability outcome) study, increased efficacy was confined to the patients who had serum albumin values less than 4 g/dL and diabetes [13]. In addition, the on-line hemodiafiltration method failed to yield a significantly different prognosis compared with low-flux or high-flux dialysis and was effective only in some patients [14]. The reason for low or no efficacy gain from these high technology dialysis methods could be a difference in water quality used in HD. Dialysate is used for HD after mixing it with water (A solution and B solution). Dialysate mixed with A and B solution becomes higher quality water by passing through a filter in the dialyzer. However, microorganisms, lipopolysaccharides, and short bacterial DNA fragments can remain and evoke inflammatory reactions [15-17]. Unwanted inflammation could be reduced by further filtering. In our study, three different qualities of water produced no difference in MCP-1 expression. Ultrapure water more highly purified through an additional filter (N2) was not effective in reducing MCP-1 expression compared to other qualities of water examined in the study. Currently many institutions are using high-flux dialysis with ultrapure water, whereas sterile water is only used for hemodiafiltration. Studies comparing ultrapure water and sterile water can elucidate whether the quality of water used during HD influences the inflammatory reaction.
There have been reports that higher quality water reduces the inflammatory reaction [18]. Lederer et al., also reported that use of ultrapure water for more than 3 years decreased inflammatory reactions and cardiovascular complications [19]. This effect seems not to occur for a single use but does occur with long-term replacement of high quality water. Di Iorio et al., reported that additional ultrafiltration improved dialysis water purity, reducing inflammation [20]. However, definitive comparison between standard and ultrapure water could be difficult using a low-flux dialyzer because there is less back diffusion or back filtration in low flux dialysis compared with high flux dialysis [21]. The inflammatory reaction difference between standard and ultrapure water needs to be examined with a high flux dialyzer. However, the comparison is not possible because standard water could be harmful in high-flux dialysis.
There are some limitations in this study. First, the small number of patients makes definitive comparison difficult. Second, strict inclusion criteria is not proper to generalize this result to all dialysis conditions. Third, to clarify the difference introduced by water quality, high flux dialysis would have to be used. However, high flux dialysis cannot be applied with standard water due to ethical issue. Fourth, there is a limitation of collecting sample only once before and after HD treatment to avoid unnecessary burden on the patient. If the measurements were taken after a longer term of repeated use of higher quality water, they could produce different results. Fifth, the quality of ultrapure water would be more improved by using two filters. However, the water quality between N1 and N2 systems would not be as significantly different as expected. To clarify the inflammatory effect from water quality, sterile water needs to be included as well as ultrapure water. Finally, additional measurement of other inflammatory marker would support the current result, although the mRNA and protein expressions of MCP-1 were similar,
CONCLUSION
In conclusion, there were no differences in the MCP-1 response in HD patients when the number of filtration steps was increased. Further study with especially ultrapure and sterile water over a longer period and with more participants is needed to clarify whether inflammatory reactions change in response to the water quality.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The Institutional Review Board of Inje University, Ilsan-Paik Hospital approved this study (IB-3-1209-040).
HUMAN AND ANIMAL RIGHTS
Animals did not participate in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2008.
CONSENT FOR PUBLICATION
Informed verbal consent from each study participant was obtained.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
This work was supported by the Gambro Korea in 2013.


