All published articles of this journal are available on ScienceDirect.
A Randomized Controlled Study of Remote Ischemic Preconditioning for the Prevention of Contrast-Induced Nephropathy
Abstract
Introduction:
Remote Ischemic Preconditioning (RIPC) is a technique which applies brief periods of reversible ischemia and reperfusion to limbs provoking adaptive protective responses to distant organs like Heart, Kidneys and Brain.
Methods:
Its efficacy in the prevention of Contrast Nephropathy was tested in our open-label, randomized and sham- controlled study. 100 patients with Chronic Kidney Disease Stages 1-3a requiring Contrast agent for Percutaneous Coronary Interventions were included. Subjects were randomized in to a 1:1 ratio to receive either Remote Ischemic Preconditioning (RIPC) or sham preconditioning.
Results & Discussion:
Both groups were treated with Intravenous saline therapy before contrast exposure. The primary end point was contrast-Induced AKI. Baseline characteristics were comparable in both groups. Mean GFR in ml/min was similar in both groups (RIPC 54.8+/- 9 ml/min; Control 54.8+/- 9 ml/min) . Contrast-induced AKI occurred in 19/50 patients in control group and 6/50 in IPC group (p = 0.005).Hemodialysis was required only in control group (4/50) (p = 0.058). 30 day rehospitalization was more in control group (8 / 50) than RIPC group (1/50) (p = 0.01). RIPC was well- tolerated without adverse effects.
Conclusion:
In conclusion, remote ischemic preconditioning applied before contrast exposure prevents contrast-induced acute kidney injury in CKD 3a. This simple procedure can be added to intravenous saline therapy for nephroprotection.
1. INTRODUCTION
Contrast induced Acute Kidney Injury (CI-AKI) is a serious complication of iodinated contrast media used in Percutaneous Coronary Interventions. Its incidence is variably reported between 1-30% which makes it one of the most common causes of hospital-acquired acute kidney injury [1]. What constitutes a diagnostic threshold of serum creatinine rise in CI-AKI has been a contentious issue. Earlier publications used a cut off value of > 0.5 mg /dl increase of serum creatinine for diagnosing CI-AKI. But recently, in consonance with the internationally accepted definition of AKI according to KDIGO and AKIN criteria, CI-AKI is diagnosed when serum creatinine increases by 0.3 mg /d L within 48 hours of contrast administration in the absence of an alternative explanation [2]. Multiple comorbid factors including Diabetes, old age, hypovolemia and Chronic Kidney disease increase the risk of contrast nephropathy which in turn elevates the risk of one-year mortality [3].
A variety of preventive strategies have been investigated in an effort to decrease the incidence of CI-AKI. Intravenous volume expansion with saline is the only evidence-based recommendation endorsed by the KDIGO for its prevention [4, 5]. Dopamine, fenoldopam, furosemide, mannitol, aminophylline, atrial natriuretic peptide, captopril, calcium channel blockers and alprostadil were not effective in preventing contrast-induced acute kidney injury (CI-AKI). Therefore, novel treatment strategies are required to prevent CI-AKI.
Remote Ischemic Preconditioning (RIPC) is defined as the protection conferred to a distant organ by applying transient brief episodes of ischemia followed by reperfusion in a local organ [4]. Its role in abrogating AKI has been tested extensively in Coronary and Aortic surgery with mixed results. However, several confounding factors are at play when surgery is performed under anaesthesia negating the full benefits of RIPC. We tested the effect of RIPC in the prevention of CI-AKI in patients undergoing coronary angiography through the transradial route.
2. SUBJECTS AND METHODS
This is a prospective, randomized, open-labelled, sham-controlled, single-center study. Inclusion criteria were Age >18 years and < 75 years with an estimated GFR (MDRD formula) between 45- 90 ml/min/1.73 m2.CKD stages >3b,4,5 and those already on dialysis were excluded from the study. Institutional Ethical committee clearance and informed written consent were obtained from the study subjects.
A total of 186 patients from May 2013 to April 2015 for coronary angiography in our Institute were assessed for eligibility. 100 patients were included and 86 patients were excluded. 1:1 Randomization was used in 100 patients so that half of them received Remote Ischemic Preconditioning (RIPC) and the other half of them received sham preconditioning (control group). Serum creatinine was estimated by modified Jaffe kinetic method and eGFR was calculated by MDRD formula. All study patients received intravenous saline infusion (0.9% normal saline) 6 hours prior to 6 hours after contrast exposure at the rate of 1 ml/kg/hour. Iodixanol (Visipaque) which is a nonionic, iso-osmolar contrast agent was used in both the groups. Serum creatinine was estimated by Jaffe Kinetic reaction and the coefficient of variation for its estimation was 2.3%.
One hour before the contrast exposure RIPC was performed. Each cycle of RIPC was done by placing an adult-sized BP cuff in upper arm, inflating it to a level of systolic BP plus 50 mm Hg and sustaining it for 5 minutes before deflation. A total of Four such cycles including two in each arm were completed in the intervention group. Sham procedure was performed by inflating the BP cuff below the diastolic pressure level and then deflating the cuff after 5 minutes.
2.1. Primary Outcome
Primary outcome was Contrast Nephropathy which was defined as an increase in serum creatinine by > 0.3 mg /dl within 48 hours of contrast exposure.
2.2. Secondary Outcomes
Absolute and percentage change in serum creatinine from day 0 to day 2 of contrast exposure, duration of hospital stay, requirement of hemodialysis, incidence of rehospitalisation, and one-month mortality following administration of contrast medium.
2.3. Statistical Analysis
Data analysis was done with the help of computer using Epidemiological Information Package (EPI 2010) developed by Centre for Disease Control, Atlanta. Using this software range, frequencies, percentages, means, standard deviations, chi-square, ’t’ value and 'p' values were calculated. ‘T’ test was used to test the significance of difference between quantitative variables and Yale’s and Fisher’s chi-square tests for qualitative variables. A 'p' value less than 0.05 is taken to denote significant relationship. Intention to treat analysis was done.
3. RESULTS
The mean age of subjects in the intervention (RIPC) and the control group was 58.6 +7.3 and 61.0 + 8.6 years, respectively (Table 1). Males constituted 72% and 82% of patient population in both groups, respectively.
Comorbid factors such as diabetes, hypertension, chronic kidney disease, cardiac dysfunction, contrast agent and contrast volume were comparable in both groups. In diabetics, glycemic status was similar in both groups (RIPC- HbA1C 8.3 ± 2% Vs control 8.28 ± 2%) (p- NS). Mean eGFR was similar in both groups (RIPC- 54.8± 9 ml /min vs control 54.9 ±10ml/min) (p- NS).
All patients who were randomized completed the study. On the intention to treat analysis, the primary end point, contrast induced AKI, occurred in 19 (38%) patients in the control group and 6 (12%) in the IPC group (p 0.005) (Table 2).
| Variable | Control (n=50) |
RIPC (n=50) |
P value |
|---|---|---|---|
| Age (yrs) | 58.6 ± 7.3 | 61.0 ± 8.6 | 0.1367 |
| – | – | – | – |
| Men | 41(82%) | 36(72%) | 0.8345 |
| – | – | – | – |
| BMI | 26.32 ± 3.63 | 25.47 ± 2.77 | 0.1936 |
| – | – | – | – |
| Diabetes Mellitus | 34(68%) | 35 (70%) | 1.0 |
| Hypertension | 22(44%) | 27(54%) | 0.4236 |
| Prior PCI | 17(34%) | 10(20%) | 0.1765 |
| NYHA Class 3,4 | 12(24%) | 21(42%) | 0.0889 |
| Hypotension | 1(2%) | 4(8%) | 0.181 |
| Contrast medium (ml) | 127.5 ± 61.0 | 123±53 | 0.6944 |
| Baseline Hb.% | 12.94±2.08 | 12.62 ± 2.56 | 0.5002 |
| Baseline eGFR | 54.8 ± 9.65 | 54.95 ± 10.0 | 0.9393 |
| Baseline S.Creatinine | 1.32 ± 0.3 | 1.31 ± 0.28 | 0.7828 |
| Integer CI- AKI Risk score < 5 |
14 (28%) | 15(30%) | |
| 6 – 10 | 24 (48%) | 21(42%) | |
| 11 - 15 | 6(12%) | 8 (16%) | |
| >16 | 1(2%) | 1(2%) |
| Trial Outcome | RIPC group | Control group | ‘p’ |
|---|---|---|---|
|
Primary end point Contrast Induced AKI |
6/50(14%) | 19/50(38%) | 0.0055(S) |
| Secondary endpoint | |||
| S. creatinine at Day 0 (mg/dl) | 1.32 +/- 0.3 | 1.31 +/- 0.28 | 0.7828 (NS) |
| Day 1 | 1.43 +/- 0.35 | 1.48 +/- 0.4 | 0.4776 (NS) |
| Day 2 | 1.32 ± 0.41 | 1.49 ± 0.56 | 0.0879 (NS) |
| Change in creatinine(mg/dl) | -0.002 ± 0.27 | 0.184 ± 0.429 | 0.0104 (S) |
| % of Change | -0.06 ± 21.36 | 13.01 ± 30.29 | 0.0143 (S) |
| CI-AKI in CKD 3a Hemodialysis |
10% 0(0%) |
32% 4(8%) |
0.004 (S) 0.0587(NS) |
| Hospital stay (days) | 5.06 +/- 1.43 | 5.12 +/- 1.56 | 0.8418(NS) |
| Mortality | 0(0%) | 1(2%) | 0.5 (NS) |
| Rehospitalisation | 1(2%) | 8(16%) | 0.0154 (S) |
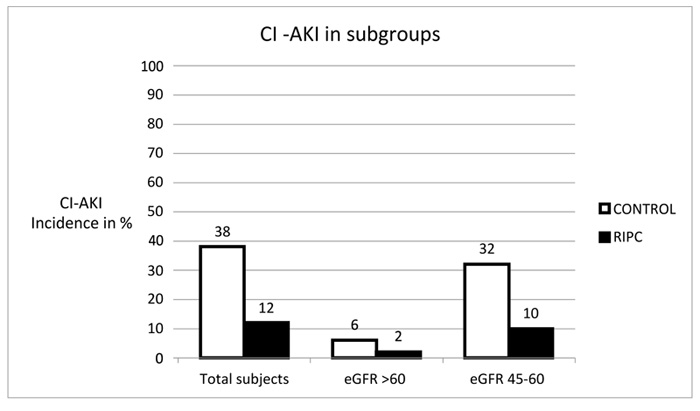
The Primary end point of contrast induced Nephropathy occurred in 14% of RIPC group whereas it was 38% in the control group (p=0.005). After contrast administration, serum creatinine increased significantly within 48 hours in control group (1.49 +0.4 mg/dL) compared with RIPC group (1.32+ 0.5mg/dl) which was statistically significant (p-0.01). In the subgroup of low GFR (45-60 ml/min/m2) incidence of CI AKI was 32% in the control group whereas it was only 10% in RIPC group (p=0.004) (Fig. 1).
Hemodialysis was required in 4 patients in control group (8%) and none from RIPC group. Mortality occurred in one patient only in control group due to complication of cardiac failure and AKI. Rehospitalisation was more in the control group (8/50) (16%) than RIPC group (1/50) (2%) and is significant (p 0.01).The common reasons for hospitalization were dyspnea and chest pain.
No major adverse events occurred in both groups during RIPC and sham- preconditioning respectively. Four patients with RIPC and two patients in control group developed temporary pain and numbness of the limb undergoing ischemia during the procedure.
4. DISCUSSION
Our study is one of the early studies from India with a Randomised Sham- controlled design to investigate the role of Remote Ischemic Preconditioning in the prevention of contrast nephropathy .
Contrast induced renal vasoconstriction affects renal parenchymal oxygenation, especially in the ischemia sensitive renal outer medulla due to release of endothelin and Angiotensin 2. Oxidative stress and Direct tubular damage also contribute to nephrotoxicity. Various treatment strategies have been explored in an effort to decrease the incidence of CI-AKI in patients undergoing contrast media administration. Volume resuscitation with either saline or bicarbonate containing fluid still remains the accepted prophylactic measure to prevent CI- AKI.
The efficacy of Ischemic Preconditioning was first explored in the field of cardiology wherein cardioprotective effects were demonstrated by inducing brief ischemia in the coronary circulation in dogs with myocardial infarction. Subsequently multiple studies studies showed that brief ischemia induced in nontarget tissue confers protection at remote sites such as lung, kidney, intestine, or Brain which lead to the concept of Remote Ischemic Preconditioning (RIPC). The protective mechanism of RIPC is complex and not fully understood. It is hypothesized that RIPC predominantly involves nociceptive, anti-inflammatory and humoral signaling pathways [6].
We followed the KDIGO classification wherein a rise in serum creatinine >0.3 mg /d L is diagnostic of CI -AKI . This improves the sensitivity of diagnosis of CI-AKI. It is at variance with other studies of contrast nephropathy where a rise of serum creatinine > 0.5 mg / dL was required for diagnosis. A cochrane meta analysis which pooled data from 29 studies of coronary artery surgery concluded that RIPC was ineffective in the prevention of major cardiovascular events [7]. However, the same cannot be extrapolated for prevention of contrast nephropathy. The crucial difference is the abrogation of pain pathways in patients under anaesthsia during Surgery. Propofol which is a common anaesthetic agent interferes with neural pathways of protection afforded by RIPC and is a confounding factor which could have nullified the benefits of
RIPC in Surgical patients [8]. Er et al, demonstrated that RIPC reduced the incidence of CI-AKI in high-risk patients with renal dysfunction undergoing Contrast administration. The primary study outcome, CI-AKI occurred in significantly fewer patients in the RIPC group than in the control group (12% versus 40%; P=0.002 [9]. Our results are in agreement with that of Gassinov et al., who showed that RIPC is beneficial in patients at intermediate or high risk, whereas no significant renoprotective effect is seen in low risk
patients with normal renal function [10]. A systematic review of 30 randomized controlled trials was conducted to investigate the effects of RIPC on the incidence and outcomes of AKI. The incidence of AKI in the RIPC group was
11.5%, which was significantly less than the 23.3% incidence in the control group (p = 0.009). The protection against AKI was mainly driven by its reduced incidence in the contrast nephropathy subgroup from 13.5% to 6.5% (P = 0.000) [11]. In the recently published EURO-CRIPS randomized study, RIPC reduced incidence of AKI in Non diabetics undergoing coronary intervention [12]. Finally, a recent Indian study with a protocol similar to ours showed that rise of serum creatinine was significantly less in the group subjected to RIPC compared to the control group [13].
5. STRENGTHS
Clinical methods, outcomes and management were predefined. Intention to treat analysis and zero attrition rate ensured that all patients who were randomized were included in the final analysis.
LIMITATIONS
Its open label design with inherent risks of performance bias.
CONCLUSION
Our study shows that RIPC confers reno protection in patients from stage 1-3 CKD. It can be applied easily and safely. However, larger studies are required to validate our results.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by Meenakshi Mission Hospital and Research Centre, Madurai, India (NBE/CNS/CET-SS/41159/20/1117213/400).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Informed consent was obtained from each participant.
AVAILABILITY OF DATA AND MATERIALS
All relevant data and materials are provided with in manuscript.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


