All published articles of this journal are available on ScienceDirect.
Porosity in Microtomography and Determination of Hounsfield Units do not Predict Calculi Fragmentation in Extracorporeal Lithotripsy: Analysis of an In-vitro Study
Abstract
Background:
Extracorporeal Shock Wave Lithotripsy (SWL) remains one of the most popular methods for treating urinary lithiasis.
Objective:
To evaluate structural and microtomographic characteristics associated with urinary calculi fragmentation in an experimental model of SWL.
Methods:
Samples consisting of at least two calculi fragments obtained from patients were submitted to urological procedures. All calculi were analyzed by X-ray diffraction and a morphological evaluation (weight, length, width, and volume measurements) was conducted along with microtomographic and conventional tomographic assessments in vitro. Thereafter, each sample was submitted to SWL in a ballistic gelatin model. The fragments were separated, using a granulometric sieve, into specimens larger than 4 mm and 2 mm, which were subsequently weighed.
Results:
Altogether, 48 urinary calculi were analyzed and were composed of whewellite (54%), struvite (22%), apatite (11%), uric acid (7%), cystine (4%), and whitlockite (2%). The fragmentation rate was 77.5% and 69.6% for samples > 4 mm and > 2 mm in size, respectively. Factors associated with a reduction in the effectiveness of SWL were volume, dry mass, and width of the calculus. The radiodensity and porosity of the samples evaluated by microtomography were not associated with the fragmentation of calculi.
Conclusion:
The volume, dry mass, and width of urinary calculi were positively correlated with the number of fragments larger than 4 and 2 mm in size obtained post-SWL.
1. INTRODUCTION
Nephrolithiasis is a common disease and despite medical progress, its prevalence has been increasing for the past few years [1]. Low fluid intake and excessive intake of protein, salt, and oxalate are important modifiable risk factors for kidney stones. Approximately 80 percent of kidney stones contain calcium and most of these stones are composed of calcium oxalate. About 10 percent of stones are composed of uric acid and another 10 percent have phosphate ammonium magnesium (struvite). At least 50 percent of individuals experience another stone within 10 years of the first occurrence [2]. Efforts to prevent stone formation are, therefore, essential.
In some circumstances, active stone removal is necessary. Decisions for upper urinary tract calculi management are based on several general aspects such as stone composition, stone size, and symptoms [2, 3]. Since its introduction in 1980, extracorporeal Shock Wave Lithotripsy (SWL) has become the first therapeutic option in most cases of upper-tract urolithiasis. Over the past 10 years, however, the use of SWL has been declining because it is not as reliably effective as more modern technology, such as ureteroscopy [4, 5].
One of the reasons for which SWL is used less and, supposedly, less effective is its inability to effectively fragment calculi with high-density values, as measured by computed tomography and expressed in Hounsfield Units (HU) [3, 6-8]. However, clinical studies report a large variation in the fragmentation of these calculi using SWL [9-11]. Recently, in vitro studies have applied various methods to analyze the predictive factors of fragmentation [12, 13].
Due to the role of SWL in the treatment of calculi, it is important to investigate predictive factors for the success of this technique. Therefore, the objective of this study was to evaluate in vitro factors associated with the fragmentation of urinary calculi in an SWL experimental model.
2. METHODS
Stone samples were obtained from patients who underwent urological surgery (percutaneous nephrolithotomy, nephrectomy, or open ureterolithotomy) from 2013-2015, at a large university hospital. Calculi that presented at least two fragments, one of them larger than 0.5 cm, were selected. At least one fragment was analyzed by X-ray diffraction with the aim to characterize the composition of the calculus (X-Ray Diffraction Empyrean model, Panalytical brand, Eindhoven, Netherlands). Length and width of the samples were measured with precision calipers, mass (in grams) of the stones was determined using a precision balance, and volume was calculated through the Archimedes' principle, by which the dry and apparent weight of the sample was obtained measuring the weight of this sample underwater. Finally, the density was calculated by dividing dry weight/volume.
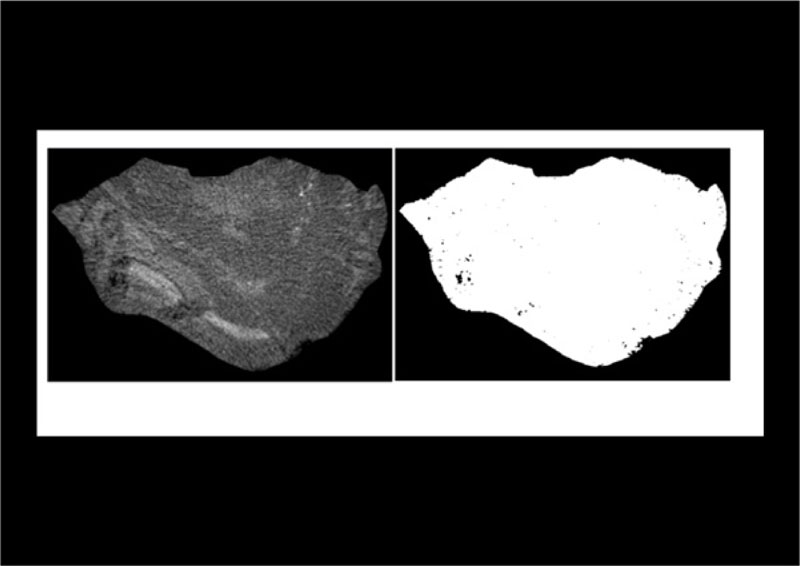
All samples were submitted to micro-computed tomography (micro-CT), using a Skyscan 1172 scanner (Kontich, Belgium). A copper and aluminum filter was used with a voxel measuring 19.8 to 20.3 µm. To assess calculus porosity, we identified an area corresponding to the region of interest, in the micro-CT image, being equivalent to the area of the calculus in each tomographic section. The region of interest was analyzed in a grayscale range from 30 to 255, obtained with the consensus agreement between two independent evaluators based on micro-CT images. The pores were recorded as dark areas outside the grayscale range. The images were analyzed by a CT analyzer software, which measured the porous area (Fig. 1). We analyzed radiodensity by immersing each sample in water for two days, in an acrylic vial, and measuring it using a 64-channel Multidetector CT scanner (Toshiba, Japan). The images were then analyzed with the OsiriX 5.8.2 software (Geneva, Switzerland). The HU was obtained by selecting the region of interest with a circular section in the tomographic image, where the calculus presented the largest diameter.
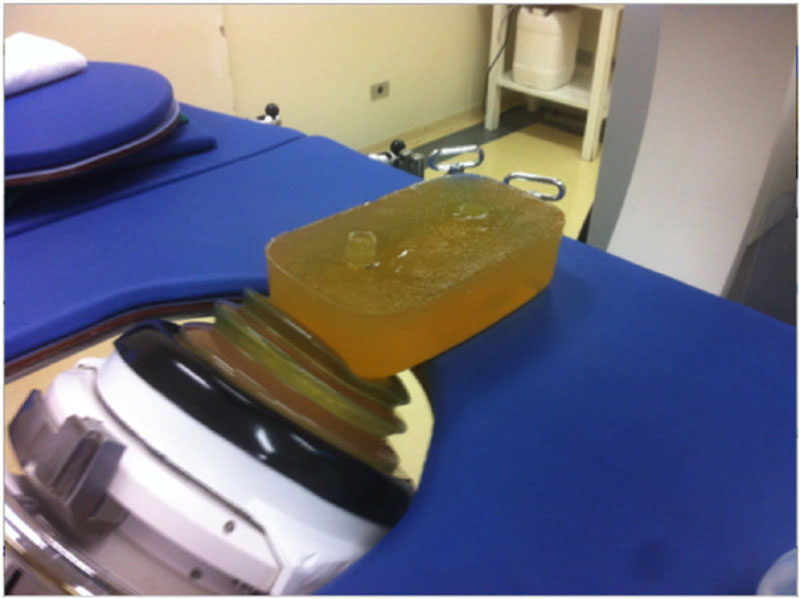
The calculi were placed in ballistic gelatin models made by following protocols previously described [14]. The mold was formed of two cylindrical shaped compartments, measuring 2.5 cm in diameter and 2 cm in depth, with approximately 9 ml volume in each half of the mold, with 7.5 cm from the lateral edge, 6 cm from the terminal edge, and 7 cm from the bottom of the mold. These measurements simulated the approximate skin-calculus distance (Fig. 2). Fragmentation was performed with the S Dornier Lithotripter apparatus (Munich, Germany), by applying 2,000 shock waves with an intensity of 80% of force (17.8 kV) using a frequency of 70 beats/minute. One compartment was used for each calculus sample, since each ballistic gelatin mold was subjected to two lithotripsy procedures on each lateral surface. Radioscopy was used to localize the calculi (Fig. 3).
At the end of each procedure, the interface between the gelatin and the bubble of the lithotripter was evaluated. Stone fragments from each compartment were collected, dried for 72 hours, and passed through a granulometric sieve, and fragments larger than 4 mm and 2 mm were separated. All fragments larger than 4 mm and 2 mm were weighed on a precision scale. The Fragmentation Rate (FR) was evaluated using the weight of the non-fragmented samples (fragments > 4 mm and fragments > 2 mm) using the formula: total weight−non-fragmented weight /total weight.
The results obtained were expressed as mean, median, minimum and maximum values, and standard deviations (quantitative variables) or as frequencies and percentages (qualitative variables). To compare groups classified based on the number of fragments or X-ray diffraction findings, for quantitative variables, the Student’s t-test for independent samples or the non-parametric Mann-Whitney test was used. To evaluate the association between quantitative variables and the number of fragments or fragmentation rates, the Spearman’s correlation coefficient was estimated. The normal distribution of the variables was assessed by the Kolmogorov-Smirnov test. The evaluation of the association between two qualitative variables was performed using Fisher's exact test. P- values <0.05 were considered statistically significant. Data were analyzed with the computer program SPSS Statistics v.20 (IBM, NY, USA).
The study was submitted to the Ethics Committee of Hospital de Clínicas of Federal University of Paraná – BR and was approved.
3. RESULTS
Forty-eight samples were analyzed. The morphological and microtomographic characteristics of the calculus, including the tomographic density (in HU), are shown in Table 1. The X-ray diffraction analysis (n= 45) demonstrated that the calculi were made of whewellite (n= 24, 54%), struvite (n= 10, 22%), apatite (n= 5, 11%), uric acid (n= 3, 7%), cystine (n= 2, 4%), and whitlockite (n= 1, 2%). Porosity of all the calculi measured by micro CT was 4.81±7.25%.
The fragmentation rate was 77.5% and 69.6%, when non-fragmented sample weights greater than 4 mm and 2 mm in size, respectively, were considered. After the fragmentation experiment, 22 samples had at least one fragment > 4 mm in size, and 40 samples had at least one fragment > 2 mm in size. Width, volume, and dry mass of the calculus showed a significant positive correlation with the number of fragments > 4 mm in size (Table 2). Porosity showed a weak, negative correlation with the number of fragments > 4 mm in size after SWL, albeit not statistically significant at the 5% level.

| Variable (n=48) | Mean±SD | Minimum | Maximum |
|---|---|---|---|
| Length of the calculus (mm) | 12.4±2.9 | 8.0 | 20.0 |
| Width of the calculus (mm) | 8.7±1.9 | 4.0 | 15.2 |
| Volume of the calculus (cm3) | 0.28±0.16 | 0.04 | 0.92 |
| Dry mass of the calculus (g) | 0.53±0.31 | 0.07 | 1.64 |
| Density of the calculus (g/cm3) | 1.93±0.20 | 1.47 | 2.39 |
| Porosity of the calculus micro CT (%) | 4.81±7.25 | 0.07 | 38.96 |
| Radiodensity (HU) | 1326±560 | 122 | 2285 |
| r | P | |
|---|---|---|
| Radiodensity | 0.06 | 0.671 |
| Length of the calculus | 0.18 | 0.215 |
| Width of the calculus | 0.33 | 0.023 |
| Volume of the calculus | 0.37 | 0.010 |
| Dry mass of the calculus | 0.37 | 0.011 |
| Density of the calculus | -0.09 | 0.550 |
| Porosity - micro CT | -0.12 | 0.403 |
| Characteristics of the calculus | Number of fragments > 4 mm |
n | Mean±SD | P |
|---|---|---|---|---|
| HU CT in vitro | 0 | 26 | 1277±562 | |
| ≥ 1 | 22 | 1385±565 | 0.512 | |
| Length of the calculus (mm) | 0 | 26 | 11.92±3.22 | |
| ≥ 1 | 22 | 12.87±2.51 | 0.267 | |
| Width of the calculus (mm) | 0 | 26 | 8.03±1.17 | |
| ≥ 1 | 22 | 9.49±2.23 | 0.009 | |
| Volume of the calculus (cm3) | 0 | 26 | 0.22±0.10 | |
| ≥ 1 | 22 | 0.34±0.19 | 0.012 | |
| Dry mass of the calculus (g) | 0 | 26 | 0.43±0.20 | |
| ≥ 1 | 22 | 0.66±0.37 | 0.014 | |
| Density of the calculus (g/cm3) | 0 | 26 | 1.95±0.21 | |
| ≥ 1 | 22 | 1.92±0.19 | 0.578 | |
| Porosity - micro CT (%) | 0 | 26 | 5.38±8.56 | |
| ≥ 1 | 22 | 4.13±5.44 | 0.492 |
| r | P | |
|---|---|---|
| Radiodensity | 0.08 | 0.595 |
| Length of the calculus | 0.37 | 0.011 |
| Width of the calculus | 0.29 | 0.045 |
| Volume of the calculus | 0.49 | <0.001 |
| Dry mass of the calculus | 0.51 | <0.001 |
| Density of the calculus | 0.10 | 0.497 |
| Porosity - micro CT | 0.14 | 0.347 |
When the factors responsible for the presence (n=22) or absence (n=26) of fragments > 4 mm after fragmentation were analyzed, only width, volume, and dry weight of the calculus were able to predict this outcome (Table 3). The length, width, volume, and dry weight of the calculus showed a positive correlation as predictors of residual fragments post-SWL > 4 mm and > 2 mm in size, as shown in Table 4.
Although, as expected, the calculi formed of calcium (whewellite + whitlockite + apatite) had higher radiodensity when compared to that of calculi that did not contain calcium (struvite + uric acid + cystine) in their composition (1,606 ± 431 vs 837 ± 312 HU, p<0.001), we found no significant difference in the fragmentation rates of residual calculi > 4 mm or > 2 mm in size. The same was observed for the number of fragments > 4 mm in size (P=0.22). Finally, calculi were divided according to their radiodensities in vitro into ≥ or < 1000 HU groups. Even after this classification, we found no difference in the fragmentation rate or the number of residual fragments > 4 mm in size.
4. DISCUSSION
In this experimental model of lithotripsy, we observed that the factors related to the lower effectiveness of fragmentation were volume, dry mass, and width of the calculus. The length of the sample was also a predictor of lithotripsy only for fragments > 2 mm, albeit slightly less effective.
The literature varies greatly regarding the effectiveness of SWL for calculi smaller than two centimeters, reporting success rates ranging from 55% to almost 90% [15-23]. Although this technique has been used for a long time, there is still a great deal of discussion on the methods used to optimize its effectiveness. According to Weld et. al. [24], the factors that mostly influence the success of SWL are the characteristics of the calculus as analyzed by computed tomography. A previous in vitro study [12] showed that the morphological findings on micro-CT (homogeneous calculi vs. non-homogenous) might correlate with the fragility of the calculus composed of calcium oxalate. In the present study, there was no correlation between the tomographic density measured in HU and the fragility of the calculi.
Our study used micro-CT to correlate calculus porosity and fragmentation. By assessing porosity, we aimed to objectively measure the findings observed in the microtomography image, where porosity is represented by the empty (or non-crystallized) space within the calculus. Therefore, the larger this space, the greater the heterogeneity in the microtomography image. No correlation between porosity and the fragmentation rate of the calculi was observed post-SWL. However, it should be noted that our study did not analyze calcium oxalate stones exclusively and that the heterogeneous image might also be related to the different compositions of the calculi. In another study, in which micro-CT was used to evaluate the internal structures in brushite calculi [13], no correlation between the fragility of the calculi and the internal structures evaluated was observed. Micro-CT has not yet been used in daily clinical practice. However, as a research technique, it may aid us to understand the microstructure of the calculus and provide some information to develop and assess new imaging methods or to improve existing techniques to study urinary calculi.
A positive correlation between the number of residual fragments and the volume of the calculus was observed. This finding was already demonstrated in previous clinical studies [25, 26]. However, this parameter is not frequently used in clinical practice. This might be due to the difficulty in measuring the volume of the calculus with the software used in clinical practice or the lack of experience of radiologists in obtaining this data, since the calculus does not have a geometric shape that can be easily measured. Since this was an in vitro experiment, the volume of the calculi was measured in our study using the Archimedes' principle and not with images obtained by CT. Another finding correlating with less fragmentation was the dry mass of the calculus. This data can be obtained almost exclusively through this type of in vitro experiments. It is also difficult to correlate this with clinical practice, as it cannot be measured in loco in the urinary calculi.
One intriguing data was the inverse correlation of fragility with the width of the calculus, both for residual fragments > 4 mm or > 2 mm in size; that is, the greater the width, the worse the results of fragmentation. There are no publications that correlate width with successful SWL. In clinical practice, the parameter taken into consideration is the length of the calculus, i.e. the larger measurement. However, there is no consensus over the efficacy of the measurement of length as a predictor of the success of lithotripsy [26]. It is also known that calculus measurements are not symmetrical; therefore, calculi with similar maximum lengths and different widths might have different masses. In this study, the measurement of calculus width was performed using precision calipers. However, this can be performed in a simple manner by any software analyzing tomography images, and thus, making it applicable to clinical settings; however, such assessment requires further investigation.
One limitation of this study is the relatively small number of samples analyzed. A larger number of samples of each type of mineral could reinforce the interpretation of our data. It should also be highlighted that this is an in vitro study. Therefore, clinical extrapolations should be carried out with caution and evaluated further in in vivo studies.
CONCLUSION
Altogether, more simple measures such as volume, width, and dry weight of the calculus correlated with the effectiveness of lithotripsy. Other data, such as the porosity obtained by micro-CT and radiodensity, did not predict the fragmentation rate. The volume and width of the calculi should be further studied as potential predictors of SWL success in a clinical setting.
LIST OF ABBREVIATIONS
| SWL | = Extracorporeal Shock Wave Lithotripsy |
| HU | = Hounsfield Units |
| Micro-CT | = Micro-Computed Tomography |
| FR | = Fragmentation Rate |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Ethics Committee of Hospital de Clínicas of Federal University of Paraná – BR with approval number 337.311.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
All patients participated on a voluntary basis and gave their informed consent.
AVAILABILITY OF DATA AND MATERIALS
The data supporting the findings of the article is available in the repository of the Library of the Federal University of Paraná at http://hdl.handle.net/1884/45312.
FUNDING
None.
CONFLCT OF INTEREST
The authors declare that they have no conflict of interest.
ACKNOWLEDGEMENTS
We thank Dr. Luiz Sérgio Santos, Dr. Maurício Zaparolli and Dr. Luiz Edison Slongo for critical review of the manuscript and helpful discussion.
We thank the Mineralogy Department of the Federal University of Paraná, Brazil, for giving space for our research.


